Advanced Synthetic Route for Racemic Nicotine: Technical Breakthroughs and Commercial Scalability
The global demand for high-purity nicotine, driven primarily by the rapidly expanding electronic cigarette industry and pharmaceutical applications, has necessitated a shift from traditional tobacco extraction to advanced chemical synthesis. Patent CN115286615A, published in late 2022, introduces a groundbreaking preparation method for racemic nicotine that addresses critical pain points regarding impurity profiles and industrial scalability. Unlike plant-derived nicotine, which often contains carcinogenic tobacco-specific nitrosamines and suffers from agricultural supply chain fluctuations, this synthetic approach utilizes readily available petrochemical derivatives to achieve a purity exceeding 99.5%. The technical significance of this patent lies in its ability to bypass harsh reaction conditions and expensive reagents, offering a robust alternative for manufacturers seeking a reliable API intermediate supplier capable of delivering consistent, contaminant-free active ingredients.
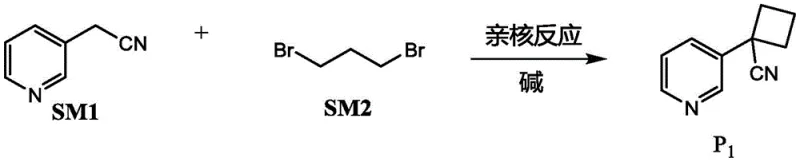
For R&D directors and process chemists, the elimination of biological impurities is paramount, but the economic feasibility of the synthesis route is equally critical for procurement strategies. This patent outlines a four-step sequence starting from 3-pyridine acetonitrile and 1,3-dibromopropane, proceeding through nucleophilic cyclization, hydrolysis, ring expansion, and finally reductive methylation. The strategic value of this methodology is its reliance on commodity chemicals rather than specialized, high-cost precursors, effectively de-risking the supply chain for large-scale production. By establishing a clear pathway that avoids the regulatory and safety hurdles associated with tobacco processing, this technology positions itself as a cornerstone for cost reduction in nicotine manufacturing, appealing directly to stakeholders focused on margin optimization and operational continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of racemic nicotine has been plagued by significant technical and economic barriers that hinder efficient commercial scale-up of complex alkaloids. Prior art methods, such as those reported in the Journal of Organic Chemistry (1990), often rely on pyrrolidine derivatives that require multi-step sequences with low overall yields and demanding reaction parameters. Other established routes, like those utilizing nicotinic acid, necessitate the use of Grignard reagents, which are notoriously moisture-sensitive and pose severe safety risks during industrial handling, requiring strictly anhydrous conditions and specialized equipment. Furthermore, approaches starting from 3-bromopyridine, while chemically viable, depend on ultra-low temperature conditions around -78°C and the use of super-strong bases like tert-butyl lithium, which are not only expensive but also present substantial storage and transportation hazards due to their pyrophoric nature.
These conventional pathways create a bottleneck for supply chain heads who must manage the logistics of hazardous materials and the high energy costs associated with cryogenic cooling. The reliance on such苛刻 conditions inevitably leads to higher production costs and increased potential for batch failures, making these methods less attractive for high-volume manufacturing. Additionally, the purification processes required to remove metal residues from Grignard reactions or byproducts from low-selectivity steps add further complexity and waste generation. Consequently, the industry has long sought a synthetic strategy that balances chemical elegance with practical manufacturability, avoiding the pitfalls of hazardous reagents and extreme thermal requirements that characterize legacy technologies.
The Novel Approach
The methodology disclosed in CN115286615A represents a paradigm shift by employing a mild, telescoped synthesis that begins with the nucleophilic coupling of 3-pyridine acetonitrile and 1,3-dibromopropane. This initial step constructs the cyclobutane ring system (Compound P1) under relatively benign conditions, utilizing common bases such as sodium hydride or potassium tert-butoxide in solvents like DMF or THF at temperatures ranging from 0°C to 55°C. This stands in stark contrast to the cryogenic requirements of previous methods, allowing the reaction to be performed in standard stainless steel reactors without the need for specialized low-temperature infrastructure. The accessibility of the starting materials, which are bulk commodity chemicals, ensures a stable supply chain and significantly lowers the raw material acquisition difficulty compared to brominated pyridines or chiral resolution agents.
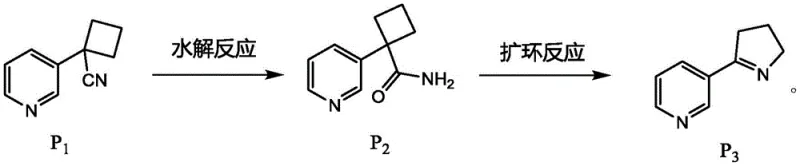
Following the formation of the cyclobutane intermediate, the process advances through a highly efficient hydrolysis and ring expansion sequence that minimizes unit operations. A distinct advantage of this novel approach is the ability to carry forward the hydrolysis product (Compound P2) directly into the ring expansion step without intermediate purification, a feature that drastically reduces solvent consumption and processing time. The ring expansion utilizes [bis(trifluoroacetoxy)iodo]benzene (PIFA) to convert the four-membered ring into the desired five-membered pyrroline structure (Compound P3) with high regioselectivity. This streamlined workflow not only enhances the overall throughput but also mitigates the accumulation of impurities that typically occur during multiple isolation steps, thereby supporting the production of high-purity intermediates essential for downstream pharmaceutical applications.
