Revolutionizing Chiral Cyclopropane Synthesis: A Scalable Copper-Catalyzed Approach for Global Supply Chains
Introduction to Advanced Asymmetric Catalysis Technology
The landscape of chiral synthesis is undergoing a significant transformation driven by the urgent need for more efficient and stable catalytic systems in the production of high-value intermediates. Patent CN1384105A introduces a groundbreaking asymmetric copper complex that addresses critical limitations in the synthesis of optically active cyclopropane carboxylates, which serve as essential building blocks for next-generation pyrethroid insecticides and beta-lactam antibiotics. This technology leverages a unique combination of an optically active bisoxazoline compound, a readily available copper salt, and a strong acid or Lewis acid activator to create a robust catalytic system. By shifting away from fragile, multi-step ligand preparations, this innovation offers a pathway to substantially lower production costs while maintaining the stringent stereochemical control required by regulatory bodies. For global procurement teams, this represents a pivotal opportunity to secure a reliable agrochemical intermediate supplier capable of delivering consistent quality without the volatility associated with traditional noble metal or complex organocatalytic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct synthesis of optically active cyclopropane carboxylic acid derivatives has relied heavily on asymmetric copper complexes utilizing ligands such as bis[2-(4,5-diphenyl-1,3-oxazolinyl)]methane, as documented in earlier literature like Tetrahedron Lett., 32, 7373 (1991). While chemically effective on a laboratory scale, these conventional methods present severe bottlenecks for industrial application due to the intricate and costly synthesis of the ligands themselves. Furthermore, the resulting copper complexes often suffer from poor stability, making their handling and storage difficult in a manufacturing environment. This instability frequently leads to batch-to-batch variability, increased waste generation, and the need for specialized containment protocols, all of which drive up the total cost of ownership for pharmaceutical and agrochemical manufacturers seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the novel approach disclosed in CN1384105A simplifies the catalyst architecture by decoupling the ligand complexity from the metal center activation. By introducing a strong acid or Lewis acid component—such as trifluoromethanesulfonic acid or boron trifluoride—to a mixture of a simple bisoxazoline ligand and an inexpensive copper salt, the system achieves high catalytic activity and stereoselectivity without the need for pre-synthesized, unstable complex ligands. This modular approach allows for the use of commodity copper compounds like copper acetate or copper chloride, which are vastly more accessible and affordable than specialized organometallic precursors. The result is a catalytic system that is not only easier to prepare but also exhibits superior robustness under reaction conditions, facilitating smoother technology transfer from pilot plants to full-scale commercial production facilities.
Mechanistic Insights into Acid-Activated Copper Catalysis
The core innovation of this technology lies in the synergistic interaction between the copper center, the chiral bisoxazoline ligand, and the acid activator. The optically active bisoxazoline compound coordinates with the monovalent or divalent copper ion to establish the chiral environment necessary for enantioselective induction. However, the critical differentiator is the addition of the strong acid or Lewis acid, which modifies the electronic properties of the copper complex, likely generating a more electrophilic metal-carbenoid species upon reaction with the diazoacetate. This activation step enhances the reactivity of the catalyst towards prochiral olefins, allowing the reaction to proceed efficiently even at lower catalyst loadings ranging from 0.0001 to 0.01 mole per mole of diazoacetate. The versatility of this mechanism is evidenced by its compatibility with a wide array of substrates, ensuring high-purity cyclopropane carboxylates regardless of the specific olefinic structure employed.
The substrate scope of this reaction is exceptionally broad, accommodating various prochiral olefins defined by general formula (2), which includes monoolefins such as isobutene and conjugated dienes like 2,5-dimethyl-2,4-hexadiene. 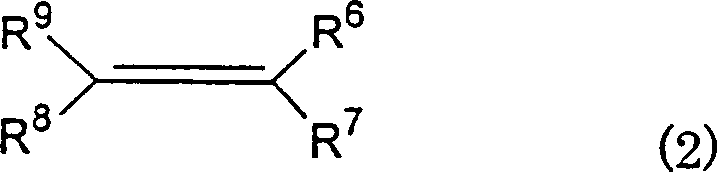 . The structural flexibility shown in the formula above allows for the introduction of diverse functional groups, including halogens, alkyl chains, and alkoxy groups, without compromising the optical purity of the final product. Experimental data from the patent indicates that the system can achieve optical purities as high as 96% e.e. for specific trans-isomers, demonstrating precise control over the stereochemical outcome. This level of control is paramount for R&D directors who must ensure that impurity profiles remain within strict limits to avoid downstream purification challenges and regulatory delays during the drug registration process.
