Revolutionizing L-Nicotine Production: A Scalable, High-Purity Synthetic Route for Global Pharmaceutical Supply Chains
Introduction to Advanced L-Nicotine Synthesis Technology
The global demand for high-purity chiral intermediates in the pharmaceutical and electronic nicotine delivery sectors has necessitated a shift away from traditional extraction methods towards robust chemical synthesis. Patent CN114644614B, published in March 2023, introduces a groundbreaking preparation method for L-nicotine that addresses critical limitations in purity and scalability. This technology enables the production of L-nicotine with an optical purity exceeding 99.9% ee, a specification that far surpasses the typical <95% purity found in plant-extracted products which often contain carcinogenic alkaloid impurities. By leveraging a concise three-step synthetic pathway involving substitution, cyclization, and asymmetric hydrogenation, this method offers a reliable pharma intermediate supplier solution that ensures consistent quality independent of agricultural variables like climate or land resources.
For R&D directors and procurement managers, the significance of this patent lies in its ability to decouple supply from the volatility of tobacco farming while simultaneously enhancing product safety profiles. The synthetic route utilizes readily available nicotinic acid esters and N-vinylpyrrolidone, avoiding the complex purification trains associated with biological sources. Furthermore, the process achieves a total synthesis yield of 50-60%, demonstrating high atom economy and operational efficiency. This report analyzes the mechanistic advantages, commercial viability, and supply chain implications of adopting this advanced manufacturing protocol for the production of high-purity L-nicotine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on extracting nicotine from tobacco leaves, a process fraught with inconsistencies and safety concerns. Since tobacco leaves contain a diverse array of alkaloids that are difficult to separate, the resulting L-nicotine typically possesses a purity of less than 95% and is contaminated with other biologically active compounds, some of which are proven carcinogens. On the synthetic front, earlier attempts to manufacture nicotine chemically have faced significant hurdles regarding safety and yield. For instance, legacy routes depicted in prior art often required the use of highly dangerous reagents such as sodium hydride (NaH) as a base.
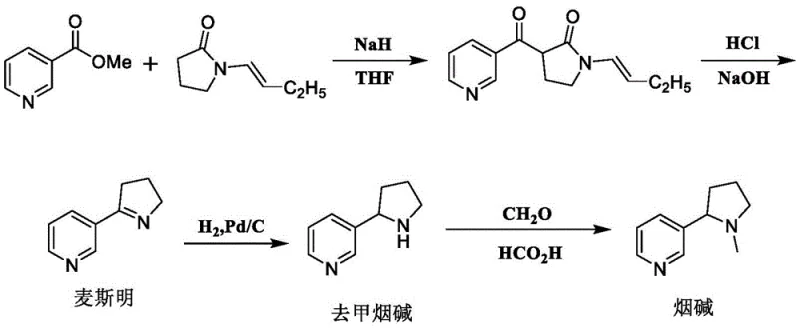
As illustrated in the reaction scheme above, older methodologies involving N-butenylpyrrolidone necessitated the in-situ generation of hazardous intermediates and produced large volumes of hydrogen gas during the reaction, creating substantial safety hazards for scale-up production. Additionally, alternative routes utilizing organometallic lithium reagents required cryogenic conditions at -78°C, leading to prohibitive energy costs and operational complexity. Other approaches involving enzymatic catalysis, while avoiding some chemical hazards, introduced trace protein residues that are difficult to detect and quantify, rendering the final product unsuitable for certain high-purity pharmaceutical applications. These cumulative drawbacks highlight the urgent need for a fully synthetic, non-biological route that guarantees both safety and exceptional purity.
The Novel Approach
The methodology disclosed in CN114644614B represents a paradigm shift by employing a streamlined, three-step chemical synthesis that eliminates the need for hazardous cryogenics or biological fermentation. The core innovation lies in the initial coupling of a nicotinic acid ester with N-vinylpyrrolidone under mild basic conditions, followed by a controlled decarboxylation to generate a stable ketone intermediate. This intermediate then undergoes a highly efficient cyclization to form Myosmine, which serves as the precursor for the final stereoselective step. Unlike resolution methods that discard 50% or more of the material as the unwanted enantiomer, this route builds chirality directly or preserves it through asymmetric catalysis, thereby maximizing material throughput.
