Advanced Manufacturing Strategy for High-Purity Bortezomib Intermediates and API
Introduction to Next-Generation Bortezomib Manufacturing
The pharmaceutical landscape for oncology therapeutics demands not only high efficacy but also robust, scalable, and cost-efficient manufacturing processes. Patent CN103497233A introduces a transformative approach to the synthesis of Bortezomib (Velcade), a potent proteasome inhibitor used in the treatment of multiple myeloma. This technology addresses critical bottlenecks in traditional production by utilizing isobutylboronic acid as a readily available starting material, diverging from the industry standard of using expensive, pre-functionalized chiral boronates. The process encompasses an eight-step synthetic sequence that integrates stereoselective halogenation, amination, and peptide coupling, culminating in a highly efficient purification strategy that eliminates the need for silica gel chromatography. By shifting the paradigm from complex starting materials to simple precursors with in-situ chirality introduction, this method offers a compelling value proposition for a reliable API intermediate supplier seeking to optimize their portfolio.
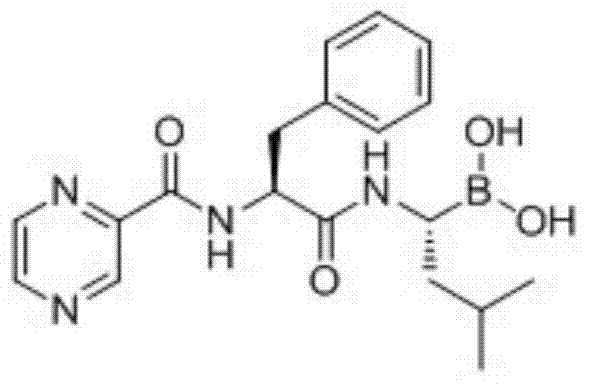
Furthermore, the strategic redesign of the purification workflow represents a significant leap forward in process chemistry. Traditional routes often suffer from low yields and high operational costs due to reliance on column chromatography, which is notoriously difficult to scale and generates substantial solvent waste. This patent details a meticulous optimization of workup procedures, employing specific aqueous washes to remove coupling byproducts like HOBt and urea derivatives without compromising the sensitive boronic ester moiety. The final product is obtained via recrystallization, achieving a purity exceeding 99.7%, which meets stringent international quality standards. This holistic improvement in both synthetic design and downstream processing positions this technology as a benchmark for cost reduction in pharmaceutical manufacturing, particularly for complex small molecules containing sensitive boron functionalities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Bortezomib has been hindered by the high cost and limited availability of key chiral building blocks. Most existing literature describes routes initiating with (1S,2S,3R,5S)-pinanediol 1-trifluoroacetic acid ammonium-3-methylbutane-1-borate, a specialized reagent that commands a premium price and introduces supply chain vulnerabilities. Additionally, the purification of intermediates in these conventional pathways heavily relies on silica gel column chromatography. While effective for laboratory-scale purification, column chromatography is fundamentally unsuited for multi-kilogram or ton-scale production due to its low throughput, high solvent consumption, and significant product loss caused by irreversible adsorption onto the silica matrix. Some alternative methods propose preparative liquid chromatography, but this similarly incurs prohibitive equipment costs and maintenance requirements, rendering the final API economically unviable for broader market access.
The Novel Approach
The methodology outlined in CN103497233A dismantles these barriers by reconstructing the synthetic tree from the ground up. By starting with isobutylboronic acid, a commodity chemical, the process decouples production from the volatility of specialized chiral boronate markets. The chirality is introduced efficiently via protection with (+)-pinanediol in the first step, setting the stage for high-fidelity stereocontrol. Crucially, the patent replaces chromatographic purification with a sophisticated liquid-liquid extraction protocol. For instance, during the condensation steps involving TBTU, the process utilizes a sequence of washes with dilute phosphoric acid, potassium carbonate, and saturated sodium chloride. This specific combination effectively removes acidic and basic impurities, including the problematic HOBt byproduct, while preserving the integrity of the boronate ester. This shift from solid-phase to liquid-phase purification dramatically enhances the commercial scale-up of complex proteasome inhibitors, ensuring that the process is not only chemically sound but also industrially pragmatic.
