Advanced Biocatalytic Production of Chiral Diols: Scaling High-Purity Intermediates for Global Pharma
Advanced Biocatalytic Production of Chiral Diols: Scaling High-Purity Intermediates for Global Pharma
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards greener, more efficient synthetic routes, particularly for high-value chiral intermediates. A pivotal development in this sector is documented in Chinese Patent CN101613666B, which discloses a novel strain of Bacillus sp. ECU0015 (CGMCC No.2874) capable of catalyzing the asymmetric hydrolysis of phenylethylene glycol cyclic carbonate. This biocatalytic breakthrough offers a robust alternative to traditional chemical synthesis, enabling the production of optically active chiral phenylethylene glycol and mandelic acid derivatives with exceptional stereocontrol. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this technology represents a significant advancement in process chemistry, combining high enantiomeric excess with environmentally benign reaction conditions that simplify downstream processing and reduce overall manufacturing complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral diols and their cyclic carbonate precursors has relied heavily on chemical methods involving organometallic catalysts or multi-step roundabout pathways. Traditional chemical synthesis often necessitates the asymmetric dihydroxylation of alkenes or the selective reduction of chiral hydroxy acids, processes that frequently require harsh reaction conditions, expensive chiral ligands, and rigorous exclusion of moisture and oxygen. Furthermore, direct asymmetric carbonylation of epoxides to form chiral cyclic carbonates has historically suffered from low optical purity, often capping at around 70% ee, which necessitates costly and yield-reducing recrystallization steps to meet pharmaceutical grade standards. The reliance on transition metals also introduces significant supply chain risks related to heavy metal residue limits in final API products, forcing manufacturers to implement complex and expensive purification protocols to ensure regulatory compliance.
The Novel Approach
In stark contrast, the novel biocatalytic approach utilizing Bacillus sp. ECU0015 leverages the inherent stereoselectivity of microbial enzymes to achieve superior results under mild conditions. This method employs resting cells or growing cells of the bacterium to catalyze the kinetic resolution or asymmetric hydrolysis of racemic substrates, effectively bypassing the need for toxic metal catalysts. The process operates efficiently in aqueous phosphate buffers at moderate temperatures ranging from 25°C to 40°C and neutral pH levels, significantly reducing energy consumption and equipment corrosion risks compared to high-pressure or cryogenic chemical methods. By directly targeting the cyclic carbonate or acetyl mandelic acid substrates, this biological route streamlines the synthesis workflow, offering a direct path to high-purity chiral building blocks that are essential for the manufacture of beta-blockers and other cardiovascular agents.
Mechanistic Insights into Cyclic Carbonate Lytic Enzyme Catalysis
The core of this technological advancement lies in the specific activity of the cyclic carbonate lytic enzyme produced by the Bacillus sp. ECU0015 strain. Unlike non-specific esterases, this enzyme exhibits profound substrate specificity and enantioselectivity towards aromatic cyclic carbonates. The mechanism involves the nucleophilic attack on the carbonyl carbon of the cyclic carbonate ring, leading to ring opening and the subsequent release of carbon dioxide and the chiral diol. The enzyme's active site is geometrically constrained to favor the binding and hydrolysis of one specific enantiomer of the substrate, leaving the other enantiomer untouched or converting it at a negligible rate, thereby driving the enantiomeric excess of the product to near-perfect levels. This high degree of discrimination is critical for pharmaceutical applications where the wrong enantiomer can be inactive or even toxic, ensuring that the resulting (S)-phenylethylene glycol meets the stringent purity requirements of modern drug development.
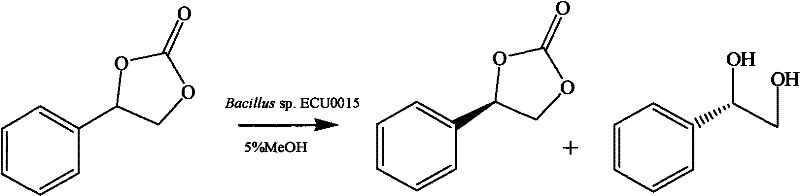
Beyond the primary reaction mechanism, the impurity profile of this biocatalytic process is remarkably clean, primarily due to the specificity of the biological catalyst which minimizes side reactions such as over-hydrolysis or polymerization. In chemical synthesis, side products often arise from competing reaction pathways or catalyst degradation, creating a complex mixture that is difficult to separate. However, in this enzymatic system, the byproduct is simply carbon dioxide, which evolves as a gas and leaves the reaction mixture, simplifying the workup procedure significantly. The remaining impurities are typically unreacted starting material, which can be easily separated from the polar diol product through standard extraction or crystallization techniques. This inherent selectivity not only improves the yield of the desired isomer but also drastically reduces the solvent usage and waste generation associated with extensive chromatographic purifications, aligning perfectly with the principles of green chemistry and sustainable manufacturing.
