Optimizing Fluazinam Intermediate Production: A Novel Normal Pressure Amination Strategy
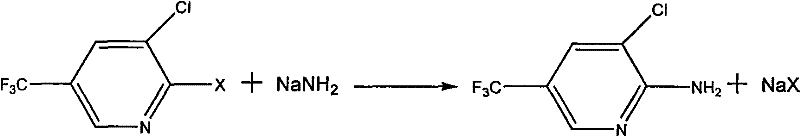
The global demand for high-efficiency fungicides continues to drive innovation in the synthesis of critical agrochemical intermediates, specifically focusing on the production of 2-amino-3-chloro-5-(trifluoromethyl)pyridine. This compound serves as a pivotal building block in the manufacturing of Fluazinam, a broad-spectrum protective fungicide widely utilized in modern agriculture. Patent CN101709051A introduces a transformative preparation method that addresses long-standing inefficiencies in this synthetic pathway. By leveraging sodium amide as a primary aminating reagent in conjunction with phase transfer catalysis, this technology enables the conversion of 2-halo-3-chloro-5-(trifluoromethyl)pyridine derivatives under remarkably moderate conditions. Unlike legacy processes that rely on hazardous high-pressure environments or suffer from dismal conversion rates, this novel approach operates effectively at normal pressure with temperatures ranging from 10°C to 220°C. For R&D directors and procurement specialists, this represents a significant opportunity to enhance the reliability of the agrochemical intermediate supplier network while simultaneously optimizing the cost structure of the final active ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this pyridine derivative has been plagued by significant technical and economic barriers that hinder efficient commercial scale-up of complex agrochemical intermediates. Early methodologies, such as those disclosed in EP0031218, relied on ammoniacal liquor as the nitrogen source, resulting in extremely low yields of approximately 25% even after prolonged reaction times of 24 hours at 125°C. Such inefficiency translates directly into excessive raw material waste and inflated production costs, making the supply chain vulnerable to price volatility. Subsequent improvements, like CN101081833A, attempted to resolve yield issues by utilizing liquefied ammonia; however, this introduced severe safety and infrastructure challenges. These high-pressure methods require specialized autoclaves capable of withstanding 3.5MPa at 130°C, necessitating rigorous working condition harshness and substantial capital investment in pressure-rated equipment. Furthermore, the handling of liquefied ammonia poses inherent safety risks regarding storage and transportation, complicating the logistics for any reliable agrochemical intermediate supplier aiming for consistent delivery.
The Novel Approach
The methodology outlined in CN101709051A fundamentally reengineers the reaction landscape by substituting hazardous high-pressure ammonia systems with a solid-state sodium amide protocol. This shift allows the reaction to proceed under normal pressure, drastically simplifying the reactor requirements and enhancing overall plant safety profiles. The integration of phase transfer catalysts plays a crucial role in overcoming the solubility limitations of sodium amide powder in organic media, thereby accelerating the reaction kinetics without the need for extreme thermal inputs. By operating within a temperature window of 120°C to 180°C, the process achieves yields exceeding 90% in optimized embodiments, a stark contrast to the quarter-yield of older atmospheric techniques. This breakthrough not only ensures cost reduction in agrochemical intermediate manufacturing but also streamlines post-treatment procedures, as the byproduct sodium halide can be easily filtered off, leaving the desired product in the filtrate for straightforward precipitation and purification.
Mechanistic Insights into Sodium Amide Mediated Nucleophilic Substitution
The core chemical transformation driving this synthesis is a nucleophilic aromatic substitution where the amino group displaces a halogen atom on the pyridine ring. In this specific system, sodium amide acts as a potent source of the nucleophilic amide ion, which attacks the electron-deficient carbon at the 2-position of the pyridine ring, activated by the electron-withdrawing trifluoromethyl and chloro groups. The presence of a phase transfer catalyst, such as quaternary ammonium salts or crown ethers, is mechanistically vital as it facilitates the transport of the amide anion into the organic phase where the substrate resides. This interfacial activity significantly lowers the activation energy required for the substitution, allowing the reaction to proceed efficiently at moderate temperatures rather than requiring the extreme conditions seen in non-catalyzed variants. The selection of the leaving group, preferably fluorine or chlorine, further influences the reaction rate, with fluorine typically offering superior leaving group ability in nucleophilic aromatic substitutions due to its high electronegativity stabilizing the Meisenheimer complex intermediate.
Controlling the impurity profile is paramount for ensuring the downstream efficacy of the fungicide, and this mechanism offers inherent advantages in purity management. By avoiding the use of aqueous ammoniacal solutions, the process minimizes the risk of hydrolysis side reactions that could generate hydroxyl-substituted byproducts or degrade the sensitive trifluoromethyl moiety. The use of anhydrous organic solvents like DMF, xylene, or decane creates a controlled environment where side reactions are kinetically suppressed. Furthermore, the stoichiometry can be precisely managed, with a molar ratio of sodium amide to substrate between 1:1 and 3:1, ensuring complete conversion of the starting material without excessive excess that would complicate workup. The resulting crude product typically exhibits purity levels above 95% after simple filtration and washing, reducing the burden on downstream purification units and ensuring that the high-purity agrochemical intermediate meets stringent specifications required by global regulatory bodies for pesticide registration.
