Advanced Synthetic Route for Racemic Nicotine: Enhancing Purity and Scalability for Global Markets
Advanced Synthetic Route for Racemic Nicotine: Enhancing Purity and Scalability for Global Markets
The rapidly expanding electronic cigarette industry and the persistent demand for pharmaceutical-grade alkaloids have necessitated a paradigm shift in how high-purity active ingredients are manufactured. Patent CN115286615A introduces a groundbreaking preparation method for racemic nicotine that fundamentally addresses the limitations of traditional extraction and synthesis techniques. This innovative approach utilizes 3-pyridine acetonitrile and 1,3-dibromopropane as primary feedstocks, executing a sequence of nucleophilic, hydrolysis, ring-expansion, and reductive methylation reactions to achieve the final target molecule. By circumventing the need for carcinogenic tobacco extracts and avoiding the extreme operational hazards associated with legacy chemical syntheses, this technology offers a robust pathway for producing high-purity intermediates. For R&D directors and supply chain leaders, this patent represents a critical opportunity to secure a reliable source of complex heterocyclic compounds that aligns with modern green chemistry principles and stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of racemic nicotine has been plagued by significant technical and economic barriers that hinder efficient commercial scale-up. As illustrated in prior art literature, such as the route depicted in Reaction Scheme 1 from the Journal of Organic Chemistry (1990), traditional methods often rely on pyrrolidine derivatives subjected to harsh reaction conditions. These legacy processes frequently necessitate the use of highly reactive and dangerous reagents like tert-butyllithium or Grignard reagents, which require strict anhydrous environments and specialized handling protocols. Furthermore, other reported pathways starting from nicotinic acid or 3-bromopyridine often demand cryogenic temperatures as low as -78°C to control selectivity, imposing massive energy costs and requiring expensive refrigeration infrastructure. The reliance on such extreme conditions not only elevates the risk profile of the manufacturing process but also complicates waste management and limits the feasible batch sizes for industrial production.
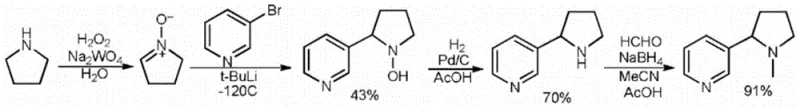
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN115286615A leverages a mild, step-wise construction of the pyrrolidine ring directly onto the pyridine scaffold. This novel strategy initiates with a straightforward nucleophilic substitution to form a cyclobutane intermediate, followed by a sophisticated ring-expansion reaction mediated by hypervalent iodine species. By operating primarily at ambient or moderately elevated temperatures (0°C to 55°C), this process eliminates the need for energy-intensive cryogenic cooling and removes the safety hazards associated with pyrophoric reagents. The result is a streamlined synthetic workflow that not only improves the overall yield to over 50% but also ensures a final product purity exceeding 99.5%, thereby providing a cost-effective and scalable solution for the mass production of high-purity racemic nicotine suitable for diverse industrial applications.
Mechanistic Insights into Hypervalent Iodine Mediated Ring Expansion
The core innovation of this synthetic route lies in the strategic transformation of the cyclobutane intermediate (Compound P2) into the five-membered pyrroline ring (Compound P3) using [bis(trifluoroacetoxy)iodo]benzene (PIFA). This hypervalent iodine reagent facilitates a Hofmann-like rearrangement under remarkably mild conditions, typically in a mixed solvent system of acetonitrile and water at room temperature. The mechanism involves the activation of the amide nitrogen, followed by a concerted migration of the carbon-carbon bond to expand the ring size, effectively converting the strained four-membered ring into the thermodynamically stable five-membered heterocycle found in nicotine. This specific catalytic step is crucial because it avoids the use of heavy metal catalysts or strong bases that could introduce difficult-to-remove impurities, thus simplifying the downstream purification process and ensuring a cleaner impurity profile for the final API intermediate.
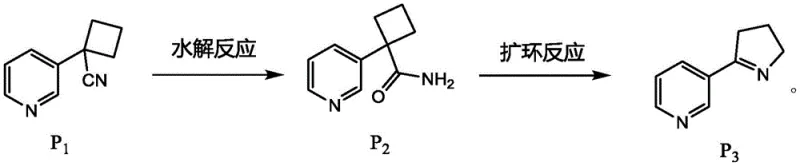
Furthermore, the control of impurities is intrinsically built into the reaction design through the selection of reagents and the sequential nature of the transformations. The initial nucleophilic cyclization uses inexpensive alkali bases like sodium hydride or potassium tert-butoxide, which are easily quenched, while the subsequent hydrolysis utilizes hydrogen peroxide and potassium carbonate, generating benign byproducts. The final reductive methylation step employs sodium borohydride and formic acid, a classic Eschweiler-Clarke modification that ensures complete conversion of the secondary amine to the tertiary amine without over-alkylation or side reactions. This rigorous control over the chemical environment at each stage minimizes the formation of regioisomers or polymeric byproducts, allowing the manufacturer to meet the stringent purity specifications required by global regulatory bodies for both pharmaceutical and consumer electronic products.