How to Synthesize (S)-Phenylethylene Glycol Efficiently
The practical implementation of this technology involves a straightforward fermentation and bioconversion protocol that is highly amenable to scale-up. The process begins with the activation of the Bacillus sp. ECU0015 strain on rich media followed by amplification in a fermentation broth optimized for enzyme production. Once the biomass is harvested, the resting cells are suspended in a buffered solution and exposed to the substrate, where the biotransformation occurs over a period of 12 to 48 hours depending on the substrate loading. The detailed standardized synthesis steps, including specific media compositions, incubation times, and extraction protocols required to replicate the high yields and ee values reported in the patent, are outlined in the technical guide below for process engineers and laboratory technicians.
- Cultivate Bacillus sp. ECU0015 in a fermentation medium containing glycerine, peptone, and meat extract at 30°C for 36 hours to generate active biomass.
- Harvest the cells via centrifugation and resuspend the resting cells in a phosphate buffer solution adjusted to pH 7.3.
- Add phenylethylene glycol cyclic carbonate substrate to the suspension and maintain oscillatory reaction at 30°C for 24 hours to achieve high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic route offers substantial strategic advantages beyond mere technical feasibility. The elimination of precious metal catalysts removes a major cost driver and supply bottleneck, as the price and availability of metals like palladium or rhodium can be highly volatile. Furthermore, the mild reaction conditions allow for the use of standard stainless steel reactors rather than specialized glass-lined or Hastelloy vessels required for corrosive chemical processes, representing a significant capital expenditure saving for manufacturing facilities. The simplicity of the downstream processing, driven by the gaseous nature of the CO2 byproduct and the high selectivity of the enzyme, translates to reduced solvent consumption and shorter batch cycle times, enhancing overall plant throughput and operational efficiency without compromising on quality.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived principally from the simplification of the synthetic route and the removal of expensive reagents. By utilizing whole-cell biocatalysts grown on inexpensive carbon sources like glycerine and peptone, the cost of goods sold (COGS) is significantly lowered compared to routes requiring stoichiometric chiral auxiliaries or rare earth catalysts. Additionally, the high enantiomeric excess achieved directly from the reaction minimizes the need for costly chiral resolution steps or recycling of unwanted isomers, effectively doubling the utility of the raw materials. The reduction in waste treatment costs, owing to the biodegradable nature of the biological catalyst and the absence of heavy metal sludge, further contributes to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Relying on a fermentable biological strain ensures a consistent and renewable source of catalytic activity, mitigating risks associated with the geopolitical instability often affecting the supply of specialty chemical reagents. The robustness of the Bacillus strain, which can survive in a wide pH range and moderate temperatures, ensures stable production performance even with minor fluctuations in process parameters. This reliability allows for better forecasting and inventory management, as the lead time for producing the biocatalyst is short and the strain can be stored for extended periods without significant loss of activity, guaranteeing continuity of supply for long-term commercial contracts.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory shake flasks to industrial fermenters, as demonstrated by the gram-level preparations described in the patent embodiments. The environmental footprint is drastically reduced, as the process avoids the generation of hazardous organic acid waste typical of chemical hydrolysis, replacing it with harmless carbon dioxide. This alignment with strict environmental regulations facilitates easier permitting and reduces the liability associated with hazardous waste disposal, making it an ideal candidate for manufacturers aiming to improve their sustainability metrics and comply with increasingly rigorous global environmental standards.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed experimental data provided in the patent documentation. These queries address common concerns regarding substrate scope, optical purity limits, and the practicalities of implementing biocatalysis in an existing chemical manufacturing infrastructure. Understanding these nuances is essential for assessing the feasibility of integrating this novel strain into your current production pipeline for chiral intermediates.
Q: What is the optical purity achievable with this biocatalytic process?
A: The process utilizing Bacillus sp. ECU0015 demonstrates exceptional stereoselectivity, consistently achieving optical purity (ee values) close to or higher than 98%, with specific embodiments reporting up to 99.8% ee for (S)-phenylethylene glycol.
Q: Does this method avoid the use of heavy metal catalysts?
A: Yes, unlike traditional chemical synthesis which often relies on organometallic catalysts, this biological method uses whole-cell biocatalysts, eliminating the need for expensive and toxic heavy metal removal steps.
Q: Can this process be scaled for industrial manufacturing?
A: The patent data indicates strong industrial applicability, with fermentation protocols designed for amplification cultivation and the use of robust resting cells that facilitate easy product purification and scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylethylene Glycol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in the synthesis of complex chiral molecules like phenylethylene glycol and mandelic acid derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and stereoselectivity demonstrated in laboratory settings are maintained at an industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to meet stringent purity specifications required by top-tier pharmaceutical clients while adhering to the highest standards of safety and environmental stewardship.
We invite you to collaborate with our technical team to explore how this innovative biocatalytic route can optimize your supply chain and reduce costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in chiral intermediate manufacturing can support your drug development goals and accelerate your time to market.
