Advanced Iodine-Catalyzed Synthesis of Triflusal for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high efficiency with stringent safety and purity standards. Patent CN101503355B introduces a transformative approach to the synthesis and refining of Triflusal, also known chemically as 2-acetoxy-4-(trifluoromethyl)benzoic acid or UR-1501. This specific innovation pivots away from the historically dominant use of corrosive sulfuric acid catalysts, opting instead for a molecular iodine-mediated acetylation process. For R&D Directors and Technical Procurement Managers evaluating the landscape of antiplatelet agent intermediates, this shift represents a critical opportunity to enhance product quality while mitigating operational risks. The patent details a method where iodine serves as a highly efficient catalyst, facilitating the reaction between 4-(trifluoromethyl)salicylic acid and acetylating agents such as acetic anhydride or acetyl chloride. By fundamentally altering the catalytic environment, this technology addresses long-standing challenges related to impurity profiles and equipment longevity, positioning it as a superior choice for modern reliable pharmaceutical intermediate supplier networks aiming to optimize their supply chains for cardiovascular therapeutics.
The structural integrity and pharmacological efficacy of Triflusal depend heavily on the precision of its acetylation. As an irreversible inhibitor of platelet cyclooxygenase, the presence of specific impurities can alter its metabolic conversion to HTB (3-hydroxy-4-trifluoro-benzoic acid), potentially impacting clinical outcomes. The methodology outlined in the referenced patent ensures that the molecular architecture remains intact during synthesis, preserving the critical trifluoromethyl group and the ester linkage necessary for biological activity. This level of chemical fidelity is paramount for downstream formulation, where consistency is key to regulatory approval. By adopting this iodine-catalyzed route, manufacturers can achieve a level of high-purity triflusal that exceeds traditional benchmarks, thereby reducing the burden on downstream purification units and accelerating the overall time-to-market for finished dosage forms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of UR-1501 has relied heavily on sulfuric acid as the primary catalyst for the acetylation step. While effective in driving the reaction to completion, this conventional approach introduces severe engineering and chemical drawbacks that complicate cost reduction in API manufacturing. The use of concentrated sulfuric acid creates a highly corrosive environment that necessitates the use of specialized, expensive reactor linings and frequent equipment maintenance, driving up capital expenditure and operational downtime. Furthermore, the quenching phase of the sulfuric acid process is notoriously hazardous; adding water to terminate the reaction generates violent exotherms that are difficult to control, particularly when scaling from laboratory benchtops to multi-ton production vessels. This thermal instability poses significant safety risks to plant personnel and increases the likelihood of batch failures due to runaway reactions. Additionally, the harsh acidic conditions promote the degradation of the sensitive salicylic acid backbone, leading to the formation of complex by-product mixtures where single maximum contaminants can exceed 4%, requiring extensive and yield-loss-inducing recrystallization cycles to meet pharmacopeial standards.
The Novel Approach
In stark contrast, the novel iodine-catalyzed methodology described in the patent data offers a streamlined and safer alternative that directly addresses the deficiencies of the sulfuric acid route. By utilizing molecular iodine in catalytic amounts, the reaction proceeds under significantly milder conditions, eliminating the aggressive corrosivity that damages infrastructure and shortens asset life. The thermal profile of the iodine-mediated reaction is much more manageable, removing the risk of violent temperature spikes during the workup phase and allowing for precise control over the crystallization process. This stability translates directly into improved process safety and operational reliability, key factors for any commercial scale-up of complex pharmaceutical intermediates. Moreover, the selectivity of the iodine catalyst minimizes side reactions, resulting in a crude product with substantially lower impurity levels. This inherent purity reduces the number of purification steps required, thereby conserving solvents and energy while maximizing the overall recovery of the valuable active intermediate. The transition to this technology represents a strategic upgrade in process chemistry that aligns with modern green chemistry principles and economic efficiency goals.
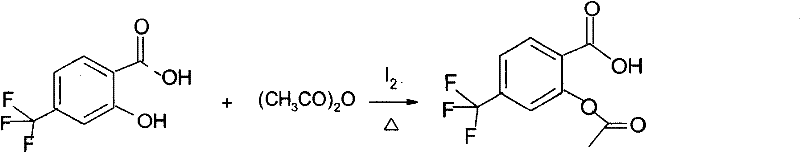
Mechanistic Insights into Iodine-Catalyzed Acetylation
The mechanistic advantage of using iodine lies in its ability to activate the acetylating agent without generating the strong protonic acidity associated with mineral acids. In this catalytic cycle, iodine likely functions as a Lewis acid, coordinating with the carbonyl oxygen of the acetic anhydride or acetyl chloride to increase the electrophilicity of the carbonyl carbon. This activation facilitates a nucleophilic attack by the phenolic hydroxyl group of the 4-(trifluoromethyl)salicylic acid substrate. Unlike the brute-force protonation mechanism of sulfuric acid, which can indiscriminately protonate various sites on the aromatic ring and lead to sulfonation or polymerization by-products, the iodine pathway is more selective. This selectivity is crucial for maintaining the integrity of the trifluoromethyl substituent, which is electron-withdrawing and deactivates the ring towards electrophilic substitution, yet requires careful handling to prevent defluorination or ring degradation under harsh conditions. The mild nature of the iodine catalyst ensures that the reaction kinetics are optimized for acetylation while suppressing competing degradation pathways, resulting in a cleaner reaction matrix that simplifies downstream processing.
