Revolutionizing L-Nicotine Production: A Deep Dive into Asymmetric Catalysis and Commercial Scalability
Revolutionizing L-Nicotine Production: A Deep Dive into Asymmetric Catalysis and Commercial Scalability
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce high-value chiral intermediates, and the recent disclosure in patent CN114644614B represents a significant leap forward in the synthesis of L-nicotine. This patent details a robust, three-step synthetic route that bypasses the limitations of traditional tobacco extraction and inefficient chemical resolution, achieving an optical purity exceeding 99.9% ee. For R&D directors and procurement specialists, this technology offers a compelling alternative to legacy methods, promising not only superior product quality but also enhanced process safety and scalability. The core innovation lies in the strategic use of N-vinylpyrrolidone as a building block and a highly selective asymmetric hydrogenation step, which collectively drive the total synthesis yield to an impressive 50-60%. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is critical for evaluating the long-term viability and cost-effectiveness of sourcing such critical alkaloids.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of L-nicotine has been plagued by significant technical and economic hurdles that impact supply chain stability and product consistency. Traditional extraction from tobacco leaves is inherently variable, dependent on agricultural cycles, climate conditions, and land resources, often resulting in a product with purity levels below 95% and contaminated with carcinogenic minor alkaloids. On the synthetic front, early chemical routes, such as the one depicted in 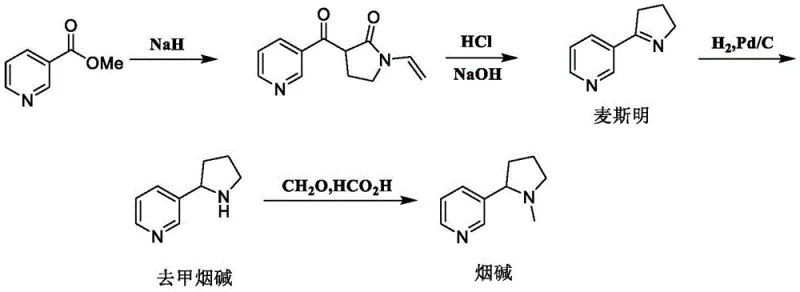 , relied on methyl nicotinate but suffered from low yields (around 40% for the myosmine intermediate) and the generation of substantial tarry byproducts that complicated purification. Furthermore, other established methods required hazardous reagents like sodium hydride or organolithium compounds operating at cryogenic temperatures of -78°C, posing severe safety risks and energy costs during scale-up. Perhaps most critically, resolution strategies for racemic nicotine inherently waste 50% of the material as the unwanted enantiomer, drastically inflating the cost of goods sold and creating significant waste disposal challenges.
, relied on methyl nicotinate but suffered from low yields (around 40% for the myosmine intermediate) and the generation of substantial tarry byproducts that complicated purification. Furthermore, other established methods required hazardous reagents like sodium hydride or organolithium compounds operating at cryogenic temperatures of -78°C, posing severe safety risks and energy costs during scale-up. Perhaps most critically, resolution strategies for racemic nicotine inherently waste 50% of the material as the unwanted enantiomer, drastically inflating the cost of goods sold and creating significant waste disposal challenges.
The Novel Approach
In stark contrast to these legacy issues, the methodology outlined in CN114644614B introduces a streamlined, high-efficiency pathway that fundamentally reshapes the manufacturing landscape for this key alkaloid. The new process utilizes a substitution reaction between a nicotinic ester and N-vinylpyrrolidone, followed by a decarboxylation step to generate a key linear intermediate, which then undergoes cyclization to form myosmine. This approach eliminates the need for cryogenic conditions and dangerous pyrophoric reagents, operating instead at moderate temperatures ranging from 30°C to 120°C using standard bases like potassium tert-butoxide. The true breakthrough, however, is the subsequent asymmetric hydrogenation of myosmine, which installs the chiral center with exceptional precision, avoiding the 50% yield penalty associated with resolution. By integrating these steps, the process achieves a total yield of 50-60% for the final L-nicotine, demonstrating a level of atom economy and operational simplicity that is ideally suited for cost reduction in API manufacturing and large-scale industrial production.
