Revolutionizing Chiral Intermediate Production with Advanced Asymmetric Copper Complex Catalysts
The landscape of chiral synthesis for critical pharmaceutical and agrochemical intermediates is undergoing a significant transformation driven by the innovations disclosed in patent CN1259322C. This pivotal intellectual property introduces a novel class of asymmetric copper complexes that fundamentally alter the economics and feasibility of producing optically active cyclopropane carboxylates. These compounds serve as the essential acid components for synthetic pyrethroid insecticides and act as key building blocks in the synthesis of beta-lactam antibiotics. Traditionally, the industrial preparation of these high-value intermediates has been hindered by the reliance on cumbersome catalyst systems that suffer from poor stability and complex preparation protocols. The technology detailed in this patent overcomes these historical bottlenecks by employing a robust catalytic system composed of an optically active bisoxazoline compound, a simple copper salt, and a strong or Lewis acid activator. For global procurement teams and R&D directors seeking a reliable agrochemical intermediate supplier, understanding the mechanistic superiority of this acid-activated copper system is paramount for securing long-term supply chain resilience.
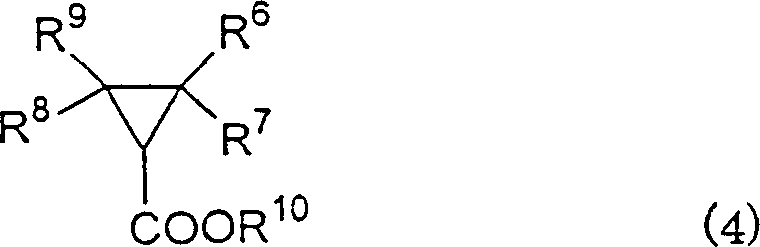
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this technological breakthrough, the industry standard for direct asymmetric cyclopropanation relied heavily on copper complexes utilizing bis[2-(4,5-diphenyl-1,3-oxazolinyl)]methane as the chiral ligand. While chemically effective in laboratory settings, these conventional methods present severe drawbacks when translated to industrial manufacturing environments. The synthesis of the requisite ligand involves multiple steps and expensive starting materials, driving up the initial capital expenditure for catalyst preparation. Furthermore, the resulting copper complexes often exhibit poor stability, making their storage and handling difficult and necessitating immediate use after preparation. This instability introduces significant operational risks, including batch-to-batch variability and potential safety hazards associated with decomposing catalytic species. For a procurement manager focused on cost reduction in pharmaceutical intermediates manufacturing, these inefficiencies translate directly into higher unit costs and unpredictable production schedules that can jeopardize downstream formulation timelines.
The Novel Approach
The patented approach described in CN1259322C offers a paradigm shift by decoupling catalyst performance from ligand complexity. By introducing a strong acid or Lewis acid component—such as trifluoromethanesulfonic acid, fluorosulfonic acid, or boron trifluide etherate—the system activates inexpensive and readily available copper compounds like copper acetate or copper chloride. This innovation eliminates the need for synthesizing elaborate, pre-formed stable complexes. The result is a catalytic system that is not only easier to prepare but also demonstrates remarkable versatility across a wide range of prochiral olefins and diazoacetates. This flexibility allows manufacturers to produce a diverse array of high-purity cyclopropane carboxylates without changing the core catalytic infrastructure. The ability to use commodity copper salts drastically reduces the raw material burden, while the enhanced stability of the acid-activated system ensures consistent performance over extended reaction times, facilitating the commercial scale-up of complex chiral intermediates.
Mechanistic Insights into Acid-Activated Asymmetric Cyclopropanation
The core mechanism driving the success of this technology lies in the synergistic interaction between the copper center, the chiral bisoxazoline ligand, and the acid activator. In traditional systems, the electronic environment of the copper atom is solely dictated by the ligand, which limits the tunability of the catalyst's Lewis acidity. In this novel system, the addition of a strong acid or Lewis acid modifies the coordination sphere of the copper, effectively increasing its electrophilicity. This heightened electrophilicity facilitates the decomposition of the diazoacetate reagent to form the reactive copper-carbenoid species more efficiently. The chiral pocket created by the bisoxazoline ligand then exerts precise stereocontrol over the approach of the prochiral olefin to the carbenoid intermediate. This dual-activation strategy ensures that the cyclopropanation proceeds with high diastereoselectivity and enantioselectivity, favoring the formation of the desired trans-isomers which are often the biologically active forms in agrochemical applications.
Furthermore, the impurity profile of the reaction is significantly improved due to the controlled generation of the carbenoid species. Uncontrolled decomposition of diazo compounds often leads to dimerization byproducts or non-selective insertion reactions, which complicate downstream purification. The acid-activated copper complex moderates this reactivity, ensuring that the carbene is transferred to the olefin substrate before side reactions can occur. This mechanistic precision is critical for reducing lead time for high-purity agrochemical precursors, as it minimizes the need for extensive chromatographic purification steps. The patent data highlights that by adjusting the nature of the acid—from Brønsted acids like triflic acid to Lewis acids like antimony pentafluoride—chemists can fine-tune the reaction kinetics to match specific substrate requirements, offering a level of process control that was previously unattainable with static ligand systems.