Mechanistic Insights into Stereoselective Alpha-Chlorination and Coupling
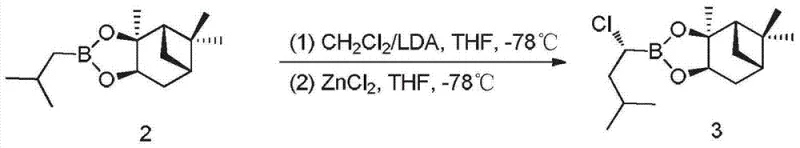
The heart of this synthetic route lies in the precise manipulation of the carbon-boron bond to establish the necessary stereochemistry. The second step involves the alpha-chlorination of the pinanediol isobutylboronate. This reaction employs Lithium Diisopropylamide (LDA) as a strong base to deprotonate the alpha-carbon adjacent to the boron atom, generating a nucleophilic organolithium species. The addition of Zinc Chloride (ZnCl2) at cryogenic temperatures (-78°C) facilitates transmetallation, forming an organozinc intermediate that is more stable and reactive towards electrophilic chlorination sources like methylene chloride. This low-temperature regime is critical for minimizing racemization and ensuring the formation of the desired (1S, 2R) configuration. The resulting chloro-boronate serves as a pivotal electrophile for the subsequent introduction of the nitrogen functionality, demonstrating a masterful application of main-group organometallic chemistry to solve stereochemical challenges.
Following the establishment of the carbon skeleton, the synthesis proceeds through peptide coupling reactions that require careful management of side reactions. The condensation of the amine intermediate with N-Boc-L-phenylalanine and subsequently with pyrazine-2-carboxylic acid utilizes TBTU (O-Benzotriazole-N,N,N',N'-tetramethyl-uronium-tetrafluoroborate) as the coupling agent. Mechanistically, TBTU activates the carboxylic acid to form an active ester, which is then attacked by the amine nucleophile. A key innovation in this patent is the post-reaction workup designed to handle the byproducts of this activation. Unlike standard protocols that might struggle with urea and HOBt residues, this process leverages the differential solubility of these impurities in acidic and basic aqueous phases. The use of 1% phosphoric acid specifically targets the removal of basic amines and coupling residues without hydrolyzing the sensitive boronic ester, a balance that is essential for maintaining high yield and purity throughout the multistep sequence.
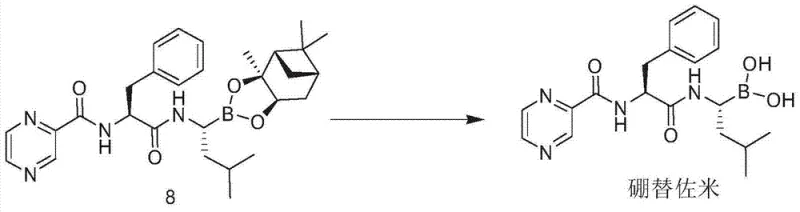
The final transformation involves the removal of the pinanediol protecting group to reveal the active boronic acid pharmacophore. This is achieved through an exchange reaction with isobutylboronic acid in the presence of dilute hydrochloric acid. This equilibrium-driven process effectively displaces the chiral diol, regenerating the free boronic acid of Bortezomib while recycling the pinanediol moiety. The choice of solvent system, typically a mixture of methanol and heptane or ethyl acetate, is optimized to precipitate the product or facilitate its extraction, further streamlining the isolation. This final step underscores the elegance of the design, where the protecting group serves its purpose of directing stereochemistry and solubility before being cleanly removed to yield the biologically active species, ready for the final recrystallization that guarantees the high-purity bortezomib required for clinical applications.
How to Synthesize Bortezomib Efficiently
Implementing this optimized synthetic route requires strict adherence to the specified reaction conditions, particularly regarding temperature control and reagent stoichiometry, to ensure the fidelity of the chiral centers. The process flows logically from the initial protection of the boron atom to the sequential construction of the peptide backbone, with each step validated for scalability. The detailed operational parameters, such as the specific molar ratios of LDA to substrate (1:1.5 to 1:2.5) and the precise temperature windows for exothermic additions, are critical for reproducibility. For process engineers and chemists looking to adopt this methodology, understanding the interplay between the boronate stability and the harsh conditions required for functionalization is key. The following guide outlines the standardized synthesis steps derived from the patent data, providing a roadmap for reducing lead time for high-purity oncology intermediates in a GMP environment.
- Protect isobutylboronic acid with (+)-pinanediol to form the chiral boronate ester intermediate.
- Perform stereoselective alpha-chlorination using LDA and ZnCl2 at low temperature (-78°C).
- Substitute chlorine with a silyl-protected amine group using LHMDS, followed by deprotection.
- Condense with N-Boc-L-phenylalanine using TBTU, then remove the Boc group with HCl.