Mechanistic Insights into Asymmetric Hydrogenation and Chiral Catalysis
The heart of this synthetic advancement lies in the third step: the enantioselective reduction of the myosmine double bond, which dictates the final optical purity of the product. This transformation is mediated by a sophisticated catalytic system generated in situ from a transition metal precursor and a specialized chiral ligand. As illustrated in 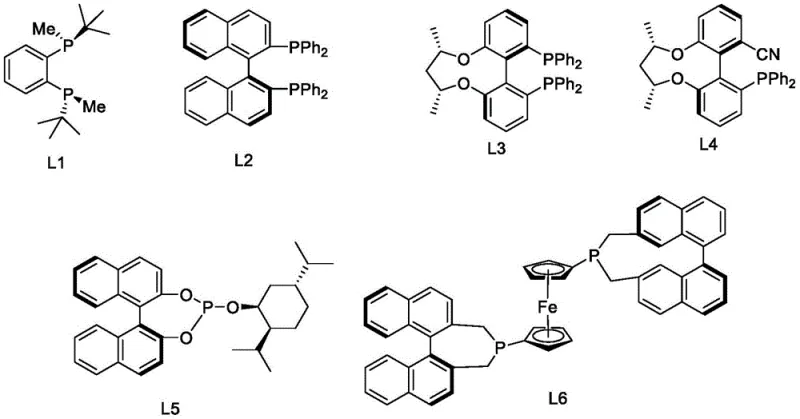 , the patent discloses a library of ligands (L1 through L6), including binaphtyl-based phosphines and ferrocene derivatives, which coordinate with metals such as Iridium, Rhodium, or Ruthenium. In the preferred embodiment, an Iridium complex paired with ligand L2 facilitates the hydrogenation under mild pressure (0.5-2.0 MPa) and temperature (20-40°C). The steric and electronic properties of these ligands create a chiral environment that strongly favors the formation of the (S)-enantiomer, effectively suppressing the formation of the (R)-isomer. This precise control over stereochemistry is what allows the process to consistently deliver L-nicotine with an optical purity greater than 99.9% ee, a specification that is difficult to achieve via extraction or resolution.
, the patent discloses a library of ligands (L1 through L6), including binaphtyl-based phosphines and ferrocene derivatives, which coordinate with metals such as Iridium, Rhodium, or Ruthenium. In the preferred embodiment, an Iridium complex paired with ligand L2 facilitates the hydrogenation under mild pressure (0.5-2.0 MPa) and temperature (20-40°C). The steric and electronic properties of these ligands create a chiral environment that strongly favors the formation of the (S)-enantiomer, effectively suppressing the formation of the (R)-isomer. This precise control over stereochemistry is what allows the process to consistently deliver L-nicotine with an optical purity greater than 99.9% ee, a specification that is difficult to achieve via extraction or resolution.
Beyond stereocontrol, the mechanism also offers significant advantages regarding impurity profiling and downstream processing. The use of homogeneous catalysis in a controlled solvent system (such as THF or methanol) ensures that side reactions, such as over-reduction of the pyridine ring or polymerization of the pyrrolidine moiety, are minimized. Following the hydrogenation, the intermediate nornicotine is subjected to a methylation step using formaldehyde and formic acid (Eschweiler-Clarke conditions), which is a robust and well-understood transformation. The combination of high-selectivity hydrogenation and clean methylation results in a crude product that requires minimal purification, typically just a pH adjustment and distillation. This streamlined impurity profile is crucial for regulatory compliance in pharmaceutical applications, where strict limits on genotoxic impurities and residual solvents must be maintained, thereby reducing the burden on QC labs and accelerating batch release times.
How to Synthesize L-Nicotine Efficiently
The synthesis of L-nicotine via this patented route is designed for operational robustness, moving from readily available starting materials to the final chiral product in just three distinct chemical transformations. The process begins with the coupling of a nicotinic ester and N-vinylpyrrolidone, followed by cyclization to myosmine, and concludes with the critical asymmetric hydrogenation and methylation sequence. This logical progression minimizes unit operations and maximizes throughput, making it an attractive candidate for technology transfer. For detailed laboratory protocols and specific reaction parameters, please refer to the standardized guide below.
- Substitution and Decarboxylation: React nicotinic ester with N-vinylpyrrolidone under basic conditions followed by acid treatment to form the key intermediate (Formula II).