How to Synthesize Optically Active Cyclopropane Carboxylates Efficiently
The synthesis protocol outlined in the patent provides a robust framework for implementing this technology in a production setting. The process begins with the in situ generation of the catalyst, where the bisoxazoline ligand and copper salt are mixed in a solvent such as ethyl acetate or toluene, followed by the addition of the acid activator. This mixture is stirred to ensure complete complexation before the introduction of the substrates. The reaction is typically conducted under an inert atmosphere to prevent oxidation of the copper center, with temperatures maintained between -20°C and 130°C depending on the boiling point of the olefin solvent. The diazoacetate is added slowly to manage the exotherm and maintain high selectivity.
- Prepare the asymmetric copper complex catalyst by mixing an optically active bisoxazoline compound, a copper salt, and a strong or Lewis acid in a suitable organic solvent.
- Combine the catalyst solution with a prochiral olefin substrate under an inert atmosphere at controlled temperatures ranging from -20°C to 130°C.
- Slowly add the diazoacetate reagent to the reaction mixture to initiate cyclopropanation, followed by isolation and purification of the optically active ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this catalytic technology represents a strategic opportunity to optimize the cost structure of chiral intermediate production. The primary driver of value is the substitution of expensive, custom-synthesized ligands with commodity copper salts and commercially available acids. This shift fundamentally alters the bill of materials, leading to substantial cost savings that compound over large production volumes. Additionally, the simplified catalyst preparation reduces the labor and equipment time required in the upstream phase of manufacturing. Because the catalyst system is more stable and tolerant of varying conditions, the risk of batch failure is minimized, ensuring a more reliable supply of critical intermediates for downstream drug and pesticide synthesis. This reliability is essential for maintaining continuous operation in large-scale facilities where downtime is prohibitively expensive.
- Cost Reduction in Manufacturing: The elimination of complex ligand synthesis steps removes a significant cost center from the production workflow. By utilizing inexpensive copper sources like copper acetate or copper chloride, manufacturers can achieve a leaner cost base without compromising on the quality of the final product. The ability to use lower catalyst loadings, as demonstrated in the patent examples where effective turnover is achieved with minute quantities of copper, further drives down the cost per kilogram of the active pharmaceutical ingredient or agrochemical. This economic efficiency allows companies to remain competitive in price-sensitive markets while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Dependence on specialized, single-source ligands creates vulnerability in the supply chain. By switching to a system based on widely available chemical commodities, procurement teams can mitigate the risk of supply disruptions. The robustness of the acid-activated catalyst also means that transportation and storage requirements are less stringent compared to sensitive pre-formed complexes. This flexibility allows for larger inventory buffers and more resilient logistics planning, ensuring that production lines remain operational even during periods of global supply chain volatility.
- Scalability and Environmental Compliance: The process operates under relatively mild conditions and uses standard organic solvents that are easily recovered and recycled, aligning with modern green chemistry principles. The high selectivity of the reaction reduces the generation of hazardous waste streams associated with byproduct removal. From a scalability perspective, the liquid-phase nature of the catalyst allows for easy integration into continuous flow reactors or large batch vessels. This adaptability ensures that the technology can grow with demand, supporting the transition from pilot-scale validation to multi-ton commercial production without the need for fundamental process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric copper complex technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of this asymmetric copper complex over conventional catalysts?
A: Unlike conventional methods requiring complex and unstable ligands like bis[2-(4,5-diphenyl-1,3-oxazolinyl)]methane, this patented technology utilizes readily available copper compounds activated by strong or Lewis acids. This significantly simplifies catalyst preparation, enhances stability during storage and handling, and reduces overall production costs while maintaining high optical purity.
Q: Can this process be scaled for industrial production of pyrethroid intermediates?
A: Yes, the process is highly amenable to commercial scale-up. The reaction conditions are mild, utilizing common organic solvents such as toluene or ethyl acetate, and the catalyst loading can be optimized to very low levels (0.0001 to 0.01 mole per mole of diazoacetate), making it economically viable for large-scale manufacturing of agrochemical intermediates.
Q: What level of optical purity can be achieved with this catalytic system?
A: The patented method demonstrates exceptional stereoselectivity. Experimental data indicates that specific configurations of the bisoxazoline ligand combined with appropriate acid activators can achieve optical purities exceeding 96% e.e. for certain trans-isomers, ensuring high-quality standards required for pharmaceutical and agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Cyclopropane Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in laboratory examples can be faithfully reproduced on an industrial scale. We understand the critical importance of stringent purity specifications and rigorous QC labs in the production of chiral intermediates, particularly for regulated industries like pharmaceuticals and agrochemicals. Our commitment to quality assurance means that every batch of optically active cyclopropane carboxylate we produce meets the highest international standards for optical purity and chemical composition.
We invite you to collaborate with us to leverage this advanced catalytic technology for your specific product needs. Whether you are looking to optimize an existing route or develop a new process for a complex chiral building block, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technologically superior, positioning your organization for long-term success in the competitive global market.