Mechanistic Insights into PIFA-Mediated Ring Expansion and Reductive Methylation
The core chemical innovation of this patent resides in the transformation of the cyclobutane derivative P2 into the pyrroline scaffold P3 via a hypervalent iodine-mediated ring expansion. Mechanistically, this reaction likely proceeds through the activation of the amide nitrogen in P2 by the electrophilic iodine species, generating a reactive nitrenium ion or an N-iodo intermediate that facilitates the migration of the carbon-carbon bond. This rearrangement effectively expands the four-membered ring to a five-membered ring, constructing the pyrrolidine backbone characteristic of nicotine. The use of PIFA in a mixed solvent system of acetonitrile and water allows for precise control over the reaction kinetics at room temperature, avoiding the thermal degradation often seen in high-temperature rearrangements. This mild condition is crucial for maintaining the integrity of the pyridine ring and preventing the formation of polymeric byproducts, ensuring a clean reaction profile that simplifies downstream workup.
Following the ring expansion, the final conversion to racemic nicotine (P4) involves a sequential reduction and methylation strategy that exemplifies process efficiency. The imine double bond in Compound P3 is first reduced using sodium borohydride or sodium triacetoxyborohydride to generate the secondary amine, demethylated nicotine. This reduction is conducted at 0°C to ensure chemoselectivity, preventing the over-reduction of the pyridine ring which could compromise the final product's identity. Subsequently, the secondary amine undergoes Eschweiler-Clarke methylation using formaldehyde and formic acid at elevated temperatures (80-90°C). This classic transformation installs the N-methyl group with high fidelity, completing the nicotine structure. The combination of these two steps in a single pot or sequential manner without isolating the unstable secondary amine intermediate further contributes to the high overall yield and purity observed in the experimental examples.
How to Synthesize Racemic Nicotine Efficiently
The synthesis of racemic nicotine described in this patent offers a practical blueprint for laboratory and pilot-scale production, emphasizing operational simplicity and safety. The protocol begins with the careful addition of base to a mixture of nitrile and dibromide, followed by a controlled temperature ramp to initiate cyclization. Subsequent steps leverage the crude nature of intermediates to accelerate the timeline, particularly the direct use of the hydrolysis product in the expansion reaction. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below which encapsulates the critical process controls identified in the patent examples.
- Perform nucleophilic reaction between 3-pyridine acetonitrile and 1,3-dibromopropane using a base like sodium hydride to form compound P1.
- Conduct hydrolysis on compound P1 using potassium carbonate and hydrogen peroxide in DMSO to obtain compound P2 without purification.
- Execute ring expansion on compound P2 using [bis(trifluoroacetoxy)iodo]benzene to generate the pyrroline intermediate P3.
- Complete the synthesis by reducing P3 with sodium borohydride followed by methylation with formaldehyde and formic acid to yield racemic nicotine (P4).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement managers and supply chain directors tasked with optimizing cost structures and ensuring material availability. The primary driver of value is the substitution of expensive, specialty starting materials with low-cost, widely available commodity chemicals. By utilizing 3-pyridine acetonitrile and 1,3-dibromopropane, manufacturers can decouple their production costs from the volatile pricing of halogenated pyridines or chiral resolving agents. Furthermore, the elimination of cryogenic requirements removes a significant energy burden from the manufacturing process, as there is no longer a need for extensive refrigeration capacity or the handling of dry ice and liquid nitrogen, leading to substantial operational expenditure savings.
- Cost Reduction in Manufacturing: The process design inherently lowers production costs by minimizing the number of isolation and purification steps. The ability to telescope the hydrolysis and ring expansion reactions without purifying the intermediate Compound P2 significantly reduces solvent usage, waste disposal costs, and labor hours associated with filtration and drying. Additionally, the avoidance of precious metal catalysts or hazardous organometallic reagents like Grignard reagents eliminates the need for expensive metal scavenging processes and specialized waste treatment protocols, resulting in a leaner and more cost-effective manufacturing footprint.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key raw materials are bulk chemicals produced by numerous global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate ambient temperatures and standard solvents, means that production can be easily transferred between different manufacturing sites without requiring bespoke equipment modifications. This flexibility ensures continuous supply even in the face of regional disruptions, providing a secure foundation for long-term procurement contracts and inventory planning.
- Scalability and Environmental Compliance: The mild reaction profile facilitates straightforward scale-up from kilogram to multi-ton quantities using standard chemical processing equipment. The absence of highly toxic reagents and the generation of fewer hazardous byproducts align with modern green chemistry principles, simplifying regulatory compliance and environmental permitting. This eco-friendly profile not only reduces the liability associated with hazardous material handling but also enhances the corporate sustainability metrics of the final product, a growing requirement for downstream customers in the consumer electronics and pharmaceutical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on purity standards, process safety, and scalability potential for interested partners.
Q: What are the primary advantages of this synthetic route over traditional tobacco extraction?
A: Unlike tobacco extraction which carries carcinogenic impurities and supply volatility, this chemical synthesis offers high purity (>99.5%) and eliminates biological contaminants, ensuring consistent quality for pharmaceutical and electronic cigarette applications.
Q: Does this process require expensive cryogenic conditions?
A: No, the process operates under mild conditions ranging from 0°C to 85°C, avoiding the ultra-low temperatures (-78°C) and hazardous reagents like tert-butyl lithium required in prior art methods, significantly lowering equipment and energy costs.
Q: Is intermediate purification required between every step?
A: A key efficiency of this patent is that compound P2 can be directly subjected to the ring expansion reaction without isolation or purification, streamlining the workflow and reducing solvent waste and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Racemic Nicotine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this synthetic pathway for the global nicotine market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the >99.5% purity benchmark essential for high-end applications.
We invite pharmaceutical and electronic cigarette manufacturers to collaborate with us to leverage this advanced technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in process chemistry can drive value and reliability for your business.