. The structural flexibility shown in the formula above allows for the introduction of diverse functional groups, including halogens, alkyl chains, and alkoxy groups, without compromising the optical purity of the final product. Experimental data from the patent indicates that the system can achieve optical purities as high as 96% e.e. for specific trans-isomers, demonstrating precise control over the stereochemical outcome. This level of control is paramount for R&D directors who must ensure that impurity profiles remain within strict limits to avoid downstream purification challenges and regulatory delays during the drug registration process.
How to Synthesize Optically Active Cyclopropane Carboxylates Efficiently
Implementing this synthesis route requires careful attention to the order of addition and the choice of activator to maximize catalyst longevity and selectivity. The process generally involves mixing the copper salt and ligand in a solvent such as ethyl acetate or toluene, followed by the addition of the acid activator to generate the active species in situ. This operational simplicity reduces the need for isolated intermediate handling, thereby minimizing exposure to air and moisture which could degrade sensitive catalytic species. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles optimized for different substrate classes, are outlined in the guide below to ensure reproducible results across different manufacturing sites.
- Prepare the catalyst by mixing an optically active bisoxazoline ligand with a monovalent or divalent copper compound in a suitable organic solvent.
- Activate the copper complex by adding a strong acid or Lewis acid, such as trifluoromethanesulfonic acid or boron trifluoride, to the mixture.
- React the activated catalyst with a prochiral olefin and a diazoacetate at temperatures between -20°C and 130°C to yield the optically active cyclopropane product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this acid-activated copper catalyst system offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage is the drastic simplification of the raw material supply chain; by replacing exotic, custom-synthesized ligands with commodity copper salts and commercially available acids, manufacturers can significantly mitigate supply risk. This shift eliminates the dependency on single-source suppliers for complex ligands, thereby enhancing supply chain reliability and reducing the lead time for high-purity agrochemical intermediates. Furthermore, the robust nature of the catalyst reduces the frequency of reactor cleaning and maintenance caused by catalyst decomposition, leading to higher overall equipment effectiveness and reduced downtime during production campaigns.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the substitution of expensive catalytic precursors with low-cost commodity chemicals. Since the copper compounds used, such as copper acetate or copper chloride, are produced on a massive industrial scale, their price point is significantly lower than specialized organometallic complexes. Additionally, the elimination of complex ligand synthesis steps removes several unit operations from the upstream supply chain, resulting in substantial cost savings in terms of labor, energy, and solvent consumption. The ability to operate with lower catalyst loadings while maintaining high yields further contributes to a leaner cost structure, making the final API or agrochemical active ingredient more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the fragility of specialized reagents; however, the components of this novel catalyst system are stable, shelf-stable, and widely sourced. This inherent stability ensures that production schedules are not disrupted by reagent degradation or shipping delays associated with temperature-sensitive materials. Moreover, the tolerance of the system to various reaction conditions allows for greater flexibility in manufacturing planning, enabling facilities to adapt quickly to fluctuating demand without compromising product quality. This resilience is critical for maintaining long-term contracts with multinational partners who prioritize consistent delivery performance over marginal price differences.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns well with green chemistry principles by reducing the waste burden associated with ligand synthesis. The simplified workflow generates fewer by-products and allows for more efficient solvent recovery systems, which is increasingly important as environmental regulations tighten globally. The technology has been demonstrated to work effectively in continuous reaction processes, which are inherently safer and more scalable than batch operations for exothermic reactions involving diazo compounds. This scalability ensures that the commercial scale-up of complex chiral intermediates can be achieved smoothly from pilot quantities to multi-ton annual production capacities without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric cyclopropanation technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the advantages of this copper catalyst over conventional methods?
A: Unlike conventional methods requiring complex and unstable ligands like bis[2-(4,5-diphenyl-1,3-oxazolinyl)]methane, this patent utilizes readily available copper salts combined with simple bisoxazoline ligands and acid activators, significantly simplifying preparation and improving industrial stability.
Q: What types of substrates are compatible with this cyclopropanation process?
A: The process demonstrates broad substrate scope, effectively converting various prochiral olefins including monoolefins like isobutene and conjugated dienes into high-value optically active cyclopropane carboxylates with excellent enantioselectivity.
Q: Is this technology suitable for large-scale commercial production?
A: Yes, the method supports continuous reaction processes and utilizes inexpensive, commercially available copper compounds, making it highly suitable for commercial scale-up of complex chiral intermediates required in the agrochemical and pharmaceutical industries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Cyclopropane Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced catalytic technologies requires more than just laboratory success; it demands rigorous process engineering and quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify enantiomeric excess and impurity profiles for every batch of high-purity cyclopropane carboxylates we produce. Our commitment to technical excellence ensures that our clients receive intermediates that meet the highest global standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific product needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic system for your specific use case. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in asymmetric synthesis can drive value and efficiency in your supply chain.
