Optimizing Benzotriazole UV Absorber Production via Novel Two-Step Reduction Technology
The chemical industry constantly seeks more efficient pathways for producing high-performance UV absorbers, specifically 2-(2-hydroxyphenyl)-2H-benzotriazole derivatives, which are critical for stabilizing polymers against degradation. Patent CN1455773A introduces a groundbreaking methodology that addresses long-standing inefficiencies in the synthesis of these vital compounds. By employing a sophisticated two-step reduction strategy, this technology transforms the production landscape from a hazardous, multi-solvent operation into a streamlined, cost-effective process. The core innovation lies in the seamless transition from an o-nitrophenylhydroxyphenylazo precursor to the final benzotriazole ring structure without the need for intermediate isolation. This approach not only enhances the theoretical yield but also fundamentally alters the economic model of manufacturing these specialty chemicals. For global procurement teams and R&D directors, understanding this shift is crucial for securing a reliable benzotriazole supplier capable of meeting stringent quality and volume demands.
Historically, the synthesis of benzotriazole UV absorbers has been plagued by significant technical and economic bottlenecks that hindered scalable production. Conventional methods relying solely on hydrazine reduction often necessitated the use of high-boiling, expensive solvents such as diethylene glycol diethyl ether or dimethyl ether. These solvents not only drove up raw material costs but also introduced severe safety hazards, including the potential for peroxide formation and explosion at elevated temperatures exceeding 100°C. Furthermore, alternative routes utilizing zinc powder frequently suffered from poor atom economy, requiring vast excesses of zinc and alkali metals—often up to 4 equivalents—which created a massive burden on waste treatment facilities. The necessity to switch solvents between reaction stages in older protocols further exacerbated product loss through filtration and crystallization steps, eroding overall process efficiency and profitability.
In stark contrast, the novel approach detailed in the patent data utilizes a hybrid reduction system that elegantly bypasses these historical limitations. The process initiates with a first reduction step using hydrazine hydrate in a biphasic system comprising a low-cost non-polar solvent, such as xylene or toluene, and water. This eliminates the reliance on dangerous ether solvents while maintaining effective reaction kinetics at moderate temperatures between 50°C and 140°C. Crucially, the method allows for a direct telescoping into the second reduction step. Without isolating the N-oxide intermediate or exchanging the solvent system, zinc powder and sulfuric acid are introduced directly into the reaction mixture. This continuity prevents the mechanical loss of product associated with filtration and drying, ensuring that the theoretical yield is maximized while simultaneously simplifying the operational workflow for plant managers.
Mechanistic Insights into Two-Step Reduction Cyclization
The mechanistic pathway of this synthesis offers profound insights into how impurity profiles can be tightly controlled to achieve high-purity polymer additives. The first stage involves the selective reduction of the nitro group on the azo benzene derivative (Formula II) to form the N-oxide intermediate (Formula III). By carefully controlling the addition rate of hydrazine hydrate and maintaining the temperature within the optimal 60°C to 100°C range, the formation of unwanted by-products such as azoxy compounds or over-reduced amines is minimized. The presence of a phase transfer catalyst, such as tetrabutylammonium chloride, can further accelerate this interfacial reaction, ensuring complete conversion of the starting material as monitored by HPLC. This precision in the first step is vital, as residual nitro compounds can interfere with the subsequent cyclization, leading to colored impurities that are difficult to remove in the final application.
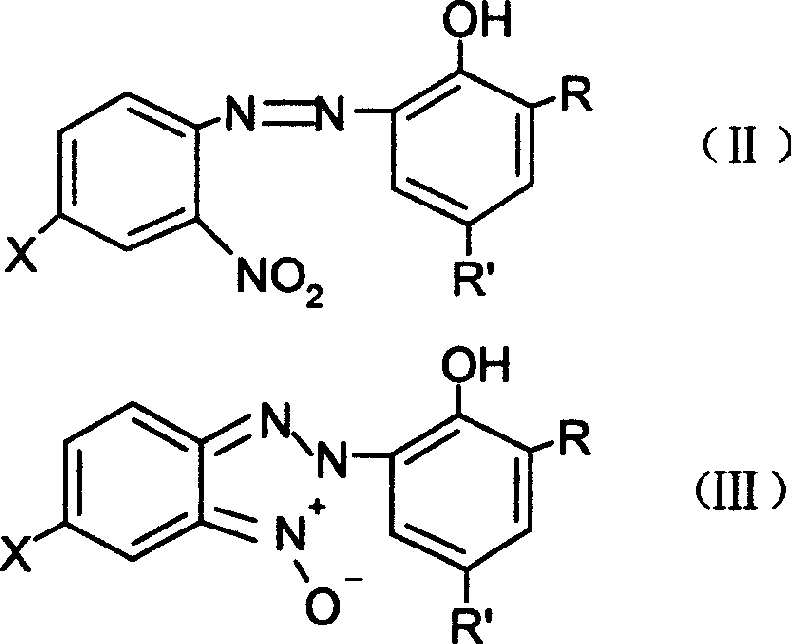
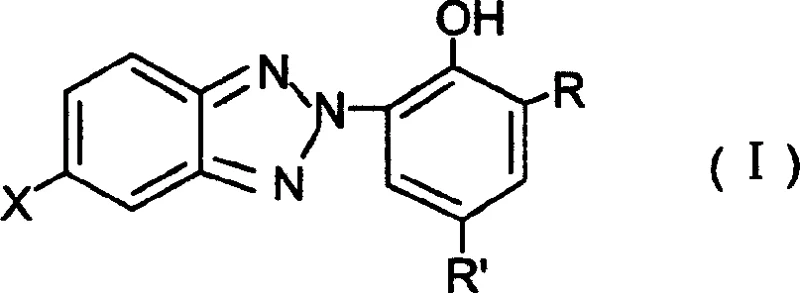
Following the formation of the N-oxide, the second reduction step triggers the intramolecular cyclization that forms the triazole ring. The addition of zinc powder and sulfuric acid creates a localized reducing environment that facilitates the dehydration and ring closure. A key mechanistic advantage here is the ability to perform this reaction in the existing organic-aqueous mixture. The acid neutralizes the base from the first step and activates the zinc surface, promoting the reduction of the N-O bond and the subsequent attack of the nitrogen on the ortho-position to close the ring. Because the solvent system remains consistent, the solubility parameters are maintained, preventing premature precipitation of the intermediate which could trap impurities. This results in a final product, Formula I, that exhibits superior clarity and UV absorption characteristics, meeting the rigorous specifications required for high-end automotive coatings and agricultural films.