- Couple with pyrazine-2-carboxylic acid, followed by final boronate deprotection and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented process offers distinct advantages that directly impact the bottom line and supply security. The primary driver of cost efficiency is the substitution of expensive, specialized starting materials with commodity chemicals like isobutylboronic acid. This shift not only lowers the direct material cost but also mitigates the risk of supply disruptions associated with niche reagents. Furthermore, the elimination of silica gel chromatography and preparative HPLC represents a massive reduction in operational expenditure. Chromatographic processes are resource-intensive, requiring vast quantities of solvents and stationary phases, along with significant labor for column packing and fraction collection. By replacing these unit operations with liquid-liquid extractions and crystallizations, the process becomes inherently more scalable and less dependent on specialized equipment, leading to substantial cost savings in manufacturing overhead.
- Cost Reduction in Manufacturing: The economic benefits of this route are multifaceted, stemming from both raw material selection and process intensification. By avoiding the purchase of pre-chiralized boronate esters, manufacturers can leverage the lower market price of isobutylboronic acid. Additionally, the removal of chromatographic purification steps drastically reduces solvent consumption and waste disposal costs, which are often hidden but significant expenses in API production. The ability to use standard reactor vessels for extractions and crystallizations, rather than dedicated chromatography columns, maximizes asset utilization and throughput. This streamlined approach ensures that the cost of goods sold (COGS) is minimized, allowing for more competitive pricing in the generic drug market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available reagents and robust reaction conditions. Isobutylboronic acid and common coupling agents like TBTU are produced by multiple global suppliers, reducing the risk of single-source dependency. The process conditions, such as the use of common solvents like dichloromethane, ethyl acetate, and heptane, further simplify logistics and inventory management. Moreover, the high yield and purity achieved through recrystallization reduce the need for reprocessing or reworking batches, which can cause significant delays in delivery schedules. This reliability is crucial for meeting the rigorous demand forecasts of pharmaceutical partners and ensuring uninterrupted availability of this life-saving medication.
- Scalability and Environmental Compliance: The transition from batch chromatography to continuous or semi-continuous extraction and crystallization aligns perfectly with modern green chemistry principles and regulatory expectations. Chromatography generates large volumes of mixed solvent waste that are difficult to recycle, whereas the aqueous washes and organic solvent layers in this new process are more amenable to separation and recovery. The reduced solvent load and the absence of silica gel waste simplify environmental compliance and lower the carbon footprint of the manufacturing process. This scalability ensures that production can be ramped up from pilot plant to commercial tonnage without encountering the engineering bottlenecks typical of chromatographic purifications, facilitating a smoother path to market approval and commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Bortezomib synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the route. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source. The answers reflect a commitment to transparency and technical excellence, ensuring that all stakeholders have a clear understanding of the process capabilities and quality outcomes.
Q: Why is isobutylboronic acid preferred over traditional starting materials for Bortezomib synthesis?
A: Traditional routes often rely on expensive, pre-formed chiral boronate esters like (1S,2S,3R,5S)-pinanediol 1-trifluoroacetic acid ammonium-3-methylbutane-1-borate. This novel process starts with inexpensive isobutylboronic acid and introduces the chiral pinanediol protecting group in situ, significantly reducing raw material costs and improving supply chain reliability.
Q: How does this process improve purification compared to standard methods?
A: Conventional methods frequently utilize silica gel column chromatography or preparative HPLC, which are difficult to scale, consume large amounts of solvents, and result in product loss due to adsorption. This patented method replaces column chromatography with optimized liquid-liquid extraction washes (using phosphoric acid and potassium carbonate) and a final recrystallization step, achieving purity greater than 99.7% suitable for industrial production.
Q: What represents the critical stereochemical control point in this synthesis?
A: The critical stereochemical control occurs during the alpha-chlorination step (Step 2) using LDA and ZnCl2 at -78°C. This ensures the correct (1S, 2R, 3R, 5S) configuration is established early in the sequence, which is maintained through the subsequent amination and peptide coupling steps to ensure the biological activity of the final proteasome inhibitor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bortezomib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the delivery of high-quality oncology therapeutics. Our team of expert chemists has extensively analyzed the innovations presented in CN103497233A and possesses the technical capability to implement this optimized pathway at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with state-of-the-art reactors capable of handling cryogenic reactions and sensitive organometallic chemistry, supported by rigorous QC labs that enforce stringent purity specifications to guarantee that every batch meets the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing process can be tailored to your specific supply chain needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this chromatography-free route. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your product portfolio's competitiveness and reliability. Together, we can drive the next generation of affordable and accessible cancer treatments to the patients who need them most.