- Cyclization to Myosmine: Treat the intermediate with a base to induce ring closure, yielding Myosmine with high efficiency.
- Asymmetric Hydrogenation and Methylation: Perform chiral hydrogenation on Myosmine using specific metal-ligand complexes, followed by methylation to obtain final L-nicotine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend far beyond simple chemical yield. By shifting away from agriculture-dependent extraction and inefficient resolution processes, manufacturers can secure a more predictable and stable supply of high-purity L-nicotine. The elimination of the resolution step alone represents a massive improvement in material efficiency, as it removes the inherent 50% loss of valuable feedstock associated with separating racemates. This direct synthesis approach ensures that every kilogram of raw material contributes to the final product, significantly lowering the effective cost of raw materials per kilogram of output. Furthermore, the use of commodity chemicals like ethyl nicotinate and N-vinylpyrrolidone, rather than custom-synthesized or hazardous reagents, mitigates supply risk and simplifies logistics, ensuring consistent availability even during market fluctuations.
- Cost Reduction in Manufacturing: The economic argument for this process is driven by the drastic simplification of the workflow and the elimination of wasteful steps. By avoiding the need for cryogenic cooling (-78°C) and hazardous reagents like sodium hydride, the process reduces energy consumption and safety infrastructure costs. Additionally, the high yield of the cyclization step (reported up to 97.8%) and the efficient hydrogenation minimize the volume of waste solvents and byproducts that require treatment. This reduction in waste generation translates directly into lower environmental compliance costs and reduced fees for hazardous waste disposal, contributing to substantial overall cost savings without compromising on quality.
- Enhanced Supply Chain Reliability: Dependence on tobacco crops introduces volatility due to weather patterns, pestilence, and geopolitical factors affecting agricultural regions. This fully synthetic route decouples production from these external variables, allowing for year-round manufacturing in controlled facility environments. The starting materials are stable, shelf-stable liquids or solids that can be sourced from multiple global suppliers, reducing the risk of single-source bottlenecks. This reliability is essential for maintaining continuous production schedules for downstream products like e-liquids or pharmaceutical formulations, ensuring that lead times for high-purity pharmaceutical intermediates remain short and predictable regardless of the harvest season.
- Scalability and Environmental Compliance: From a scale-up perspective, the reaction conditions are exceptionally forgiving, operating at near-ambient pressures and moderate temperatures that are easily managed in standard stainless steel reactors. The absence of pyrophoric reagents removes the need for specialized handling equipment, facilitating a smoother transition from pilot plant to commercial scale. Moreover, the process generates significantly less three-waste (waste water, gas, and solid) compared to resolution methods that require multiple acid-base swings and generate large volumes of salt waste. This cleaner profile aligns with modern green chemistry principles and stringent environmental regulations, making it easier to obtain operating permits and maintain a sustainable manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: What is the optical purity achievable with this new synthesis method?
A: According to patent CN114644614B, this method achieves an optical purity of greater than 99.9% ee, significantly surpassing the <95% purity typical of traditional extraction methods.
Q: How does this route improve upon traditional resolution methods?
A: Traditional resolution methods often suffer from a maximum theoretical yield of 50% due to the discard of the unwanted enantiomer. This asymmetric synthesis route avoids that loss, offering a total yield of 50-60% for the target L-isomer directly.
Q: Are there safety concerns with the reagents used in this process?
A: Unlike prior art routes that utilize hazardous reagents like sodium hydride (NaH) or organolithium compounds at cryogenic temperatures, this process operates at moderate temperatures (30-120°C) with safer, commercially available bases.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Nicotine Supplier
The technological advancements described in CN114644614B underscore the potential for producing L-nicotine with unprecedented purity and efficiency, setting a new benchmark for the industry. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chiral syntheses like this can be translated into reliable supply chains. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, including the >99.9% ee targets demanded by top-tier pharmaceutical and electronic cigarette clients. We understand that consistency is key, and our process engineering teams are dedicated to optimizing every step of the value chain to deliver premium quality intermediates.
We invite you to discuss how this innovative route can be integrated into your supply strategy to achieve superior cost-performance ratios. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data, route feasibility assessments, and samples to validate the quality of our L-nicotine against your internal standards. Let us partner with you to secure a sustainable and high-quality source of this critical chemical building block.
