Advanced Manufacturing of Stable Rabeprazole Sodium for Global Pharmaceutical Supply Chains
Advanced Manufacturing of Stable Rabeprazole Sodium for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously demands higher standards for Proton Pump Inhibitors (PPIs), particularly regarding stability and purity profiles which directly impact shelf-life and bioavailability. Patent CN102260244B introduces a groundbreaking methodology for producing a stable Rabeprazole Sodium compound that addresses the chronic instability issues associated with traditional benzimidazole derivatives. This innovation is not merely a incremental improvement but a fundamental shift in how we approach the crystallization of hygroscopic API intermediates. By achieving a moisture content of less than 1‰ and maintaining chemical purity up to 99.9%, this technology offers a robust solution for manufacturers seeking to minimize degradation products during storage and formulation. The strategic importance of this patent lies in its ability to deliver a product with exceptional optical purity (99.96% ee), ensuring consistent therapeutic efficacy while simplifying the quality control burden for downstream drug product manufacturers.
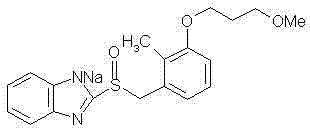
For R&D Directors and technical decision-makers, the structural integrity of the active ingredient is paramount. The provided structural formula illustrates the core benzimidazole scaffold substituted with a methoxypropoxy chain and a critical sulfinyl group, which is notoriously susceptible to oxidation and hydrolysis.  . The novelty of the invention described in CN102260244B is not just the molecule itself, but the specific crystal lattice arrangement achieved through a novel solvent engineering approach. This specific polymorph demonstrates remarkable resistance to environmental stressors, maintaining its physical and chemical properties even under high humidity (92.5% RH) and elevated temperatures. Such stability is crucial for global supply chains where temperature control during logistics cannot always be guaranteed, thereby reducing the risk of batch rejection and financial loss due to stability failures.
. The novelty of the invention described in CN102260244B is not just the molecule itself, but the specific crystal lattice arrangement achieved through a novel solvent engineering approach. This specific polymorph demonstrates remarkable resistance to environmental stressors, maintaining its physical and chemical properties even under high humidity (92.5% RH) and elevated temperatures. Such stability is crucial for global supply chains where temperature control during logistics cannot always be guaranteed, thereby reducing the risk of batch rejection and financial loss due to stability failures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Rabeprazole Sodium has been plagued by significant processing challenges that compromise both yield and product quality. Traditional patents, such as WO03101452, rely heavily on freeze-drying (lyophilization) processes to isolate the sodium salt from aqueous solutions. While effective at removing water, freeze-drying is an extremely energy-intensive operation with a prolonged production cycle, leading to exorbitant operational expenditures and limited throughput capacity. Furthermore, other conventional methods described in patents like US5045552 involve repeated concentration steps to remove solvents, a practice that frequently induces thermal decomposition and discoloration of the sensitive benzimidazole core. These syrupy residues often adhere to reactor walls, causing significant material loss and complicating cleaning validation protocols. Additionally, extraction-based methods utilizing dichloromethane require massive volumes of halogenated solvents and saturated brine to overcome the high water solubility of the sodium salt, creating severe environmental disposal challenges and increasing the complexity of waste management.
The Novel Approach
In stark contrast, the methodology disclosed in CN102260244B circumvents these bottlenecks through a sophisticated anti-solvent crystallization strategy that eliminates the need for freeze-drying or harsh concentration. The process begins by dissolving the crude material in a controlled aqueous system enhanced with a minute quantity of dimethylformamide (DMF), which acts as a co-solvent to ensure complete solubilization without promoting degradation. This solution is then introduced into a pre-cooled mixture of acetonitrile and methyl ethyl ketone, creating a thermodynamic environment that favors the rapid and orderly precipitation of the desired crystal form. This approach not only drastically reduces energy consumption by avoiding high-vacuum distillation and lyophilization but also ensures that the product is isolated as a free-flowing powder rather than a sticky residue. The result is a streamlined workflow that significantly shortens the manufacturing timeline while inherently protecting the chemical integrity of the sulfinyl linkage from hydrolytic attack.
