Revolutionizing Proton Pump Inhibitor Synthesis: High-Enantioselective Preparation of (S)-Pantoprazole
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has found a significant breakthrough in the technology disclosed in patent CN102603716A. This patent introduces a sophisticated method for the preparation of (S)-pantoprazole, a critical proton pump inhibitor intermediate, utilizing a highly enantioselective catalytic oxidation process. Unlike conventional synthetic routes that often struggle with racemic mixtures and complex purification steps, this innovation leverages a specific titanium-based chiral catalyst system to directly oxidize the sulfide precursor. The core advancement lies in the ability to conduct this oxidation without the addition of any organic bases, a constraint that traditionally complicates downstream processing. By achieving an enantiomeric excess (ee) value of up to 100 percent, this method sets a new benchmark for optical purity in the manufacturing of gastrointestinal therapeutics. For global procurement teams and R&D directors, this represents a pivotal shift towards more efficient, compliant, and scalable production methodologies for high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of single-enantiomer pantoprazole has been fraught with significant technical and economic challenges that hinder large-scale commercial viability. Traditional approaches often relied on the resolution of racemic pantoprazole, a process inherently limited by a maximum theoretical yield of 50 percent, which is economically inefficient for high-volume manufacturing. Furthermore, existing asymmetric oxidation methods frequently necessitated the use of substantial quantities of special organic bases to facilitate the reaction kinetics. The presence of these bases introduces severe complications during the workup phase, requiring extensive washing and neutralization steps to remove residual amines that could contaminate the final product. Additionally, many prior art methods suffered from inconsistent chiral purity, often yielding products with insufficient ee values that required further, costly recrystallization cycles. The reliance on complex biochemical methods or expensive chiral zirconium complexes also imposed heavy burdens on supply chain stability and raw material sourcing, making the final API cost-prohibitive for generic drug manufacturers seeking to optimize their margins.
The Novel Approach
The methodology outlined in CN102603716A fundamentally disrupts these established paradigms by introducing a streamlined, base-free oxidation protocol. This novel approach utilizes a chiral ligand, specifically (1R, 2S)-1-amino-2-indanol, in conjunction with titanium tetraisopropylate to form a highly active catalytic species in situ. The reaction proceeds smoothly in solvents such as acetonitrile, using cumene hydroperoxide as the oxidant, without the need for auxiliary organic bases. This simplification is not merely a procedural tweak but a transformative change that drastically reduces the complexity of the reaction mixture. By eliminating the base, the formation of emulsions and salt byproducts is minimized, allowing for a much cleaner phase separation during extraction. The result is a robust process capable of delivering (S)-pantoprazole with exceptional optical purity directly from the reactor. This efficiency translates directly into operational excellence, offering a reliable pharmaceutical intermediates supplier pathway that aligns with modern green chemistry principles and cost-reduction mandates.
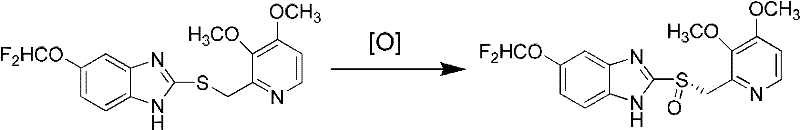
Mechanistic Insights into Titanium-Catalyzed Asymmetric Oxidation
To fully appreciate the technical superiority of this process, one must delve into the coordination chemistry governing the catalytic cycle. The active catalyst is generated through the complexation of titanium tetraisopropylate with the chiral amino-alcohol ligand, creating a rigid chiral environment around the metal center. When the pantoprazole thioether substrate coordinates to this titanium complex, the steric bulk of the indanol moiety effectively blocks one face of the sulfur atom. As the cumene hydroperoxide approaches to transfer an oxygen atom, it is forced to attack from the less hindered trajectory, dictated by the specific stereochemistry of the (1R, 2S)-ligand. This precise spatial control ensures that the oxygen insertion occurs exclusively to form the S-configured sulfoxide, thereby suppressing the formation of the unwanted R-enantiomer. The absence of organic bases is crucial here, as bases can compete for coordination sites on the titanium or alter the electronic properties of the hydroperoxide, potentially degrading the enantioselectivity. By maintaining a neutral environment, the catalyst retains its structural integrity and chiral induction capability throughout the reaction duration.
Furthermore, the impurity profile of the resulting (S)-pantoprazole is significantly improved due to the specificity of this mechanistic pathway. In traditional non-selective oxidations, over-oxidation to the sulfone is a common side reaction that is difficult to separate from the desired sulfoxide. However, the controlled kinetics of this titanium-mediated system favor the formation of the sulfoxide while minimizing further oxidation. The reaction conditions, typically maintained between 10°C and 20°C, provide a thermal window that balances reaction rate with selectivity. Lower temperatures help to freeze out unfavorable transition states that might lead to racemization or byproduct formation. This level of control over the reaction landscape means that the crude product emerging from the reactor already possesses high purity, reducing the load on downstream purification units. For quality assurance teams, this mechanistic robustness offers greater confidence in batch-to-batch consistency, a critical factor for regulatory filings and long-term supply agreements.