How to Synthesize Racemic Nicotine Efficiently
The synthesis of racemic nicotine via this patented route is designed for operational simplicity and high throughput, making it an ideal candidate for technology transfer from laboratory to pilot plant. The process begins with the alkylation of 3-pyridine acetonitrile, proceeds through a one-pot hydrolysis and ring expansion sequence, and concludes with a reductive methylation. Each step has been optimized to maximize yield while minimizing the need for intermediate isolation, particularly between the hydrolysis and ring expansion stages where the crude product can often be carried forward directly. For detailed procedural parameters, including exact stoichiometric ratios, solvent volumes, and temperature ramping profiles, please refer to the standardized synthesis guide provided below.
- Perform nucleophilic reaction between 3-pyridine acetonitrile and 1,3-dibromopropane under alkaline conditions to form compound P1.
- Conduct hydrolysis of compound P1 using potassium carbonate and hydrogen peroxide in DMSO to obtain amide intermediate P2.
- Execute ring expansion on P2 using [bis(trifluoroacetoxy)iodo]benzene (PIFA) to generate the pyrroline derivative P3.
- Complete the synthesis by reducing P3 and performing methylation using formaldehyde and formic acid to yield racemic nicotine (P4).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic methodology offers profound advantages in terms of cost structure and logistical reliability. By shifting away from extraction-dependent models and hazardous legacy syntheses, manufacturers can decouple their production capacity from agricultural variables and volatile reagent markets. The use of commodity chemicals such as 3-pyridine acetonitrile and 1,3-dibromopropane ensures a stable supply of raw materials, mitigating the risk of shortages that often plague specialty reagent-dependent processes. Additionally, the mild reaction conditions translate directly into lower capital expenditure (CAPEX) for reactor infrastructure, as standard stainless steel vessels can be used without the need for specialized cryogenic jackets or inert gas manifolds required for pyrophoric reagents.
- Cost Reduction in Manufacturing: The elimination of ultra-low temperature requirements and hazardous reagents like tert-butyllithium drastically reduces operational expenditures. Without the need for expensive cryogenic cooling systems or specialized safety containment for pyrophoric materials, the energy consumption per kilogram of product is significantly lowered. Furthermore, the ability to carry out the ring expansion without purifying the intermediate amide reduces solvent usage and labor time, leading to substantial overall cost savings in the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials ensures a consistent and predictable supply chain, free from the seasonal fluctuations associated with tobacco extraction. The robustness of the chemical process means that production schedules are less likely to be disrupted by reagent availability issues or strict transportation regulations governing hazardous goods. This stability allows for better long-term planning and inventory management, ensuring that downstream customers receive their orders on time without unexpected delays caused by raw material scarcity.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions facilitates easier scale-up from kilogram to multi-ton quantities without encountering the heat transfer or mixing limitations often seen in exothermic cryogenic reactions. Moreover, the avoidance of heavy metal catalysts and the generation of benign byproducts simplify waste treatment protocols, ensuring compliance with increasingly strict environmental regulations. This green chemistry approach not only reduces the environmental footprint but also lowers the costs associated with waste disposal and regulatory reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, safety profiles, and scalability potential for prospective partners evaluating this technology for their supply chains.
Q: How does this new synthetic route improve upon traditional nicotine synthesis methods?
A: Unlike conventional methods that require harsh cryogenic conditions (-78°C) and hazardous reagents like tert-butyllithium or Grignard reagents, this patented process operates under mild temperatures (0°C to 55°C) using stable, commercially available raw materials, significantly enhancing operational safety and reducing equipment costs.
Q: What is the expected purity and yield of the racemic nicotine produced?
A: The patent data demonstrates a total yield exceeding 50% across multiple examples, with final product purity consistently achieving greater than 99.5%, meeting stringent specifications for pharmaceutical and electronic cigarette applications without requiring complex chiral resolution.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the elimination of ultra-low temperature requirements and the use of non-purified intermediates for subsequent steps streamline the workflow, making it highly scalable for commercial production while minimizing waste generation and energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Racemic Nicotine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global fine chemicals market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to practice is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of racemic nicotine adheres to the highest quality standards required by the pharmaceutical and electronic cigarette industries.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific application needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability.
