Advanced Base-Free Asymmetric Oxidation for Commercial Scale-Up of Complex Chiral Sulfoxides
Introduction to High-Enantioselectivity Preparation Technology
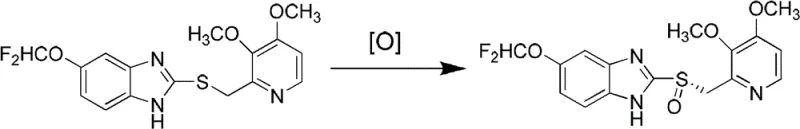
The pharmaceutical industry continuously seeks robust synthetic routes for proton pump inhibitors (PPIs), with (S)-pantoprazole standing out as a critical therapeutic agent for treating gastric acid-related disorders. A pivotal advancement in this domain is documented in patent CN102603716B, which discloses a novel method for preparing (S)-pantoprazole with exceptional enantioselectivity. Unlike conventional approaches that often rely on complex resolution techniques or biochemical transformations requiring specialized equipment, this invention introduces a streamlined chemical oxidation pathway. The core innovation lies in the direct oxidation of 5-difluoromethoxy-2-[[(3,4-dimethoxy-2-pyridyl)-methyl]-sulfenyl]-1H-benzimidazole (pantoprazole thioether) using an oxidant in the presence of a chiral reagent, critically without the addition of any organic bases. This base-free condition is a significant departure from prior art, addressing long-standing issues regarding side reactions and purification difficulties. The patent reports that the ee value of the resulting (S)-pantoprazole can reach an impressive 100 percent, demonstrating the efficacy of this titanium-catalyzed asymmetric oxidation strategy for producing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-enantiomer pantoprazole has been fraught with technical and economic challenges that hinder efficient commercial manufacturing. Traditional methods primarily fall into three categories: chiral resolution of racemates, biochemical oxidation, and asymmetric oxidation requiring basic additives. The chiral resolution route, while conceptually simple, suffers from a theoretical maximum yield of only 50 percent for the desired isomer, necessitating costly recycling processes for the unwanted enantiomer. Furthermore, the lock-out operations involved in splitting racemic mixtures are often laden with trivial details and result in low raw material availability efficiency. Biochemical methods, utilizing microorganisms or enzymes for selective oxidation, present another set of hurdles; they demand special experimental installations and rigorous control over biological parameters, making them relatively too loaded down with trivial details and trouble for large-scale industrial application. Additionally, existing asymmetric oxidation methods often require a large amount of special organic bases to facilitate the reaction. This reliance on bases increases the complexity of cost and operation, as it introduces additional steps for neutralization and salt removal, potentially leading to product degradation or racemization during the workup phase.
The Novel Approach
The methodology outlined in CN102603716B offers a transformative solution by eliminating the need for organic bases during the critical oxidation step. This novel approach utilizes a chiral ligand, specifically (1R, 2S)-1-amino-2-indanol or its enantiomer, in conjunction with titanium tetraisopropylate to form a highly active chiral catalyst in situ. By directly oxidizing the sulfide precursor with cumene hydroperoxide under these specific catalytic conditions, the process achieves high stereoselectivity without the interference of basic species. This simplification of the reaction matrix not only enhances the optical purity of the final product but also drastically simplifies the downstream processing. The absence of organic bases means fewer impurities are generated, and the quenching process becomes more straightforward, typically involving a simple alkaline aqueous wash followed by acidification. This shift represents a significant leap forward in process chemistry, offering a more direct, cost-effective, and environmentally friendlier pathway for the synthesis of high-value chiral sulfoxides used in modern medicine.
Mechanistic Insights into Titanium-Catalyzed Asymmetric Oxidation
The success of this synthesis hinges on the precise formation of a chiral titanium complex that dictates the stereochemical outcome of the oxygen transfer. The mechanism begins with the reaction between the chiral ligand, such as (1R, 2S)-1-amino-2-indanol, and titanium tetraisopropylate. This interaction generates a bitooth ligand titanium complex catalyst, which creates a rigid chiral environment around the metal center. When the pantoprazole thioether substrate coordinates to this titanium center, the bulky chiral ligand sterically hinders one face of the sulfur atom. Consequently, when the oxidant, cumene hydroperoxide, approaches the complex, it is forced to attack the sulfur from the less hindered face. This directed attack ensures that the oxygen atom is transferred with high fidelity to produce the (S)-enantiomer of the sulfoxide. The patent specifies that the molar ratio of the components is critical, with a preferred ratio of pantoprazole thioether to chiral ligand to titanium tetraisopropylate to oxidant being approximately 1:1:0.5:2.32. Maintaining this stoichiometry ensures that the active catalytic species is formed in sufficient concentration to drive the reaction to completion while minimizing non-selective background oxidation.
