Advanced Biocatalytic Production of (S)-CHBE: A Strategic Upgrade for Statin Manufacturing
Advanced Biocatalytic Production of (S)-CHBE: A Strategic Upgrade for Statin Manufacturing
The pharmaceutical industry's relentless pursuit of efficient, sustainable, and cost-effective synthetic routes for key chiral intermediates has led to significant advancements in biocatalysis. A pivotal development in this domain is documented in patent CN103589665A, which discloses a novel bacterial strain, Rhodococcus qingshengii ZJB-12028, specifically engineered for the asymmetric reduction of ethyl 4-chloroacetoacetate (COBE). This biocatalytic innovation targets the production of ethyl (S)-4-chloro-3-hydroxybutyrate, commonly abbreviated as (S)-CHBE, a critical chiral building block for the synthesis of atorvastatin calcium, the active ingredient in the blockbuster drug Lipitor. The structural integrity and stereochemistry of this molecule are paramount for its biological activity, as illustrated by its specific molecular configuration.
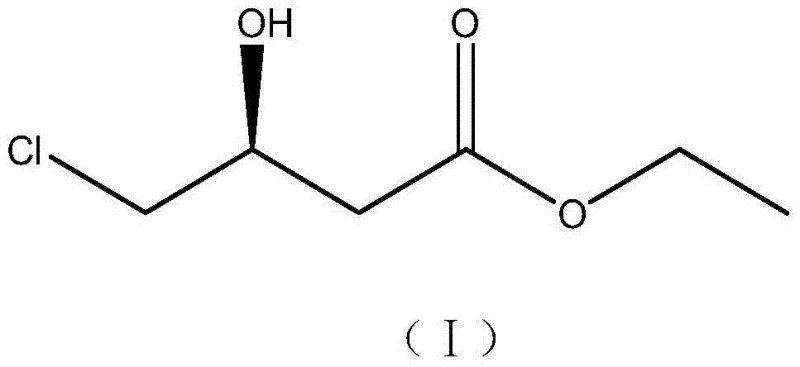
For R&D directors and process chemists, the significance of this patent lies not just in the isolation of a new microorganism, but in the demonstration of a robust, scalable, and highly stereoselective biotransformation process. The strain exhibits exceptional stability and strict stereoselectivity, yielding products with optical purity exceeding 99% e.e. under mild reaction conditions. This represents a paradigm shift from traditional chemical synthesis, offering a greener alternative that aligns with modern environmental regulations and the growing demand for sustainable pharmaceutical manufacturing practices. The ability to produce such high-value intermediates with minimal byproduct formation underscores the industrial potential of this technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-CHBE has relied heavily on chemical catalysis, particularly asymmetric hydrogenation using chiral transition metal complexes. While effective, these conventional methods present substantial logistical and economic challenges for large-scale manufacturing. The most prominent issue is the reliance on expensive noble metal catalysts, such as ruthenium-BINAP complexes, which not only drive up raw material costs but also introduce the risk of heavy metal contamination in the final API. Furthermore, chemical hydrogenation typically requires high-pressure reactors and stringent safety protocols to handle hydrogen gas, necessitating significant capital investment in specialized infrastructure. The separation and purification steps to remove trace metal residues add further complexity and cost to the downstream processing, often requiring additional chromatography or scavenging steps that reduce overall yield.
The Novel Approach
In stark contrast, the biocatalytic approach detailed in the patent utilizes whole cells of Rhodococcus qingshengii as a self-contained catalytic factory. This method operates under ambient pressure and moderate temperatures, typically between 20°C and 45°C, drastically reducing energy consumption and safety hazards associated with high-pressure operations. The use of a biological catalyst eliminates the need for toxic heavy metals, thereby simplifying the purification workflow and ensuring a cleaner impurity profile for the final product. Moreover, the substrate, ethyl 4-chloroacetoacetate (COBE), is readily available and inexpensive, making the overall process economically attractive. The inherent chirality of the enzyme system within the bacteria ensures that the reduction occurs with high specificity, directly yielding the desired (S)-enantiomer without the need for complex resolution steps that limit theoretical yields to 50% in racemic processes.
Mechanistic Insights into Microbial Asymmetric Reduction
The core of this technological breakthrough lies in the enzymatic machinery of the Rhodococcus qingshengii strain, specifically its carbonyl reductase activity. The biotransformation involves the stereoselective reduction of the ketone group in COBE to a hydroxyl group, creating the chiral center essential for statin activity. This reaction is driven by the cofactor NAD(P)H, which acts as the hydride donor. To make this process industrially viable, the patent describes an integrated cofactor regeneration system. Instead of adding stoichiometric amounts of expensive NAD(P)H, the process employs a secondary dehydrogenase enzyme within the cell that oxidizes a cheap cosubstrate, such as glucose, to regenerate the reduced cofactor continuously. This cyclic mechanism allows a catalytic amount of cofactor to drive the conversion of large quantities of substrate.