Mechanistic Insights into Controlled Crystallization and Impurity Exclusion
The success of this synthesis route relies on the precise manipulation of solubility parameters and nucleation kinetics to exclude water molecules from the final crystal lattice. By maintaining the reaction mixture at low temperatures (5°C-10°C) throughout the addition and incubation phases, the process suppresses the kinetic energy of the molecules, allowing for the formation of a highly ordered crystal structure with minimal defects. This ordered lattice is inherently hydrophobic enough to reject water inclusion, which is the primary driver of instability in Rabeprazole Sodium. The mechanism effectively traps the sodium ion and the rabeprazole anion in a stable configuration that resists hydration, as evidenced by the Karl Fischer titration results showing moisture levels consistently below 0.2%. This level of dryness is critical because even trace amounts of water can catalyze the decomposition of the sulfinyl group into sulfide or sulfone impurities, which are strictly regulated in pharmaceutical specifications.
Furthermore, the specific solvent ratio of acetonitrile to methyl ethyl ketone (5:3) plays a pivotal role in impurity profiling. This binary solvent system creates a selective precipitation environment where the target compound reaches supersaturation before potential by-products or unreacted starting materials. As the crystals grow over the 15-20 hour incubation period, a phenomenon known as Ostwald ripening occurs, where smaller, less perfect crystals dissolve and redeposit onto larger, more stable crystals. This self-purification mechanism ensures that the final solid possesses a chemical purity of up to 99.9% and an optical purity exceeding 99.9% ee. For R&D teams, this means the process acts as its own purification step, reducing the reliance on additional recrystallization cycles or chromatographic separations, thereby enhancing the overall mass balance and efficiency of the synthetic route.
How to Synthesize Stable Rabeprazole Sodium Efficiently
The implementation of this crystallization technology requires strict adherence to temperature controls and solvent ratios to replicate the high-quality outcomes described in the patent data. The process is designed to be robust yet sensitive to deviations, meaning that precise metering of the anti-solvent and careful monitoring of the cooling ramp are essential for success. Operators must ensure that the initial aqueous phase is fully clarified before introduction to the organic phase to prevent the entrapment of insoluble particulates which could act as unwanted nucleation sites. The following guide outlines the standardized operational parameters derived from the patent embodiments, serving as a foundational protocol for scaling this chemistry from the laboratory bench to pilot plant reactors. Detailed standard operating procedures regarding agitation speeds, filtration rates, and drying endpoints should be developed based on specific equipment configurations.
- Dissolve crude Rabeprazole Sodium in water (5-10 volumes) containing a small percentage of DMF (0.5%-1%), stir for 30 minutes, filter, and cool the filtrate to 5°C-10°C.
- Prepare a mixed solvent system of acetonitrile and methyl ethyl ketone (5: 3 ratio, 18-20 volumes relative to substrate) and cool this mixture to 5°C-10°C.
