Advanced Manufacturing of 2-(2-Hydroxyphenyl)-2H-Benzotriazole Derivatives for High-Performance Polymer Stabilization
The global demand for high-performance ultraviolet (UV) stabilizers continues to surge as polymer manufacturers seek to extend the service life of plastics and coatings exposed to harsh environmental conditions. At the forefront of this chemical sector is the synthesis of 2-(2-hydroxyphenyl)-2H-benzotriazole derivatives, a critical class of compounds renowned for their ability to absorb harmful UV radiation and dissipate it as thermal energy. A pivotal advancement in the manufacturing of these essential additives is detailed in Chinese Patent CN1226293C, which outlines a novel preparation method that fundamentally restructures the traditional synthetic workflow. This patent introduces a sophisticated two-step reduction strategy that replaces hazardous, high-temperature ether solvents with a safer, cost-effective biphasic system comprising non-polar solvents and water. For industry stakeholders, this technological shift represents more than just a procedural update; it signifies a major leap forward in process safety, environmental compliance, and overall manufacturing economics, addressing long-standing pain points in the production of high-purity polymer additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benzotriazole UV absorbers has been plagued by significant operational hazards and inefficiencies inherent to older reduction protocols. Conventional methodologies frequently rely on high-boiling ether solvents, such as diethylene glycol diethyl ether or dimethoxyethane, which necessitate reaction temperatures exceeding 100°C, often reaching up to 130°C to drive the reduction to completion. These extreme thermal conditions not only accelerate unwanted side reactions that generate complex impurity profiles but also introduce severe safety risks due to the propensity of ether solvents to form explosive peroxides upon heating. Furthermore, traditional routes typically demand a massive excess of reducing agents, specifically utilizing up to 4 equivalents of zinc powder and equivalent amounts of base, which creates a substantial burden on downstream waste treatment facilities and drastically inflates the cost of goods sold through excessive raw material consumption and hazardous waste disposal fees.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN1226293C pioneers a streamlined biphasic reaction system that operates under significantly milder and safer conditions. By utilizing a solvent mixture composed of inexpensive non-polar hydrocarbons like o-xylene or toluene combined with water and an alkaline compound, the process effectively lowers the required reaction temperature to a manageable range of 60°C to 100°C. This innovative approach eliminates the explosion risks associated with ether peroxides and simplifies the reaction setup by removing the need for complex solvent exchange procedures between steps. The method allows for the direct conversion of the o-nitrophenyl hydroxyphenyl azo-compound into the final benzotriazole structure through a telescoped sequence, thereby minimizing product loss during filtration and crystallization stages while simultaneously reducing the consumption of metallic reducing agents to near-stoichiometric levels.
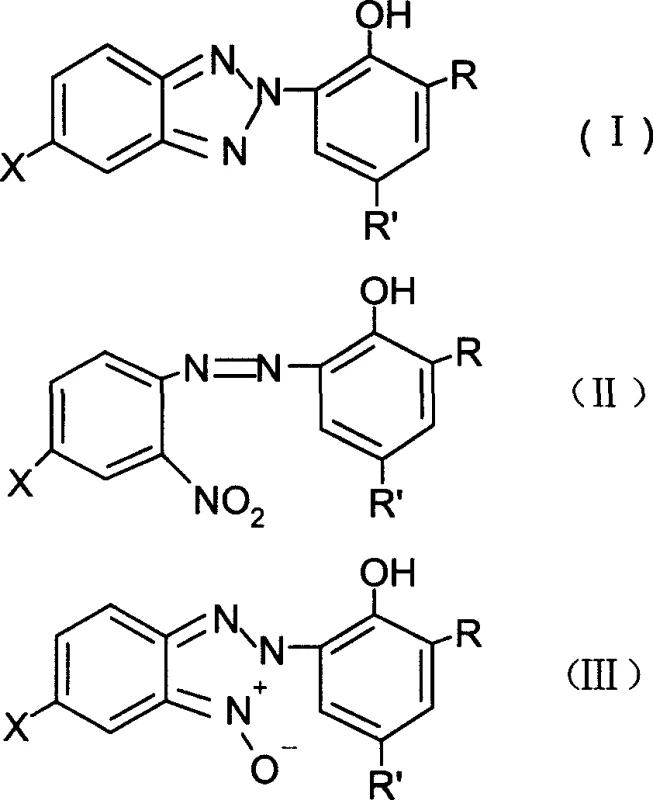
Mechanistic Insights into Two-Step Reduction Cyclization
The core of this synthetic breakthrough lies in its elegant two-step reduction mechanism, which meticulously controls the transformation of the nitro-azo precursor into the stable benzotriazole ring system. The first stage involves the selective reduction of the nitro group in the starting material (Formula II) using hydrazine hydrate as the primary reducing agent within the biphasic solvent system. In this step, the presence of a phase transfer catalyst, such as tetrabutylammonium chloride or resorcinol, plays a critical role in facilitating the interaction between the aqueous hydrazine and the organic-soluble substrate, ensuring high conversion rates to the N-oxide intermediate (Formula III) without over-reduction or decomposition. This controlled environment prevents the formation of azo-hydrazo byproducts that often plague less optimized reduction processes, thereby securing a cleaner reaction profile for the subsequent cyclization step.
Following the formation of the N-oxide intermediate, the process transitions seamlessly into the second reduction phase, where the actual ring closure occurs to form the target benzotriazole structure (Formula I). This step utilizes zinc powder and sulfuric acid added directly to the reaction mixture, leveraging the acidic conditions to promote the intramolecular cyclization of the reduced amine species. The mechanistic efficiency here is highlighted by the drastic reduction in zinc usage; whereas older methods required a four-fold excess to drive the reaction, this optimized protocol achieves complete conversion with only 1 to 1.3 equivalents of zinc powder. This stoichiometric precision not only minimizes the generation of zinc sulfate waste but also simplifies the post-reaction workup, as the removal of unreacted zinc and aqueous byproducts becomes a straightforward filtration and separation task rather than a complex purification challenge.
