Advanced Purification Technology for High-Purity Triflusal Manufacturing and Commercial Scale-Up
Introduction to Advanced Triflusal Purification Technologies
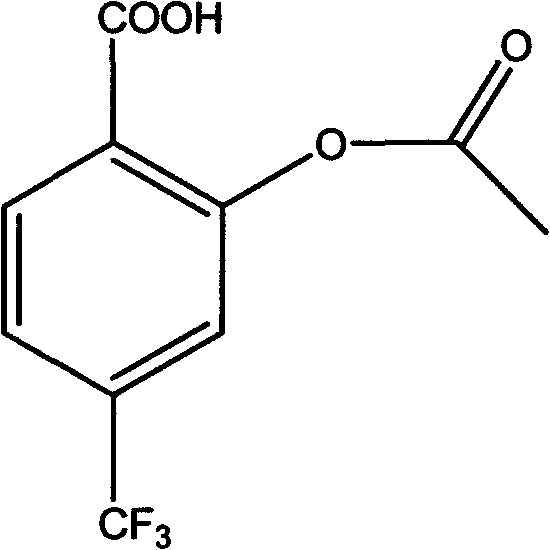
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients (APIs) and their critical intermediates to ensure patient safety and regulatory compliance. In the realm of cardiovascular therapeutics, Triflusal (2-acetoxy-4-(trifluoromethyl)benzoic acid) stands out as a potent antiplatelet aggregation agent, offering superior efficacy and a better safety profile compared to traditional aspirin. However, the manufacturing of high-purity Triflusal has historically been plagued by complex impurity profiles arising from self-polymerization during synthesis. Patent CN101177398B introduces a groundbreaking refinement methodology that addresses these longstanding challenges through a sophisticated two-step purification protocol. This technical insight report analyzes the proprietary process detailed in the patent, highlighting its potential to revolutionize the supply chain for reliable pharmaceutical intermediates supplier networks globally. By shifting from hazardous, low-yield solvent systems to a robust tetrahydrofuran (THF) and cyclohexane framework, manufacturers can achieve purity levels exceeding 99.5% while significantly enhancing operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Triflusal, as disclosed in earlier patents such as US4096252, relied heavily on acid-catalyzed esterification using sulfuric acid or acetyl chloride. A fundamental chemical flaw in these acidic conditions is the propensity for self-polymerization, where the carboxyl group of one molecule reacts with the hydroxyl group of another, creating dimeric or polymeric byproducts. These autohemagglutination products typically constitute 5-10% of the crude reaction mass and are notoriously difficult to separate due to their structural similarity to the target molecule. Conventional purification attempts involved repeated recrystallization using petroleum ether and diethyl ether systems. This approach is fraught with inefficiencies; diethyl ether's extremely low boiling point of 36°C limits the operating temperature window, causing rapid solvent evaporation and inconsistent solvent ratios during reflux. Furthermore, the high volatility and flammability of ether pose severe safety risks in large-scale industrial environments, often necessitating expensive explosion-proof infrastructure and limiting batch sizes to mitigate hazard exposure.
The Novel Approach
The innovative process outlined in patent CN101177398B fundamentally reengineers the purification workflow to bypass these thermodynamic and safety bottlenecks. Instead of struggling to separate polymeric impurities via crystallization alone, the new method employs a selective alkaline dissolution strategy as the primary purification step. By dissolving the crude product in a weak alkaline aqueous solution at controlled low temperatures (0-10°C), the target Triflusal converts to a soluble salt while the polymeric byproducts, which lack the necessary solubility characteristics in this medium, remain as solid insoluble impurities. These impurities are rapidly filtered off before the solution is acidified to recover the Triflusal crystals. This is followed by a secondary recrystallization using a THF and cyclohexane mixed solvent system. This solvent pair offers a much higher boiling point and thermal stability, allowing for consistent reflux conditions without the rapid composition shifts seen in ether-based systems. The result is a streamlined process that delivers Triflusal with a content greater than 99.5%, effectively eliminating the need for multiple, yield-depleting recrystallization cycles.
Mechanistic Insights into Alkaline Dissolution and Solvent Engineering
The core mechanism driving the success of this purification technology lies in the differential solubility of the target molecule versus its polymeric impurities in weakly alkaline media. Triflusal possesses both a carboxylic acid group and an ester linkage. Under mild alkaline conditions (using bases like sodium bicarbonate, sodium carbonate, or triethylamine), the carboxylic acid moiety is deprotonated to form a water-soluble carboxylate salt. Crucially, the polymeric byproducts formed during the initial acidic esterification—often esters formed between the acid of one molecule and the phenol of another—exhibit significantly lower solubility in these specific aqueous alkaline conditions. This allows for a physical separation via filtration that is far more efficient than chromatographic or crystalline separation. The process must be tightly controlled, with the entire dissolution and acidification sequence completed within 30 minutes at temperatures between 0-30°C to prevent hydrolysis of the sensitive acetoxy group, which could degrade the product back to the starting salicylic acid derivative.
Following the alkaline cleanup, the second stage leverages the synergistic properties of the THF and cyclohexane binary solvent system. Tetrahydrofuran (THF) has a boiling point of 65.40°C, which is approximately 30 degrees higher than diethyl ether. This elevated boiling point is critical for industrial scalability. During reflux, THF vapors condense efficiently, maintaining a stable solvent composition over extended periods. Experimental data indicates that even after 3 hours of reflux at 70°C with a 1:8 volume ratio, the solvent composition remains nearly unchanged. In contrast, ether systems suffer from preferential evaporation of the lower boiling component, altering the solubility parameters of the solution and leading to unpredictable crystallization outcomes. The THF/cyclohexane system provides a tunable solubility environment where the ratio can be adjusted (from 1:6 to 1:12) to optimize the crystalline rate for crude products of varying initial qualities, ensuring consistent recovery of high-purity material regardless of feedstock variations.
