Advanced Transposition Strategy for Commercial Brassinolide Production and Side Chain Engineering
Introduction to Advanced Steroid Side Chain Engineering
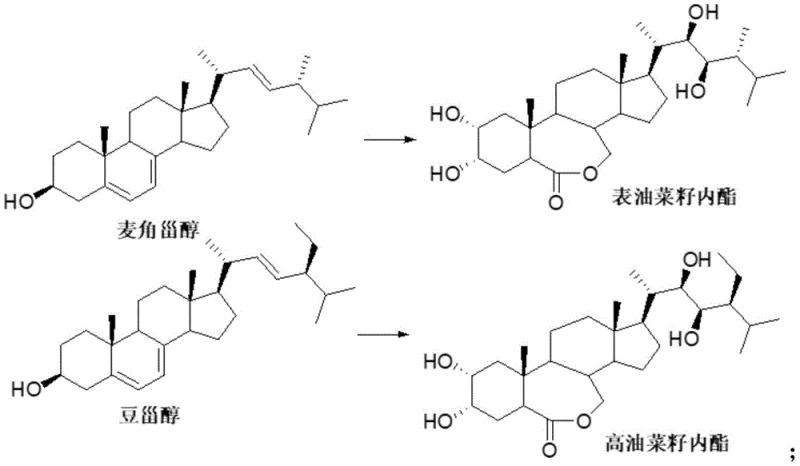
The agricultural sector continuously demands high-efficiency plant growth regulators to enhance crop yield and stress resistance, with brassinolide standing out as the most biologically active member of the brassinosteroid family. However, the scarcity of natural brassinolide in plant sources, where extraction yields are infinitesimally low, has necessitated the development of robust chemical synthesis routes. Patent CN114031666A introduces a groundbreaking methodology for the transposition of substituents at the 24-position of steroid compounds, specifically addressing the challenging conversion from an alpha configuration to a thermodynamically more stable beta configuration. This innovation represents a significant leap forward for any reliable agrochemical intermediate supplier seeking to optimize the production of high-value plant hormones. By leveraging a strategic oxidation-enolization sequence, this technology overcomes the historical bottlenecks associated with side-chain chirality, offering a pathway that is both chemically elegant and industrially viable for the manufacture of natural brassinolide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of natural brassinolide has been plagued by severe inefficiencies, primarily stemming from the reliance on extraction from rape pollen, a process that is economically prohibitive due to the requirement of processing hundreds of kilograms of biomass to obtain mere milligrams of product. Furthermore, existing total synthesis strategies, such as those reported in earlier literature involving multi-step constructions of the side chain, suffer from cumulative yield losses and extreme difficulties in controlling the stereochemistry at the C-24 position. These conventional routes often require complex chiral auxiliaries or resolution steps that drastically increase the cost of goods and extend the production timeline, making them unsuitable for cost reduction in plant growth regulator manufacturing on a commercial scale. The inherent rigidity of the steroid nucleus combined with the flexibility of the side chain creates a formidable challenge for traditional synthetic organic chemistry, often resulting in mixtures of diastereomers that are arduous to separate.
The Novel Approach
In stark contrast to these cumbersome traditional pathways, the novel approach detailed in the patent utilizes a semi-synthetic strategy that capitalizes on the availability of epibrassinolide, an analog that possesses the correct steroid core but lacks the specific 24-beta-methyl configuration. This method ingeniously bypasses the need to build the side chain from scratch by instead modifying an existing side chain through a precise transposition mechanism. By oxidizing the adjacent hydroxyl group to a ketone, the method creates an enolizable system that allows for the epimerization of the 24-methyl group under controlled basic conditions. This shift from a kinetic alpha-product to a thermodynamic beta-product simplifies the synthesis dramatically, reducing the number of steps and eliminating the need for expensive chiral catalysts, thereby providing a scalable solution for the commercial scale-up of complex steroid intermediates.
Mechanistic Insights into Base-Catalyzed Enolization and Transposition
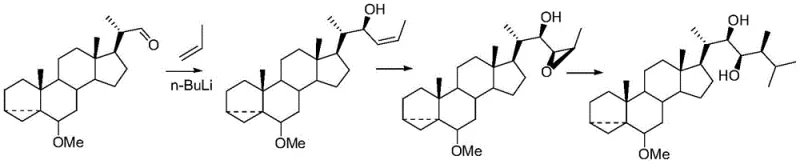
The core of this technological advancement lies in the subtle manipulation of keto-enol tautomerism to achieve stereochemical inversion at the C-24 position. The process initiates with the selective oxidation of the C-23 hydroxyl group to a ketone, typically using mild oxidants like Dess-Martin periodinane to preserve the integrity of the sensitive lactone ring and other hydroxyl functionalities. Once the ketone is formed, the introduction of a strong base, such as potassium tert-butoxide, facilitates the formation of an enolate intermediate. This enolate allows for the rotation around the C-23/C-24 bond, enabling the methyl substituent to adopt the energetically favored beta-orientation upon reprotonation. This mechanism effectively corrects the stereochemical error present in the starting material without requiring bond cleavage or reconstruction of the carbon skeleton, showcasing a sophisticated application of physical organic chemistry principles to solve a practical manufacturing problem.
