Advanced Cyclization Technology for Commercial Scale-up of Complex Pharmaceutical Intermediates
The global demand for high-purity fluoroquinolone antibiotics continues to drive innovation in process chemistry, particularly for critical molecules like Levofloxacin and Ofloxacin. Patent CN100412075C introduces a transformative approach to the synthesis of these anti-infective agents, addressing long-standing inefficiencies in industrial manufacturing. By utilizing tetrafluorobenzoic acid derivatives as the primary starting material, this methodology redefines the cyclization strategy, moving away from fragmented multi-step procedures towards a more integrated and thermally controlled process. For R&D Directors and Supply Chain Heads, the implications are profound: a reduction in unit operations directly correlates to lower operational expenditure and enhanced batch consistency. This report analyzes the technical merits of this patent, highlighting how its specific reaction conditions and solvent management strategies offer a viable pathway for reliable pharmaceutical intermediate supplier partnerships seeking to optimize their production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for fluoroquinolones, as illustrated in prior art, often suffer from significant logistical and chemical inefficiencies that hinder commercial scalability. A common historical approach involves distinct reaction vessels for different stages of the synthesis, necessitating frequent solvent switches between polar aprotic solvents and aqueous systems. This fragmentation creates substantial bottlenecks, including high solid transfer volumes, increased dust generation, and complex solvent recovery systems that struggle to achieve satisfactory recycling rates. Furthermore, conventional methods frequently rely on simultaneous ring closure at elevated temperatures ranging from 120°C to 150°C for extended periods of 4 to 5 hours. Such harsh thermal conditions inevitably promote side reactions, leading to impurities that compromise product quality and appearance. The reliance on large quantities of glacial acetic acid for hydrolysis further exacerbates environmental concerns and increases separation difficulties, making the overall process less attractive for modern green chemistry standards.
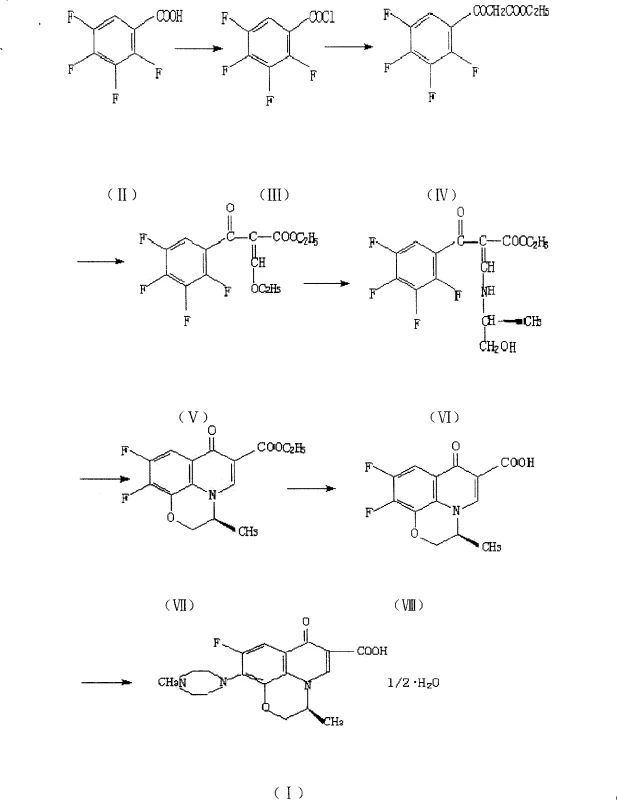
The Novel Approach
In stark contrast, the methodology disclosed in CN100412075C employs a strategic stepwise cyclization technique that fundamentally alters the reaction landscape. Instead of forcing simultaneous ring formation under continuous high heat, this novel approach separates the cyclization into two distinct thermal phases within a single solvent system. The first ring is formed at a moderate temperature range of 50°C to 90°C, followed by the introduction of N-methyl piperazine and a subsequent temperature increase to 120°C to 130°C for the second ring closure. This precise thermal modulation drastically shortens the high-temperature exposure time to merely 0.1 to 1.0 hour, significantly minimizing thermal degradation and side product formation. By maintaining a consistent solvent environment throughout the transformation from the acrylate intermediate to the final ester, the process eliminates the need for intermediate isolation and solvent swapping, thereby streamlining the workflow and enhancing overall yield consistency.