How to Synthesize High-Purity Triflusal Efficiently
Implementing this refined synthesis route requires precise control over temperature and stoichiometry to maximize the removal of polymeric impurities while preserving the integrity of the acetoxy group. The process begins with the careful selection of a weak base, such as sodium bicarbonate or triethylamine, to facilitate the selective dissolution of the crude API intermediate. Operators must ensure rapid filtration of the insoluble polymeric fraction immediately after dissolution to prevent re-equilibration. Following acidification and isolation of the intermediate solid, the critical recrystallization step utilizes a heated mixture of THF and cyclohexane. The detailed standardized synthesis steps, including specific mass ratios, temperature gradients, and drying protocols required to replicate the patent's >99.5% purity results, are outlined in the technical guide below.
- Dissolve crude Triflusal in a weak alkaline aqueous solution (e.g., sodium bicarbonate) at 0-10°C, filter rapidly to remove insoluble polymeric impurities, then acidify to precipitate purified crystals.
- Dry the intermediate solid under vacuum at low temperature to remove moisture content below 0.3%.
- Recrystallize the dried solid in a refluxing mixture of tetrahydrofuran (THF) and cyclohexane (volume ratio 1: 6 to 1:12), followed by cooling crystallization and vacuum drying to obtain >99.5% purity Triflusal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel purification method represents a strategic opportunity to optimize cost structures and mitigate operational risks. The elimination of diethyl ether from the process removes a significant safety liability, reducing the need for specialized explosion-proof equipment and lowering insurance and compliance costs associated with handling highly volatile solvents. Furthermore, the improved stability of the THF/cyclohexane solvent system allows for more predictable batch cycles, reducing the variability that often leads to production delays. By simplifying the purification train from multiple recrystallizations to a single robust cycle, manufacturers can significantly reduce solvent consumption and waste generation, aligning with modern green chemistry initiatives and reducing environmental disposal fees.
- Cost Reduction in Manufacturing: The implementation of this alkaline dissolution technique drastically reduces the loss of product associated with traditional multi-step recrystallization. Conventional methods often suffered from cumulative yield losses, with overall recovery rates hovering near 50% due to the necessity of repeating purification cycles to meet purity specs. By effectively removing the bulk of polymeric impurities in the first filtration step, the new process achieves substantially higher overall yields, directly translating to a lower cost of goods sold (COGS) per kilogram of finished API intermediate. Additionally, the ability to recover and reuse the THF/cyclohexane solvent system more efficiently than the ether/petroleum ether mix further contributes to long-term operational savings.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents and the regulatory hurdles of using hazardous solvents. Diethyl ether is subject to strict controls due to its use in illicit drug manufacture and its extreme flammability. By substituting this with THF and cyclohexane, which are commodity chemicals with robust global supply chains, manufacturers can secure a more reliable flow of raw materials. The process's tolerance for variations in crude feedstock quality—adjustable via solvent ratios—also means that production is less likely to be halted by minor fluctuations in upstream synthesis quality, ensuring a steady output of high-purity pharmaceutical intermediates for downstream drug formulation.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden thermal hazards, particularly with low-boiling solvents. The higher boiling point of the THF/cyclohexane system facilitates safer scale-up, as heat transfer and condensation are more manageable at 65-70°C compared to 36°C. This thermal margin allows for larger batch sizes without proportional increases in safety infrastructure costs. Moreover, the reduction in solvent waste and the elimination of ether emissions contribute to a smaller environmental footprint, helping companies meet increasingly stringent environmental, social, and governance (ESG) targets and regulatory discharge limits without requiring complex abatement technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Triflusal refinement technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method outperforms legacy techniques in terms of purity, safety, and yield. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for commercial manufacturing.
Q: How does the new refining method improve Triflusal purity compared to traditional methods?
A: Traditional acid-catalyzed esterification often produces 5-10% self-polymerized byproducts which are difficult to remove. The novel method utilizes a weak alkaline dissolution step where these polymeric impurities remain insoluble and are filtered out prior to recrystallization, consistently achieving purity levels greater than 99.5%.
Q: Why is the THF and cyclohexane solvent system preferred over petroleum ether and ether?
A: Ether has a very low boiling point (36°C), leading to significant evaporation losses and difficulty in maintaining solvent ratios during industrial reflux, alongside high flammability risks. The THF/cyclohexane system offers a higher boiling point (THF at 65.4°C), ensuring stable solvent composition during reflux, safer operation, and improved crystallization yields.
Q: What is the expected yield improvement with this purification technology?
A: While conventional recrystallization methods using ether systems often result in overall yields around 50% due to multiple crystallization steps and solvent losses, this optimized process demonstrates overall yields ranging from approximately 58% to 81% depending on specific base and solvent ratio conditions, representing a substantial efficiency gain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triflusal Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. While the patent CN101177398B outlines the theoretical framework for high-purity Triflusal production, our team possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this chemistry to life. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, consistently delivering the >99.5% purity benchmarks required by global regulatory bodies. We understand the critical nature of cardiovascular intermediates and maintain a supply chain resilient enough to handle the complexities of fluorinated chemistry and sensitive esterifications.
We invite pharmaceutical partners and contract manufacturers to engage with our technical procurement team to discuss how this advanced purification technology can be integrated into your supply chain. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technical excellence can drive value and reliability in your Triflusal sourcing strategy.