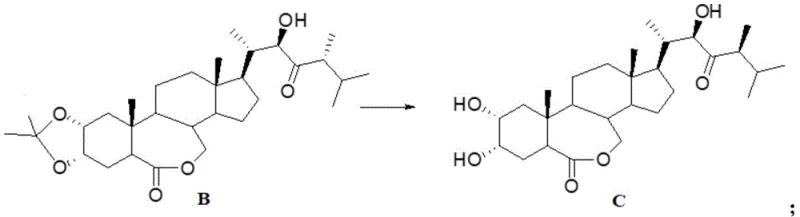
Furthermore, the control of impurities in this process is meticulously managed through the regulation of reaction temperature and the choice of solvent systems. The patent specifies maintaining reaction temperatures between 0°C and 10°C during the transposition step, which is critical for minimizing side reactions such as over-enolization or degradation of the lactone moiety. By quenching the reaction carefully with water and subsequently hydrolyzing the protecting groups under acidic conditions, the process ensures that the final product retains high purity with minimal formation of regioisomers or elimination byproducts. This rigorous control over the reaction environment ensures that the impurity profile remains within acceptable limits for agrochemical applications, reducing the burden on downstream purification processes like chromatography and crystallization.
How to Synthesize Natural Brassinolide Efficiently
The synthesis of natural brassinolide via this transposition route involves a logical sequence of protection, oxidation, transposition, and reduction steps that can be executed with standard laboratory equipment. The process begins with the protection of the vicinal diols on the A-ring to prevent unwanted oxidation, followed by the critical oxidation of the side chain hydroxyl. The subsequent transposition step is the heart of the innovation, where the stereochemistry is corrected, and finally, the ketone is reduced back to the hydroxyl group to yield the target molecule. For R&D teams looking to implement this, the detailed standardized synthesis steps see the guide below, which outlines the specific reagents, stoichiometry, and workup procedures required to achieve optimal yields and purity.
- Protect the 2,3-dihydroxyl groups of epibrassinolide using dibutyl acetonide to form the ketal intermediate.
- Oxidize the 23-hydroxyl group to a ketone using Dess-Martin periodinane under mild conditions.
- Perform base-catalyzed enolization with potassium tert-butoxide to transpose the 24-alpha-methyl to the beta-configuration, followed by hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented method offers substantial strategic benefits by shifting the supply chain dependency from scarce natural extracts or complex custom syntheses to readily available semi-synthetic starting materials. The ability to utilize epibrassinolide, which is more accessible and cost-effective to produce than natural brassinolide, fundamentally alters the cost structure of the final API. This transition allows for significant cost savings in manufacturing by reducing the number of synthetic steps and avoiding the use of precious metal catalysts that often require expensive removal processes. Additionally, the robustness of the reaction conditions implies a higher success rate in production batches, leading to more predictable inventory planning and reduced waste generation, which aligns with modern sustainability goals in the fine chemical industry.
- Cost Reduction in Manufacturing: The elimination of complex chiral synthesis steps and the use of common reagents like potassium tert-butoxide and Dess-Martin periodinane significantly lowers the raw material costs associated with production. By avoiding the need for cryogenic conditions or exotic catalysts, the operational expenditure related to energy consumption and specialized equipment is also drastically reduced. This streamlined approach minimizes the overall processing time and labor intensity, contributing to a leaner manufacturing model that enhances profit margins for high-purity brassinolides without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on a semi-synthetic route starting from commercially available epibrassinolide mitigates the risks associated with agricultural variability and extraction bottlenecks. This ensures a consistent and uninterrupted supply of the intermediate, which is crucial for meeting the seasonal demands of the agrochemical market. The simplified process flow reduces the likelihood of batch failures and delays, thereby reducing lead time for high-purity brassinolides and allowing suppliers to respond more agilely to market fluctuations and customer requirements.
- Scalability and Environmental Compliance: The reaction conditions described, operating at near-ambient temperatures and using standard organic solvents, are inherently easier to scale from kilogram to tonnage levels compared to sensitive low-temperature processes. The method avoids the generation of heavy metal waste streams often associated with alternative oxidation or reduction methods, simplifying wastewater treatment and environmental compliance. This green chemistry profile not only reduces disposal costs but also aligns with increasingly stringent global regulations regarding chemical manufacturing, making it a future-proof choice for long-term production strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this transposition technology in an industrial setting. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of adopting this route for your specific production needs.
Q: What is the primary advantage of this transposition method over traditional total synthesis?
A: The primary advantage is the utilization of commercially available epibrassinolide as a starting material, bypassing the complex construction of the steroid core and focusing solely on the difficult 24-position stereochemistry, thereby significantly shortening the synthetic route.
Q: How does the process ensure high stereochemical purity at the 24-position?
A: The process relies on thermodynamic control during the enolization step. By converting the 23-hydroxyl to a ketone and treating it with a base like potassium tert-butoxide, the system equilibrates to the more stable 24-beta-methyl configuration, effectively correcting the alpha-configuration of the starting material.
Q: Is this method suitable for large-scale industrial manufacturing?
A: Yes, the method utilizes standard reagents such as Dess-Martin periodinane and potassium tert-butoxide under moderate temperature conditions (0-10°C), avoiding cryogenic requirements or exotic catalysts, which facilitates straightforward scale-up and process safety management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brassinolide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this transposition technology in democratizing access to high-quality plant growth regulators. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a manufacturing environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of brassinolide delivered meets the highest standards of efficacy and safety required by the global agrochemical industry.
We invite you to collaborate with us to leverage this advanced synthesis route for your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can support your supply chain goals with reliability and precision.