- Add the cooled aqueous stock solution into the cold organic mixed solvent, maintain incubation for 15-20 hours to allow complete crystallization, then filter and dry to obtain the stable crystal form.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel manufacturing process represents a significant opportunity to optimize total landed costs and mitigate supply risks. The elimination of freeze-drying equipment removes a major capital expenditure barrier and drastically reduces the utility costs associated with long-cycle vacuum operations. By shifting to a standard crystallization workflow, manufacturers can utilize existing general-purpose reactor trains rather than dedicating specialized lyophilizers, thereby increasing asset utilization rates and production flexibility. This operational efficiency translates directly into a more competitive pricing structure for the final API intermediate, allowing buyers to secure high-quality materials without the premium typically associated with complex, low-yield processes. Moreover, the simplified workup reduces the consumption of expensive reagents and solvents, further driving down the variable cost per kilogram of production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of energy-intensive unit operations and the reduction of solvent usage. Traditional methods often require vast quantities of dichloromethane and saturated brine solutions, which incur high procurement costs and substantial waste disposal fees. By replacing these with a recyclable acetonitrile-MEK system, the process significantly lowers the environmental compliance burden and associated taxes. Additionally, the high yield and purity achieved in a single crystallization step minimize the need for reprocessing off-spec batches, ensuring that raw material inputs are converted into saleable product with maximum efficiency. This lean manufacturing approach results in substantial cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Stability is the cornerstone of supply chain reliability, and the superior moisture resistance of this Rabeprazole Sodium crystal form offers distinct logistical advantages. Products manufactured via this method are less susceptible to degradation during transit and storage, reducing the incidence of out-of-specification results upon arrival at the customer's site. This robustness allows for longer shelf-life assignments and reduces the frequency of quality testing required during inventory rotation. For global buyers, this means a lower risk of stockouts caused by batch failures and a more predictable supply cadence. The ability to store the material under ambient conditions without specialized climate control further simplifies warehouse management and reduces overhead costs for both the supplier and the buyer.
- Scalability and Environmental Compliance: From a scalability perspective, the process is inherently designed for large-scale industrial application. It avoids the use of hazardous halogenated solvents like dichloromethane, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. The solvents employed are common in the fine chemical industry and can be easily recovered and reused through standard distillation columns, minimizing the generation of hazardous waste streams. This green chemistry profile not only facilitates smoother regulatory approvals in key markets like the EU and North America but also future-proofs the supply chain against potential bans or restrictions on specific chemical classes. The straightforward nature of the crystallization allows for seamless scale-up from 100 kgs to multi-ton annual production capacities without requiring fundamental changes to the process physics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this stable Rabeprazole Sodium compound. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a transparent view of the technology's capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their finished dosage forms. The answers reflect the consensus on performance metrics such as purity, stability, and process robustness observed across multiple experimental batches.
Q: What distinguishes this Rabeprazole Sodium crystal form from conventional methods?
A: Unlike conventional methods that rely on freeze-drying or repeated concentration which often lead to decomposition and high moisture retention, this patented process utilizes a specific anti-solvent crystallization technique. This results in a crystal lattice with exceptionally low moisture content (less than 1‰) and superior chemical stability under high humidity conditions.
Q: How does this synthesis method impact the optical purity of the final API?
A: The controlled crystallization environment prevents racemization and degradation of the chiral sulfinyl group. The process consistently yields Rabeprazole Sodium with an optical purity exceeding 99.9% ee and chemical purity up to 99.9%, significantly reducing the burden on downstream purification steps.
Q: Is this manufacturing process scalable for industrial production?
A: Yes, the method avoids energy-intensive freeze-drying and complex extraction procedures involving large volumes of halogenated solvents. By using standard crystallization tanks and common organic solvents like acetonitrile and MEK, the process is highly amenable to scale-up from pilot batches to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rabeprazole Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality through expert process engineering. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate crystallization parameters of Rabeprazole Sodium are maintained with absolute precision at every scale. Our facilities are equipped with state-of-the-art stainless steel reactors and rigorous QC labs capable of verifying stringent purity specifications, including the critical moisture content and optical purity metrics defined in CN102260244B. We are committed to delivering a supply of high-purity Rabeprazole Sodium that meets the exacting standards of the global pharmaceutical industry, providing you with a partner who understands both the chemistry and the commerce of API manufacturing.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain to drive value and efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this stable crystal form. We encourage potential partners to contact us directly to obtain specific COA data from our recent pilot batches and to receive detailed route feasibility assessments tailored to your specific volume requirements. Let us collaborate to secure a stable, cost-effective, and high-quality supply of this critical gastrointestinal therapeutic intermediate for your upcoming projects.