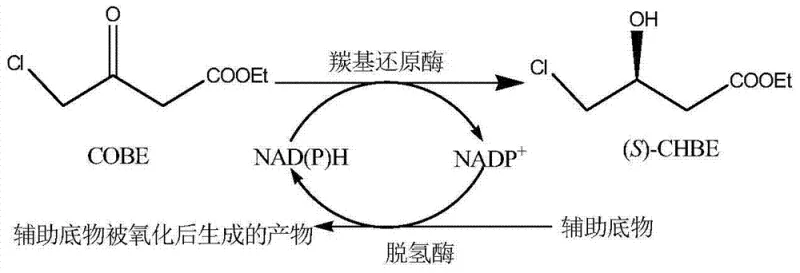
From a mechanistic standpoint, the control of impurities is superior in this biological system compared to chemical alternatives. Chemical reduction can often lead to over-reduction or side reactions at the chloro-group, generating dehalogenated byproducts that are difficult to separate. However, the enzyme's active site provides a specific steric environment that shields the chloro-group while selectively targeting the carbonyl moiety. This precise molecular recognition ensures that the chlorine atom remains intact, preserving the structural integrity required for subsequent coupling reactions in the statin synthesis pathway. The result is a crude product with a significantly cleaner profile, reducing the burden on downstream purification units and increasing the overall mass balance efficiency of the manufacturing line.
How to Synthesize (S)-CHBE Efficiently
The implementation of this biocatalytic route involves a streamlined fermentation and transformation protocol that is amenable to standard pharmaceutical manufacturing equipment. The process begins with the cultivation of the bacterial strain in a nutrient-rich medium to generate sufficient biomass, followed by the harvesting of wet cells which serve as the biocatalyst. These cells are then suspended in a buffered aqueous system containing the substrate and a cosubstrate for cofactor regeneration. The reaction proceeds under gentle stirring at controlled temperatures, after which the product is extracted using organic solvents and purified via distillation. For a detailed, step-by-step technical guide on optimizing fermentation parameters and transformation conditions, please refer to the standardized protocol below.
- Cultivate Rhodococcus qingshengii ZJB-12028 in a fermentation medium containing glucose and yeast extract at 28-30°C to obtain wet cell biomass.
- Prepare a transformation system by suspending the wet cells in a phosphate or citrate buffer (pH 6.0-7.5) and adding the substrate ethyl 4-chloroacetoacetate (COBE).
- Add a cosubstrate such as glucose for cofactor regeneration, maintain the reaction at 28-35°C for 2-4 hours, and isolate the product via extraction and distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this biocatalytic technology offers compelling strategic advantages that extend beyond simple technical feasibility. The shift from precious metal catalysis to renewable biological systems fundamentally alters the cost structure of producing high-purity pharmaceutical intermediates. By eliminating the dependency on volatile markets for rare earth metals like ruthenium, manufacturers can achieve greater price stability and predictability in their raw material sourcing. Furthermore, the simplified downstream processing resulting from the absence of heavy metal contaminants reduces the consumption of auxiliary chemicals and solvents, leading to substantial operational expenditure savings. This efficiency gain is critical for maintaining competitiveness in the generic pharmaceutical market where margin compression is a constant challenge.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of expensive chiral chemical catalysts with self-replicating microbial cells. Since the biocatalyst is produced via fermentation using low-cost carbon sources like glucose and peptone, the unit cost of the catalyst is negligible compared to noble metal complexes. Additionally, the mild reaction conditions eliminate the need for high-pressure autoclaves and the associated maintenance and safety compliance costs. The high conversion efficiency and selectivity mean that less raw material is wasted on byproducts, maximizing the yield per batch and effectively lowering the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of domestically cultivable biological strains rather than imported specialty chemicals. The fermentation-based production of the catalyst can be scaled up rapidly to meet surging demand, providing a flexible buffer against market fluctuations. The robustness of the Rhodococcus qingshengii strain ensures consistent performance across different batches, reducing the risk of production delays caused by catalyst variability. This reliability allows supply chain planners to optimize inventory levels and reduce safety stock requirements, freeing up working capital and improving the overall agility of the supply network in responding to customer orders.
- Scalability and Environmental Compliance: As global regulatory bodies tighten restrictions on pharmaceutical waste and emissions, the environmental profile of a manufacturing process becomes a key license to operate. This biocatalytic route generates significantly less hazardous waste compared to chemical hydrogenation, as it avoids toxic metal residues and operates in aqueous media. The reduced environmental footprint simplifies the permitting process for facility expansions and minimizes the costs associated with waste treatment and disposal. This alignment with Green Chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is increasingly valued by downstream partners and investors in the healthcare sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the fit of this technology within your existing manufacturing portfolio and supply chain strategy.
Q: What is the optical purity of (S)-CHBE produced by this method?
A: The patented process using Rhodococcus qingshengii ZJB-12028 achieves an enantiomeric excess (e.e.) value of greater than 99%, ensuring high stereochemical purity required for statin synthesis.
Q: How does this biocatalytic route compare to chemical hydrogenation?
A: Unlike chemical methods requiring expensive ruthenium catalysts and high-pressure hydrogen, this biological route operates under mild atmospheric conditions using renewable whole-cell catalysts, significantly lowering safety risks and equipment costs.
Q: What substrates are used for cofactor regeneration in this process?
A: The process utilizes inexpensive cosubstrates such as glucose, fructose, or sucrose to regenerate the NAD(P)H cofactor in situ, eliminating the need for external addition of costly reducing agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-CHBE Supplier
The technological potential of Rhodococcus qingshengii-mediated synthesis represents a significant opportunity for optimizing the production of statin intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of (S)-CHBE delivered meets the exacting standards required for API synthesis. We are committed to bridging the gap between cutting-edge biocatalytic research and reliable commercial supply.
We invite you to engage with our technical team to explore how this advanced biocatalytic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits relevant to your operation. We encourage potential partners to contact our technical procurement team to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and our proven track record in delivering high-quality pharmaceutical intermediates.