Impurity control is another vital aspect of this mechanistic framework, particularly concerning the prevention of over-oxidation to the sulfone or the formation of the (R)-enantiomer. The reaction conditions described, specifically operating at temperatures between 10°C and 20°C, are optimized to balance reaction rate with selectivity. Lower temperatures generally favor higher enantioselectivity by reducing the thermal energy available for non-selective pathways, while the specific choice of acetonitrile as a solvent helps stabilize the transition state. The patent data indicates that deviating from these optimal conditions, such as lowering the temperature to -20°C or changing the solvent to dichloromethane without adjusting other parameters, can lead to a decrease in both yield and ee value. Furthermore, the strict exclusion of water during the reaction phase is emphasized, as water can hydrolyze the titanium catalyst, rendering it inactive and leading to poor conversion. The subsequent purification steps, involving pH adjustment to 7.5–8 and extraction with ethyl acetate, are designed to remove residual titanium species and unreacted starting materials, ensuring the final product meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize (S)-Pantoprazole Efficiently
Implementing this synthesis route requires careful attention to the order of addition and temperature control to maximize the efficiency of the chiral catalyst. The process begins with the preparation of the catalyst solution, where the chiral ligand and titanium tetraisopropylate are reacted in an anhydrous solvent like acetonitrile for 10 to 60 minutes. Once the active complex is formed, the pantoprazole thioether is introduced, followed by the slow addition of the oxidant to manage the exotherm and maintain selectivity. The reaction is typically allowed to proceed for 2 to 4 hours at room temperature or slightly below. Following the oxidation, the reaction mixture is quenched with an alkaline aqueous solution, such as ammonia water, to decompose excess oxidant and facilitate phase separation. The detailed standardized synthesis steps, including specific workup procedures like acidification with glacial acetic acid and recrystallization from ethyl acetate and petroleum ether, are essential for achieving the reported 100% ee value and high yields.
- Prepare the chiral catalyst by reacting (1R, 2S)-1-amino-2-indanol with titanium tetraisopropylate in acetonitrile at 10-20°C for 15 minutes.
- Add pantoprazole thioether and slowly drip cumene hydroperoxide into the reaction mixture, maintaining the temperature between 10°C and 20°C for 2 to 4 hours.
- Quench the reaction with alkaline aqueous solution, separate layers, adjust pH to neutrality with acetic acid, extract with ethyl acetate, and recrystallize to obtain pure (S)-pantoprazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this base-free asymmetric oxidation technology presents substantial opportunities for optimizing the supply of critical pharmaceutical intermediates. The elimination of organic bases and the use of common, commercially available reagents like cumene hydroperoxide and titanium tetraisopropylate significantly reduce the complexity of the raw material portfolio. This simplification translates directly into enhanced supply chain reliability, as the risk of bottlenecks associated with sourcing specialized chiral auxiliaries or enzymes is mitigated. Furthermore, the streamlined workup procedure, which avoids complex salt formation and extensive washing steps required by basic conditions, leads to a drastic simplification of the manufacturing process. This efficiency gain allows for faster batch turnover times and reduced consumption of solvents and utilities, contributing to significant cost savings in API manufacturing without compromising on the quality of the final product.
- Cost Reduction in Manufacturing: The removal of organic bases from the reaction protocol eliminates the need for expensive neutralization agents and the associated waste disposal costs. By avoiding the formation of stable salts that require difficult breakdown procedures, the process reduces the consumption of acids and solvents during the purification phase. Additionally, the high enantioselectivity achieved (up to 100% ee) minimizes the loss of material due to the formation of the unwanted (R)-isomer, thereby improving the overall atom economy of the synthesis. This high efficiency ensures that a greater proportion of the starting thioether is converted into the valuable (S)-product, effectively lowering the cost per kilogram of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including titanium tetraisopropylate and cumene hydroperoxide, are commodity chemicals with robust global supply chains. This contrasts sharply with biochemical methods that rely on fragile enzymes or resolution methods that require specific chiral acids which may have limited suppliers. The robustness of the chemical catalyst system ensures that production can be sustained even during fluctuations in the availability of niche reagents. Moreover, the process operates under mild conditions (10°C to 20°C), which reduces the dependency on specialized cryogenic equipment, further enhancing the flexibility and resilience of the manufacturing infrastructure against operational disruptions.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and the absence of hazardous organic bases make this process highly amenable to commercial scale-up. The ability to run the reaction in standard reactors without the need for exotic containment systems facilitates the transition from pilot plant to multi-ton production scales. From an environmental perspective, the reduction in solvent usage and the avoidance of heavy metal catalysts or biological waste streams align with green chemistry principles. This compliance with stringent environmental regulations reduces the regulatory burden on the manufacturing site and ensures long-term sustainability of the supply chain for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific patent technology. These insights are derived directly from the experimental data and claims presented in CN102603716B, providing clarity on the operational parameters and expected outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production lines. The answers reflect the specific advantages of the titanium-aminoindanol catalytic system over traditional methods.
Q: What is the primary advantage of the base-free oxidation method described in CN102603716B?
A: The primary advantage is the elimination of organic bases during the oxidation step, which simplifies the workup procedure, reduces the formation of salt byproducts, and significantly lowers the risk of racemization compared to traditional methods requiring basic conditions.
Q: Which chiral ligand provides the highest enantiomeric excess in this synthesis?
A: According to the patent data, using (1R, 2S)-1-amino-2-indanol as the chiral ligand in combination with titanium tetraisopropylate yields (S)-pantoprazole with an enantiomeric excess (ee) value reaching up to 100% under optimized conditions.
Q: How does this process impact the scalability of pantoprazole production?
A: The process utilizes readily available reagents like cumene hydroperoxide and operates at mild temperatures (10-20°C), avoiding cryogenic conditions. This makes the reaction highly suitable for commercial scale-up, ensuring consistent quality and reduced operational complexity for large-batch manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Pantoprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation proton pump inhibitors. Our technical team has extensively analyzed the potential of the base-free asymmetric oxidation route described in CN102603716B and possesses the expertise to implement this advanced chemistry at an industrial level. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of (S)-pantoprazole. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity analysis via HPLC, to guarantee that every batch meets the highest international standards for pharmaceutical use.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis that demonstrates the economic benefits of switching to this high-efficiency method. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain security and product quality.