Mechanistic Insights into Stepwise Cyclization and Hydrolysis
The core innovation of this patent lies in the mechanistic control over the heterocyclic ring formation, which is critical for ensuring the structural integrity of the fluoroquinolone core. The reaction begins with the nucleophilic attack of L-aminopropanol or DL-aminopropanol on the tetrafluorobenzoyl acrylate derivative, facilitated by a base such as potassium carbonate or triethylamine. The initial cyclization at lower temperatures allows for the formation of the oxazine ring without triggering premature decomposition of the sensitive fluorine-substituted aromatic system. Following this, the addition of N-methyl piperazine targets the remaining reactive site, and the subsequent temperature ramp to 120°C to 130°C drives the closure of the pyridone ring. This sequential mechanism ensures that each bond formation occurs under its optimal thermodynamic conditions, preventing the cross-reactivity that often plagues one-pot high-temperature syntheses. The result is a cleaner reaction profile with a markedly improved impurity spectrum, which is a key metric for R&D Directors evaluating process robustness.
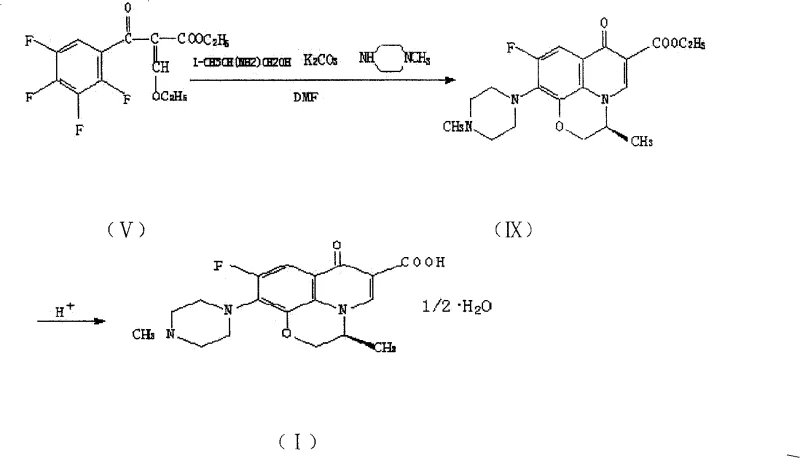
Impurity control is further enhanced by the modified hydrolysis protocol, which occurs after the full heterocyclic system has been established. In traditional methods, hydrolysis is often attempted on partially cyclized intermediates, leading to incomplete conversion and difficult separations. However, in this optimized route, the hydrolysis is performed on the fully formed ethyl ester intermediate, which exhibits superior solubility characteristics in the acidic hydrolysis medium. The use of concentrated hydrochloric acid under reflux conditions ensures rapid and complete cleavage of the ester group, converting it to the free carboxylic acid without the need for excessive acetic acid. Following hydrolysis, the pH is carefully adjusted to 7.0 using alkali, allowing for efficient extraction of the final product. This sequence not only improves the chemical purity but also simplifies the downstream processing, reducing the burden on purification teams and ensuring that the final API intermediate meets stringent quality specifications.
How to Synthesize Levofloxacin Efficiently
The implementation of this synthesis route requires careful attention to solvent selection and thermal profiling to maximize the benefits of the patented method. The process is designed to be operationally simple, allowing for the conversion of the crude acrylate intermediate directly into the final product with minimal intervention. By utilizing solvents such as DMF, DMSO, or isoamyl alcohol, the reaction maintains homogeneity throughout the critical cyclization phases, which is essential for heat transfer and reaction kinetics. The following guide outlines the standardized operational framework derived from the patent examples, serving as a baseline for process engineers looking to adapt this technology for large-scale production. Detailed standard operating procedures regarding specific reagent grades and equipment specifications should be consulted to ensure full compliance with safety and quality standards.
- Dissolve the crude 2-(2,3,4,5-tetrafluorobenzoyl)-3-ethoxy-ethyl acrylate in a polar aprotic solvent and cool to freezing temperatures before adding L-aminopropanol.