The superiority of this novel approach is evident in its operational simplicity and environmental profile. By avoiding the use of explosive sodium hydride and replacing it with safer alkali bases like potassium tert-butoxide or sodium hydroxide, the process significantly reduces the risk profile for commercial manufacturing. Furthermore, the elimination of biological fermentation steps ensures that the final product is free from protein residues, a critical quality attribute for regulatory compliance in sensitive applications. The result is a robust, scalable process capable of delivering L-nicotine with >99.9% optical purity, setting a new benchmark for cost reduction in pharma intermediate manufacturing.
Mechanistic Insights into Asymmetric Hydrogenation and Chiral Catalysis
The cornerstone of achieving >99.9% optical purity in this synthesis is the asymmetric hydrogenation step, which transforms the achiral or racemic Myosmine intermediate into the desired chiral L-nicotine scaffold. This transformation is mediated by a sophisticated catalyst system generated in situ from a transition metal precursor and a specialized chiral ligand. The patent specifies the use of metals such as Iridium (e.g., Ir(COD)Cl2), Rhodium, or Ruthenium, coordinated with bulky, electron-rich phosphine ligands. These ligands create a chiral environment around the metal center, effectively differentiating between the two faces of the substrate during the hydrogen addition.
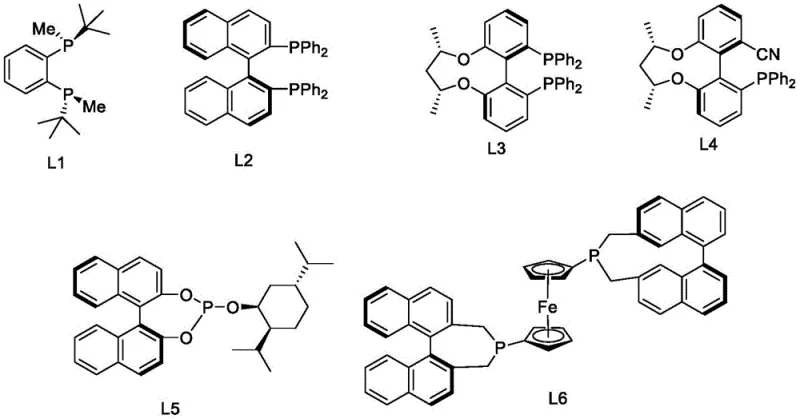
As shown in the ligand structures above, the steric bulk and electronic properties of ligands like L2 (BINAP derivatives) or L5/L6 (ferrocene-based or bulky phosphines) are critical for inducing high enantioselectivity. During the catalytic cycle, the metal-hydride species coordinates to the imine or enamine functionality of the Myosmine derivative. The chiral pocket formed by the ligand forces the hydrogen transfer to occur exclusively from one spatial direction, thereby establishing the (S)-configuration at the chiral center with exceptional fidelity. This mechanism bypasses the need for classical resolution, where half the material is lost, and instead drives the reaction towards a single enantiomer with high efficiency. The subsequent N-methylation using formaldehyde and formic acid (Eschweiler-Clarke type conditions) proceeds without racemization, preserving the high optical purity established in the hydrogenation step.
From an impurity control perspective, this catalytic system is advantageous because it operates under homogeneous conditions that allow for precise tuning of reaction parameters such as temperature (20-40°C) and hydrogen pressure (1.0-1.5 MPa). This controllability minimizes the formation of side products like over-reduced species or regio-isomers, which are common pitfalls in non-selective hydrogenations. The ability to fine-tune the ligand-to-metal ratio (e.g., 1.5:1 to 10:1) provides process chemists with a powerful lever to optimize both reaction rate and stereoselectivity, ensuring that the final API intermediate meets stringent regulatory specifications for chiral purity.