How to Synthesize (S)-Pantoprazole Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and optical purity. The process begins with the careful preparation of the catalytic solution, where the stoichiometry between the titanium source and the chiral ligand must be precisely controlled to form the active dimeric or monomeric species. Following catalyst activation, the addition of the oxidant must be performed dropwise to manage the exotherm and prevent local hot spots that could degrade the chiral catalyst. The detailed standardized synthesis steps below outline the critical operational windows for temperature, timing, and reagent addition rates that have been proven to deliver the reported 100% ee values. Operators should note that the exclusion of moisture is paramount, as water can hydrolyze the titanium alkoxide and deactivate the catalyst, leading to poor conversion rates.
- Prepare the chiral catalyst by reacting (1R,2S)-1-amino-2-indanol with titanium tetraisopropylate in acetonitrile at low temperature (0°C to 20°C) for 15 minutes.
- Add pantoprazole thioether and slowly drip cumene hydroperoxide into the reaction mixture, maintaining the temperature between 10°C and 20°C for 2 to 4 hours.
- Quench the reaction with alkaline aqueous solution, extract with organic solvents like ethyl acetate, adjust pH, and recrystallize to obtain pure (S)-pantoprazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this base-free asymmetric oxidation technology offers profound advantages that extend well beyond the laboratory bench. For procurement managers tasked with optimizing the cost of goods sold (COGS), the elimination of organic bases represents a direct reduction in raw material expenditure. Organic amines and specialized bases are often high-cost items that require careful handling and disposal; removing them from the bill of materials immediately improves the gross margin profile of the intermediate. Moreover, the simplified workup procedure—characterized by straightforward extraction and crystallization rather than complex chromatographic separations—drastically reduces solvent consumption and processing time. This efficiency gain allows manufacturing facilities to increase throughput without expanding physical infrastructure, effectively lowering the unit cost of production. The ability to source readily available reagents like titanium tetraisopropylate and cumene hydroperoxide further enhances supply chain resilience, mitigating the risks associated with specialty chemical shortages.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of expensive organic bases and the associated neutralization agents. In traditional methods, the cost of purchasing, storing, and disposing of these bases, along with the acids needed to quench them, adds a significant hidden tax to the manufacturing process. By operating in a base-free environment, the process eliminates these line items entirely. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting material to byproducts like sulfones or racemates, ensuring that a higher percentage of input mass is converted into saleable product. This improvement in atom economy directly correlates to substantial cost savings in raw material procurement, making the final API more competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on exotic or single-source reagents. This novel method relies on commodity chemicals that are widely produced and available from multiple global vendors, reducing the risk of supply disruption. The robustness of the reaction conditions, which tolerate a reasonable range of temperatures and do not require cryogenic cooling below -20°C, means that the process can be easily transferred between different manufacturing sites without extensive re-validation. This flexibility is invaluable for multinational corporations seeking to diversify their supplier base or scale up production rapidly to meet market demand. The simplified purification steps also reduce the dependency on specialized resin columns or chiral HPLC packs, which often have long lead times and limited availability.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding waste generation and solvent usage, this process offers a clear path to greener manufacturing. The reduction in solvent volume required for extraction, due to the cleaner reaction profile, lowers the facility's volatile organic compound (VOC) emissions. Furthermore, the absence of nitrogen-containing organic bases simplifies wastewater treatment, as there is no need for complex biological degradation steps to remove amine residues. This environmental advantage facilitates easier permitting and compliance with increasingly stringent local environmental regulations. The scalability of the process is evidenced by its operation in common solvents like acetonitrile and toluene, which are standard in industrial reactors, ensuring that the transition from pilot scale to multi-ton commercial production is seamless and low-risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-enantioselectivity synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this intermediate.
Q: What is the enantiomeric excess (ee) achieved in this novel synthesis method?
A: According to patent CN102603716A, the method can achieve an ee value of up to 100% for (S)-pantoprazole, significantly surpassing traditional resolution methods.
Q: Why is the absence of organic bases significant in this oxidation process?
A: Eliminating organic bases simplifies the post-reaction workup, reduces the formation of difficult-to-remove salts, and lowers the overall environmental footprint and cost of manufacturing.
Q: What are the critical reaction conditions for optimal yield and purity?
A: Optimal results are obtained using acetonitrile as the solvent, maintaining reaction temperatures between 10°C and 20°C, and utilizing a molar ratio of thioether to ligand to titanium of approximately 1:1:0.5.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Pantoprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality control. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity demonstrated in the lab is maintained at the plant scale. Our facilities are equipped with state-of-the-art reactors capable of precise temperature control and inert atmosphere operations, which are critical for the success of titanium-catalyzed reactions. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch of (S)-pantoprazole meets the 100% ee target, providing our partners with the confidence needed for their own regulatory submissions.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this base-free route for your specific volume requirements. We encourage you to contact us today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments tailored to your project timelines. Together, we can redefine the standards for quality and efficiency in the production of proton pump inhibitor intermediates.