How to Synthesize 2-(2-Hydroxyphenyl)-2H-Benzotriazole Efficiently
The implementation of this patented methodology offers a robust framework for manufacturing high-purity UV absorbers with exceptional reproducibility and yield. The process begins by charging a reactor with the o-nitrophenyl hydroxyphenyl azo-compound, a non-polar solvent such as o-xylene, water, and a base like sodium hydroxide, creating the essential biphasic medium. Hydrazine hydrate is then introduced dropwise while maintaining the temperature between 60°C and 100°C to effect the first reduction, with reaction progress monitored via HPLC to ensure the complete disappearance of the starting nitro compound. Upon completion, the mixture is neutralized with acid, the aqueous layer is separated, and the organic phase is washed, preparing it immediately for the final cyclization without the need for solvent swapping or intermediate isolation.
- Perform the first reduction by reacting the o-nitrophenyl azo-compound with hydrazine hydrate in a biphasic mixture of non-polar solvent (e.g., xylene), water, and alkali at 60-100°C to form the N-oxide intermediate.
- Neutralize the reaction mixture with acid, separate the aqueous layer, and wash the organic layer to prepare for the second reduction step without solvent exchange.
- Execute the second reduction by adding zinc powder and sulfuric acid directly to the organic phase at 50-100°C to cyclize the intermediate into the final benzotriazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates into tangible strategic advantages that directly impact the bottom line and operational resilience. The shift away from expensive, specialty ether solvents to commodity-grade hydrocarbons like xylene significantly reduces raw material procurement costs and mitigates supply volatility associated with niche chemical feedstocks. Moreover, the elimination of intermediate isolation steps and the reduction in solvent exchange operations streamline the production cycle, leading to faster batch turnover times and increased facility throughput without requiring additional capital investment in new equipment. This operational efficiency ensures a more reliable supply of critical UV absorber intermediates, enabling downstream polymer manufacturers to maintain consistent production schedules even in fluctuating market conditions.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the optimization of reagent stoichiometry and waste management. By reducing the requirement for zinc powder from 4 equivalents to approximately 1.3 equivalents, the process drastically cuts the volume of heavy metal waste generated, which in turn lowers the costs associated with hazardous waste disposal and environmental compliance. Additionally, the use of cheaper, non-polar solvents instead of high-cost ethers, combined with the ability to recycle the organic phase more effectively due to the absence of complex solvent swaps, results in substantial savings in both material input and utility consumption for solvent recovery systems.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the simplified workflow enhances supply chain robustness by reducing the number of unit operations and potential failure points in the manufacturing chain. The telescoped nature of the reaction, where the intermediate is not isolated but carried forward directly, minimizes the risk of product loss during filtration and drying stages, thereby improving overall mass balance and yield consistency. This reliability allows for more accurate forecasting and inventory management, ensuring that customers receive their orders of high-purity polymer additives on time and with the consistent quality specifications required for sensitive applications in automotive and agricultural films.
- Scalability and Environmental Compliance: The safety profile of this method makes it exceptionally well-suited for large-scale commercial production, as it removes the explosion hazards linked to heating ether solvents to high temperatures. Operating at lower temperatures (60-100°C) reduces energy consumption for heating and cooling, contributing to a smaller carbon footprint for the manufacturing facility. Furthermore, the significant reduction in zinc waste and the avoidance of peroxide-forming solvents align perfectly with increasingly stringent global environmental regulations, future-proofing the supply chain against regulatory changes and ensuring long-term operational continuity without the need for costly retrofits or permit modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of how this method outperforms traditional approaches in terms of safety, efficiency, and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the new biphasic solvent system improve safety compared to traditional ether-based methods?
A: Traditional methods often utilize high-boiling ethers like diethylene glycol diethyl ether which pose significant explosion risks due to peroxide formation at temperatures exceeding 100°C. The patented biphasic system employs safer non-polar solvents like xylene or toluene mixed with water, allowing the reaction to proceed at lower temperatures (60-100°C) while eliminating the hazard of peroxide accumulation.
Q: What represents the primary cost-saving driver in this manufacturing process?
A: The primary economic advantage stems from the drastic reduction in reducing agent consumption and waste treatment costs. Conventional processes require excessive amounts of zinc powder (up to 4 equivalents) and base, generating substantial heavy metal waste. This optimized route reduces zinc usage to approximately 1 to 1.3 equivalents, significantly lowering raw material costs and downstream environmental compliance expenses.
Q: Does this process require intermediate isolation between the two reduction steps?
A: No, the process is designed as a telescoped sequence that avoids intermediate isolation. After the first reduction step forms the N-oxide intermediate, the reaction mixture is simply neutralized and washed. The second reduction proceeds directly in the same organic phase, avoiding the product loss and operational complexity associated with solvent swapping and crystallization of the intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Hydroxyphenyl)-2H-Benzotriazole Supplier
As the chemical industry evolves towards more sustainable and efficient manufacturing paradigms, NINGBO INNO PHARMCHEM stands ready to leverage cutting-edge technologies like the one described in CN1226293C to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of UV absorber intermediate meets the exacting standards required by the world's leading polymer and coating manufacturers.
We invite you to explore how our advanced capabilities can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis of 2-(2-hydroxyphenyl)-2H-benzotriazole derivatives can become a cornerstone of your strategic sourcing initiative.