How to Synthesize L-Nicotine Efficiently
The synthesis of L-nicotine via this patented route is designed for operational robustness, utilizing common industrial solvents like xylene, THF, and ethyl acetate. The process begins with the alkylation of ethyl nicotinate with N-vinylpyrrolidone, followed by acid-mediated decarboxylation to yield the linear ketone intermediate. This intermediate is then cyclized under basic conditions to form Myosmine, which is subsequently subjected to asymmetric hydrogenation and methylation. The detailed standardized synthesis steps, including specific reagent ratios, temperature profiles, and workup procedures, are outlined below to guide process implementation.
- Perform a substitution reaction between a nicotinic acid ester and N-vinylpyrrolidone under basic conditions, followed by acid-catalyzed decarboxylation to yield the key intermediate (Formula II).
- Execute a base-mediated cyclization of the Formula II intermediate to form Myosmine with high conversion efficiency.
- Conduct an asymmetric hydrogenation of Myosmine using a chiral metal-ligand catalyst system, followed by N-methylation to obtain the final L-nicotine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative advantages in terms of cost stability and supply security. Traditional extraction methods are inherently vulnerable to agricultural disruptions, including crop failures, climate change impacts, and geopolitical instability in tobacco-growing regions. By shifting to a fully synthetic pathway based on petrochemical-derived feedstocks like nicotinic esters, manufacturers can insulate their supply chains from these external volatilities. The raw materials required for this process are commodity chemicals available from multiple global suppliers, ensuring a continuous and reliable flow of inputs for large-scale production campaigns.
- Cost Reduction in Manufacturing: The economic case for this technology is driven by the elimination of expensive and wasteful processing steps. Unlike chiral resolution methods that inherently discard approximately half of the synthesized material as the unwanted enantiomer, this asymmetric synthesis builds the desired chirality directly, effectively doubling the theoretical yield from the starting materials. Furthermore, the avoidance of hazardous reagents like sodium hydride reduces the need for specialized safety infrastructure and expensive waste disposal protocols associated with reactive metal byproducts. The high overall yield of 50-60% across three steps significantly lowers the cost of goods sold (COGS) compared to lower-yielding multi-step sequences.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions contributes to greater manufacturing reliability and shorter lead times. The process operates at moderate temperatures (30-100°C) and pressures, avoiding the extreme cryogenic conditions (-78°C) required by older organolithium routes. This reduces energy consumption and allows the use of standard stainless steel reactors rather than specialized low-temperature vessels. Additionally, the absence of biological fermentation steps means there is no risk of batch failure due to microbial contamination or enzyme deactivation, leading to more predictable production schedules and consistent on-time delivery for customers.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns with modern green chemistry principles. The replacement of toxic solvents and hazardous reagents with safer alternatives simplifies the permitting process for new manufacturing facilities. The reduction in three wastes (wastewater, waste gas, solid waste) is significant, particularly when compared to resolution processes that generate large volumes of salt waste from acid-base cycling. The high purity of the final product (>99.9% ee) also minimizes the need for extensive downstream purification, further reducing solvent usage and energy intensity, making it an ideal candidate for sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this L-nicotine synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in patent CN114644614B, providing clarity on purity standards, safety protocols, and scalability potential for potential partners and licensees.
Q: How does this synthetic method compare to traditional tobacco extraction in terms of purity?
A: Traditional extraction from tobacco often yields nicotine with less than 95% purity and contains various carcinogenic alkaloid impurities. The synthetic method described in patent CN114644614B achieves an optical purity exceeding 99.9% ee, significantly reducing harmful contaminants.
Q: What are the safety advantages of this new route over previous synthetic methods?
A: Previous synthetic routes often utilized hazardous reagents like sodium hydride (NaH) which generates explosive hydrogen gas, posing significant scale-up risks. The new method employs safer bases and controlled hydrogenation conditions, eliminating these major safety hazards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability with a total synthesis yield of 50-60%. It utilizes cheap, readily available raw materials and avoids complex biological fermentation steps that introduce protein residues, ensuring a robust supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Nicotine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN114644614B can be realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific requirements of asymmetric hydrogenation, including high-pressure reactors and rigorous QC labs capable of verifying stringent purity specifications such as the >99.9% ee target.
We invite forward-thinking pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced synthesis route for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your project benefits from the highest standards of quality, safety, and efficiency in the industry.