- Raise the temperature to 50-90°C to facilitate the first ring closure, then add N-methyl piperazine and increase heat to 120-130°C for the second cyclization.
- Hydrolyze the resulting ester intermediate using acid reflux, neutralize to pH 7.0, and extract the final Levofloxacin product using organic solvents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this optimized synthesis route offers tangible strategic advantages that extend beyond mere chemical yield. The consolidation of multiple reaction steps into a streamlined two-step process significantly reduces the operational complexity of the manufacturing line. By eliminating the need for intermediate isolation and solvent switching, the facility can achieve higher throughput with existing infrastructure, effectively increasing capacity without capital expenditure on new reactors. This operational efficiency translates directly into cost reduction in API manufacturing, as labor hours, energy consumption, and solvent procurement costs are all substantially minimized. Furthermore, the simplified workflow reduces the risk of human error during material transfer, enhancing the overall reliability of the supply chain and ensuring consistent delivery schedules for downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive unit operations and the optimization of solvent usage. Traditional methods often require vast quantities of glacial acetic acid and multiple solvent types, each incurring procurement, storage, and disposal costs. By contrast, this novel method utilizes a single solvent system that can be concentrated and reclaimed mechanically with high efficiency. The removal of transition metal catalysts or complex purification steps further reduces the cost of goods sold. Additionally, the shorter reaction times mean that reactor turnover is faster, allowing for more batches to be produced within the same timeframe, thereby spreading fixed costs over a larger volume of product and achieving significant cost savings.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are prone to bottlenecks or failure points. The robustness of this synthesis route, characterized by its tolerance to standard industrial solvents and straightforward temperature controls, mitigates these risks. The use of readily available raw materials such as tetrafluorobenzoic acid derivatives ensures that sourcing remains stable even during market fluctuations. Moreover, the reduced number of processing steps decreases the likelihood of batch failures due to handling errors, ensuring a steady flow of high-purity intermediates. This reliability is crucial for maintaining just-in-time inventory levels and meeting the rigorous delivery commitments expected by global pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden inefficiencies, particularly regarding waste management. This patented method addresses environmental compliance by significantly reducing the volume of waste solvents and acidic byproducts generated per kilogram of product. The ability to reclaim and reuse the primary reaction solvent minimizes the environmental footprint, aligning with increasingly strict global regulations on industrial emissions. The simplified hydrolysis step also reduces the load on wastewater treatment facilities. These factors make the process highly scalable, allowing manufacturers to expand production from 100 kgs to 100 MT annual commercial production volumes without encountering prohibitive environmental barriers or requiring extensive new waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers provided reflect the specific advantages of the stepwise cyclization and hydrolysis protocol over conventional methods.
Q: How does the new cyclization method improve upon conventional Levofloxacin synthesis?
A: The patented method eliminates the need for solvent switching between reaction steps, allowing the entire cyclization process to occur in a single reactor. This significantly reduces solid transfer operations and simplifies solvent recovery compared to traditional multi-vessel approaches.
Q: What are the critical temperature controls required for this synthesis?
A: The process requires precise stepwise temperature management: an initial low-temperature phase at 50-90°C for the first ring closure, followed by a high-temperature phase at 120-130°C for the second ring. This prevents side reactions common in continuous high-heat methods.
Q: Why is the hydrolysis step more efficient in this protocol?
A: By introducing the N-methyl piperazine group prior to hydrolysis, the resulting intermediate exhibits better solubility characteristics in the hydrolysis medium. This ensures complete conversion without the need for excessive glacial acetic acid or prolonged reaction times.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levofloxacin Supplier
The technical potential of the CN100412075C patent represents a significant opportunity for optimizing the production of fluoroquinolone intermediates, and NINGBO INNO PHARMCHEM is uniquely positioned to leverage this innovation. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are translated into industrial realities. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Levofloxacin or Ofloxacin intermediate meets the highest international standards. We understand the critical nature of API supply chains and are committed to delivering consistent quality that supports your regulatory filings and commercial launches.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your supply chain efficiency. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By engaging with us, you can access specific COA data and route feasibility assessments that will demonstrate the tangible value of this technology for your organization. Let us help you reduce lead time for high-purity pharmaceutical intermediates and secure a competitive advantage in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