Impurity control is another critical dimension where the iodine-catalyzed mechanism excels, providing R&D teams with a robust platform for quality assurance. In traditional acid-catalyzed processes, the formation of di-acetylated species or hydrolysis products is common due to the equilibrium dynamics driven by strong acids and water presence. The iodine system, operating effectively at temperatures between 50°C and 120°C, maintains a stable reaction environment where the generation of new impurities does not increase significantly even with prolonged reaction times. This stability is evidenced by patent data showing single maximum impurity levels dropping from over 4% in conventional methods to below 1% with iodine catalysis. For Quality Control laboratories, this means a more predictable impurity profile that is easier to characterize and validate. The ability to consistently produce material with purity levels approaching 98% at the crude stage significantly reduces the analytical burden and allows for tighter specification limits on the final API, ensuring that the reducing lead time for high-purity antiplatelet agents is achieved without compromising on safety or efficacy standards required by global regulatory bodies.
How to Synthesize Triflusal Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of operations that can be readily integrated into existing multipurpose chemical manufacturing facilities. The process begins with the charging of 4-(trifluoromethyl)salicylic acid and the acetylating agent into a dry reactor under an inert nitrogen atmosphere to prevent moisture interference. Molecular iodine is then introduced in a precise molar ratio, typically ranging from 0.002 to 0.1 equivalents, ensuring sufficient catalytic activity without excessive reagent costs. The mixture is heated to the optimal temperature range, monitored closely via Thin Layer Chromatography (TLC) to determine the endpoint, ensuring complete conversion of the starting material. Upon completion, the reaction is quenched carefully with ice water, inducing crystallization of the product which is then filtered, washed, and dried. The detailed standardized synthesis steps, including specific solvent volumes, stirring rates, and temperature ramps validated for reproducibility, are provided in the technical guide below for immediate reference by process engineers.
- Combine 4-(trifluoromethyl)salicylic acid with acetic anhydride or acetyl chloride in a reactor under nitrogen protection.
- Add molecular iodine as the catalyst with a molar ratio ranging from 0.002 to 0.1 relative to the substrate.
- Heat the mixture to 50°C-120°C, monitor via TLC, then quench with ice water and crystallize to obtain pure Triflusal.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of the iodine-catalyzed Triflusal synthesis offers compelling advantages that extend beyond simple yield metrics to impact the total cost of ownership and supply chain resilience. The elimination of corrosive sulfuric acid removes a major source of equipment degradation, leading to extended reactor lifecycles and reduced maintenance intervals, which translates into substantial cost savings over the long term. Furthermore, the milder reaction conditions reduce the energy load required for cooling and temperature control, contributing to a more sustainable and economically efficient manufacturing footprint. For Supply Chain Heads, the robustness of this process means fewer batch failures and more consistent delivery schedules, mitigating the risk of stockouts for critical cardiovascular medications. The simplified purification workflow also reduces the consumption of organic solvents and the volume of hazardous waste generated, aligning with increasingly strict environmental regulations and lowering disposal costs. These factors combine to create a supply proposition that is not only technically superior but also commercially more viable in a competitive global market.
- Cost Reduction in Manufacturing: The shift to an iodine catalyst fundamentally alters the cost structure by eliminating the need for corrosion-resistant alloys and frequent equipment replacements associated with sulfuric acid usage. Additionally, the higher crude purity reduces the number of recrystallization cycles needed, saving significant amounts of solvents, energy, and labor hours per kilogram of product. This efficiency gain allows for a more competitive pricing model without sacrificing margin, providing a clear economic incentive for switching to this advanced synthetic route.
- Enhanced Supply Chain Reliability: Process stability is the cornerstone of a reliable supply chain, and the iodine-catalyzed method offers superior control over reaction parameters compared to the volatile exotherms of traditional methods. This predictability ensures that production timelines are met consistently, reducing the variability in lead times that often plagues complex intermediate manufacturing. By minimizing the risk of batch rejection due to impurity spikes, manufacturers can maintain higher inventory turnover rates and respond more agilely to fluctuations in market demand for antiplatelet therapies.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this technology is inherently designed for scalability. The absence of violent exothermic quenching makes it safer to operate at larger volumes, while the reduction in hazardous waste streams simplifies compliance with environmental discharge permits. This ease of scale-up ensures that supply can be ramped up rapidly to meet commercial needs without requiring prohibitive investments in new safety infrastructure or waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and sourcing of Triflusal using this advanced iodine-catalyzed methodology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this intermediate into their broader portfolio of cardiovascular assets. The answers reflect a commitment to transparency and technical accuracy, ensuring that partners have a clear understanding of the capabilities and limitations of the synthesis route.
Q: Why is iodine preferred over sulfuric acid for Triflusal synthesis?
A: Iodine catalysis offers milder reaction conditions, significantly reduces equipment corrosion, and minimizes the formation of impurities compared to traditional sulfuric acid methods, resulting in higher purity crude products.
Q: What is the expected purity of Triflusal using this novel method?
A: Experimental data indicates that the iodine-catalyzed process can achieve crude purities of approximately 98%, with single maximum impurities controlled below 1%, surpassing conventional techniques.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process eliminates violent exothermic risks associated with acid quenching and uses non-corrosive catalysts, making it highly suitable for safe commercial scale-up and continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triflusal Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of the iodine-catalyzed Triflusal process are fully realized at an industrial level. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the high standards required for pharmaceutical applications. We understand the critical nature of cardiovascular intermediates and are committed to delivering consistent quality that supports your regulatory filings and commercial success.
We invite you to collaborate with us to optimize your supply chain for Triflusal and related antiplatelet agents. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics. By leveraging our manufacturing prowess and this innovative technology, we can together drive efficiency and reliability in the global supply of essential cardiovascular medicines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