How to Synthesize 2-Amino-3-Chloro-5-(Trifluoromethyl)Pyridine Efficiently
Implementing this synthesis route requires careful attention to reagent quality and atmospheric control to maximize the benefits of the phase transfer catalysis system. The process begins with the preparation of a reaction mixture containing the chosen solvent and catalyst, followed by the gradual addition of sodium amide powder under inert gas protection to prevent moisture ingress which could deactivate the reagent. Once the slurry is prepared, the halogenated pyridine substrate is introduced, and the system is heated to reflux conditions tailored to the specific solvent boiling point. Monitoring the reaction progress is essential to determine the optimal endpoint, typically achieved within 0.5 to 24 hours depending on the specific catalyst and temperature profile employed. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety protocols required for laboratory and pilot scale execution.
- Prepare the reaction system by adding sodium amide powder into a selected solvent such as DMF or xylene, optionally incorporating a phase transfer catalyst like methyltributylammonium chloride.
- Introduce the substrate 2-halo-3-chloro-5-(trifluoromethyl)pyridine into the mixture under nitrogen protection while maintaining stirring.
- Heat the reaction mixture to between 120°C and 180°C, monitor for completion, then filter off sodium halide salts and precipitate the product via decompression.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this normal pressure amination technology offers compelling strategic advantages that extend beyond simple chemical yield improvements. The elimination of high-pressure reactors removes a significant bottleneck in capacity expansion, allowing manufacturers to utilize standard glass-lined or stainless steel vessels that are more readily available and easier to maintain. This flexibility enhances supply chain reliability by reducing the dependency on specialized, long-lead-time equipment, thereby shortening the timeline for bringing new production lines online. Additionally, the use of solid sodium amide simplifies raw material logistics compared to managing bulk liquefied ammonia, which requires dedicated cryogenic storage and transport infrastructure. These operational simplifications translate into substantial cost savings and a more resilient supply network capable of withstanding market fluctuations and regulatory changes regarding hazardous material transport.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the reaction apparatus and the reduction in energy consumption associated with high-pressure maintenance. By operating at normal pressure, the facility avoids the capital expenditure and insurance costs linked to high-pressure autoclaves, while the improved yield reduces the cost of goods sold per kilogram of active intermediate. The ability to use common organic solvents and recoverable phase transfer catalysts further optimizes the material balance, ensuring that waste generation is minimized and raw material utilization is maximized. This holistic efficiency gain allows for a more competitive pricing structure without compromising on the quality or purity of the final agrochemical intermediate supplied to formulators.
- Enhanced Supply Chain Reliability: Stability in the supply of critical fungicide intermediates is often threatened by the complexity of the manufacturing process, but this method mitigates those risks through operational robustness. The mild reaction conditions reduce the likelihood of unplanned shutdowns due to equipment failure or safety incidents, ensuring consistent output volumes throughout the production cycle. Furthermore, the availability of sodium amide and standard organic solvents in the global chemical market ensures that raw material sourcing remains stable, preventing disruptions that could delay lead time for high-purity agrochemical intermediates. This reliability is crucial for downstream partners who depend on just-in-time delivery schedules to maintain their own formulation and packaging operations without interruption.
- Scalability and Environmental Compliance: Scaling this process from laboratory bench to commercial tonnage is straightforward due to the absence of complex pressure dynamics that often behave unpredictably at larger volumes. The simplified post-treatment, involving filtration and precipitation, generates less wastewater contaminated with ammonia compared to traditional methods, easing the burden on effluent treatment plants. This aligns with increasingly strict environmental regulations, reducing the compliance risk and potential fines associated with nitrogen-rich waste streams. The process inherently supports green chemistry principles by improving atom economy and reducing the need for hazardous reagents, making it a sustainable choice for long-term commercial scale-up of complex agrochemical intermediates in a regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, yield, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific manufacturing contexts and supply chain requirements.
Q: How does this method improve safety compared to traditional liquid ammonia processes?
A: This protocol utilizes solid sodium amide under normal pressure, eliminating the need for high-pressure autoclaves required by liquid ammonia methods, thereby significantly reducing operational risks and equipment costs.
Q: What yields can be expected from this catalytic amination route?
A: Experimental data indicates yields ranging from 83% to over 94% with purity exceeding 95%, representing a substantial improvement over older atmospheric methods that yielded only 25%.
Q: Is this process scalable for industrial manufacturing of fungicide intermediates?
A: Yes, the use of common organic solvents and standard heating equipment without high-pressure constraints makes this route highly adaptable for large-scale commercial production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-Chloro-5-(Trifluoromethyl)Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced agrochemicals depends on the availability of high-quality intermediates produced via robust and scalable pathways. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of market demand surges. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-amino-3-chloro-5-(trifluoromethyl)pyridine meets the exacting standards required for Fluazinam synthesis. We combine technical expertise with operational excellence to deliver products that empower your R&D and manufacturing teams to focus on innovation rather than supply chain uncertainties.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your production efficiency and reduce overall manufacturing costs. Let us be your partner in driving the future of sustainable and efficient agrochemical production.