How to Synthesize 2-(2-Hydroxyphenyl)-2H-Benzotriazole Efficiently
Implementing this synthesis route requires precise adherence to the reaction conditions outlined in the patent to ensure reproducibility and safety. The process begins by charging the reactor with the o-nitrophenylhydroxyphenylazo starting material, a non-polar solvent like o-xylene, water, and a base such as sodium hydroxide. Hydrazine hydrate is then added dropwise while maintaining strict temperature control to generate the N-oxide intermediate. Once the first reduction is confirmed complete, the reaction mixture is neutralized, and the aqueous layer is separated. Water is reintroduced along with zinc powder, followed by the controlled addition of sulfuric acid to effect the final cyclization. The detailed standardized operating procedures for reagent ratios, addition rates, and workup protocols are critical for maximizing yield and minimizing waste.
- Perform the first reduction by adding hydrazine hydrate to the starting azo compound in a mixture of non-polar solvent, water, and base at 50-140°C to form the N-oxide intermediate.
- Without isolating the intermediate, add water, zinc powder, and sulfuric acid directly to the reaction mixture for the second reduction step.
- Complete the cyclization at 50-100°C, separate the aqueous layer, filter residual zinc, and crystallize the final high-purity benzotriazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages that go beyond simple chemical yield. The elimination of expensive, hazardous ether solvents in favor of commodity chemicals like xylene and water drastically reduces the raw material cost base. Furthermore, the removal of the intermediate isolation step means that the production cycle time is significantly compressed. There is no longer a need for dedicated filtration, drying, and re-dissolution equipment for the intermediate, which frees up reactor capacity and allows for a higher throughput of batches per year. This increased asset utilization directly contributes to a more robust supply chain capable of responding quickly to market fluctuations in demand for UV stabilizers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial reduction in reagent consumption and waste disposal fees. By optimizing the stoichiometry of zinc powder to just 1 to 1.3 equivalents, compared to the 4 equivalents required in legacy methods, the volume of solid waste generated is cut by nearly two-thirds. Additionally, the avoidance of solvent swapping eliminates the energy costs associated with distillation and recovery of multiple solvent types. These cumulative efficiencies result in a significantly lower cost of goods sold, allowing suppliers to offer more competitive pricing without compromising on margin.
- Enhanced Supply Chain Reliability: Reliability in the supply of high-purity UV absorbers is often compromised by complex purification steps that are prone to variability. This streamlined process minimizes the number of unit operations, thereby reducing the number of potential failure points in the manufacturing line. The use of common, readily available solvents like toluene and xylene ensures that supply is not held hostage by the availability of niche specialty solvents. Consequently, lead times for high-purity benzotriazole derivatives can be reduced, providing downstream customers with greater certainty in their own production planning and inventory management.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this method inherently mitigates them. The lower reaction temperatures and the absence of peroxide-forming solvents make the process safer to operate at the 100 MT scale. From an environmental perspective, the drastic reduction in zinc sludge and alkaline waste simplifies effluent treatment, ensuring easier compliance with increasingly stringent global environmental regulations. This sustainability profile is a key differentiator for manufacturers seeking to partner with eco-conscious global brands in the automotive and packaging sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific improvements in safety, yield, and process simplicity documented in the patent literature. Understanding these details helps stakeholders evaluate the feasibility of adopting this method for their specific supply chain needs.
Q: How does this patent method improve safety compared to traditional ether-based solvents?
A: Traditional methods often utilize expensive ethers like diethylene glycol diethyl ether which pose explosion risks due to peroxide formation at high temperatures. This patented method replaces those hazardous solvents with safer, low-cost non-polar solvents like xylene or toluene mixed with water, significantly reducing thermal hazards and operational risks during the reduction phase.
Q: What are the waste treatment advantages of this zinc reduction process?
A: Conventional zinc reduction methods typically require a massive excess of zinc powder and alkali metals, leading to difficult post-treatment and high waste disposal costs. This optimized protocol utilizes a stoichiometric amount of zinc (1 to 1.3 equivalents) and eliminates the need for excess alkali neutralization, thereby drastically simplifying the effluent treatment process and lowering environmental compliance costs.
Q: Does this process require intermediate isolation between reduction steps?
A: No, a key innovation of this technology is the elimination of intermediate isolation. The N-oxide intermediate formed in the first step is directly subjected to the second reduction in the same reactor system without solvent exchange or crystallization. This telescoping of steps prevents product loss through filtrates and reduces overall processing time and equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Hydroxyphenyl)-2H-Benzotriazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this two-step reduction process are fully realized in large-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benzotriazole UV absorber meets the exacting standards required for polymer stabilization. Our commitment to process optimization allows us to deliver consistent quality while adhering to the highest safety and environmental protocols.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your product portfolio. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
