Advanced Normal-Pressure Synthesis of 2-Amino-3-Chloro-5-(Trifluoromethyl)Pyridine for Commercial Scale-Up
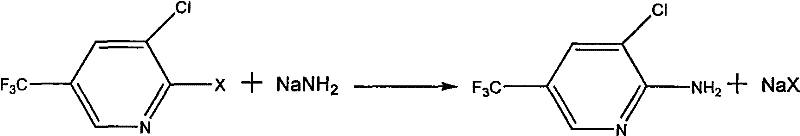
The global demand for high-performance fungicides continues to drive innovation in the synthesis of fluorinated heterocyclic intermediates, specifically within the agrochemical sector. Patent CN101709051A introduces a transformative preparation method for 2-amino-3-chloro-5-(trifluoromethyl)pyridine, a critical building block for the synthesis of Fluazinam and related protective agents. This technology represents a significant departure from legacy methodologies that relied on hazardous high-pressure conditions or suffered from prohibitively low conversion rates. By utilizing sodium amide as the primary aminating reagent in conjunction with phase transfer catalysis, the process achieves high efficiency under normal atmospheric pressure. This technical breakthrough not only simplifies the operational workflow but also aligns with modern manufacturing standards that prioritize safety and scalability. For R&D directors and procurement strategists, understanding the nuances of this synthetic route is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality without the burden of complex high-pressure infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-3-chloro-5-(trifluoromethyl)pyridine has been plagued by significant technical and economic hurdles that hindered large-scale production. Early methods, such as those disclosed in EP0031218, utilized aqueous ammonia at elevated temperatures around 125°C, resulting in dismal yields of approximately 25%, which is commercially unsustainable for high-volume manufacturing. Subsequent improvements, like those found in CN101081833A, attempted to address the yield issue by employing liquid ammonia; however, this introduced severe engineering challenges by requiring reaction conditions of 130°C under a high pressure of 3.5MPa. Such high-pressure environments necessitate specialized, expensive autoclave equipment and rigorous safety protocols, drastically increasing capital expenditure and operational complexity. Furthermore, the handling of large quantities of liquefied ammonia poses inherent safety risks regarding leakage and containment, creating substantial liability for production facilities. These conventional approaches often result in difficult post-treatment procedures where the separation of products from excess ammonia and byproducts becomes energy-intensive and time-consuming, ultimately inflating the cost of goods sold.
The Novel Approach
The methodology outlined in patent CN101709051A effectively circumvents these historical bottlenecks by shifting the reaction paradigm to a solid-liquid heterogeneous system using sodium amide powder. This novel approach operates successfully at normal pressure, thereby eliminating the need for costly high-pressure reactors and allowing the use of standard glass-lined or stainless steel vessels commonly found in fine chemical plants. The reaction proceeds smoothly within a temperature range of 10-220°C, with optimal results observed between 120-180°C, providing a wide operational window that enhances process robustness. A key innovation is the optional inclusion of phase transfer catalysts, such as quaternary ammonium salts or crown ethers, which significantly accelerate the reaction kinetics by solubilizing the ionic sodium amide in organic media. This catalytic enhancement allows for shorter reaction times and higher conversions, with experimental examples demonstrating yields exceeding 90% and purities reaching up to 98%. The post-treatment is remarkably straightforward, involving simple filtration to remove sodium halide salts followed by precipitation, which streamlines the isolation of the final high-purity agrochemical intermediate.
Mechanistic Insights into Sodium Amide Mediated Nucleophilic Substitution
The core chemical transformation driving this synthesis is a nucleophilic aromatic substitution (SnAr) where the amino group replaces a halogen atom at the 2-position of the pyridine ring. The presence of the strongly electron-withdrawing trifluoromethyl group at the 5-position and the chlorine atom at the 3-position significantly activates the pyridine ring towards nucleophilic attack, particularly at the C-2 position which is ortho to the ring nitrogen. In this mechanism, the amide ion (NH2-) derived from sodium amide acts as a potent nucleophile, attacking the electron-deficient carbon center to form a Meisenheimer-like intermediate. The subsequent elimination of the leaving group, typically fluoride or chloride depending on the starting material, restores the aromaticity of the pyridine system and yields the desired 2-amino derivative. The choice of sodium amide over ammonia is critical because the amide ion is a much stronger base and nucleophile, facilitating the reaction under milder thermal conditions compared to neutral ammonia molecules.
Furthermore, the role of the phase transfer catalyst (PTC) cannot be overstated in optimizing this mechanism for industrial application. Since sodium amide is an ionic solid with poor solubility in non-polar organic solvents like alkanes or aromatics, the reaction would otherwise be limited by mass transfer constraints at the solid-liquid interface. The PTC, such as methyltributylammonium chloride or 18-crown-6, complexes with the sodium cation or forms lipophilic ion pairs with the amide anion, effectively transporting the reactive species into the bulk organic phase where the substrate resides. This homogenization of the reaction environment ensures that the nucleophilic attack occurs rapidly and uniformly, minimizing side reactions and improving the overall impurity profile. By carefully selecting the solvent system—ranging from polar aprotic solvents like DMF to non-polar hydrocarbons like decane—chemists can fine-tune the solubility parameters to maximize the efficiency of the catalytic cycle. This mechanistic understanding allows for precise control over the reaction trajectory, ensuring that the formation of the target molecule is favored over potential degradation pathways or polymerization.
How to Synthesize 2-Amino-3-Chloro-5-(Trifluoromethyl)Pyridine Efficiently
To implement this synthesis effectively, operators must adhere to strict protocols regarding reagent addition and temperature control to ensure safety and reproducibility. The process begins with the suspension of finely pulverized sodium amide powder in a chosen solvent, such as DMF or xylene, under an inert nitrogen atmosphere to prevent moisture ingress which could deactivate the reagent. This standardized approach ensures that the exothermic nature of the amination is managed correctly while maintaining the integrity of the sensitive fluorinated intermediates throughout the production batch.
- Prepare the reaction mixture by adding sodium amide powder into a suitable solvent such as DMF, xylene, or decane, optionally including a phase transfer catalyst like methyltributylammonium chloride.
- Add 2-halo-3-chloro-5-(trifluoromethyl)pyridine to the mixture under nitrogen protection and heat to a temperature range of 120-180°C.
- Upon completion, filter the reaction mixture to remove sodium halide salts, then precipitate the product from the filtrate via decompression, followed by washing and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sodium amide-based process offers profound strategic advantages that extend beyond mere chemical yield. The most immediate benefit is the drastic reduction in capital expenditure (CAPEX) associated with reactor infrastructure, as the elimination of high-pressure requirements allows manufacturers to utilize existing standard equipment rather than investing in specialized autoclaves. This flexibility significantly lowers the barrier to entry for production and enhances the agility of the supply chain, enabling faster scale-up from pilot batches to commercial tonnage without lengthy equipment procurement lead times. Additionally, the use of solid sodium amide simplifies logistics and storage compared to the handling of bulk liquid ammonia, which requires cryogenic tanks or high-pressure cylinders, thereby reducing HazMat transportation costs and regulatory compliance burdens. The simplified post-treatment workflow, which relies on filtration and precipitation rather than complex distillation or extraction sequences, further contributes to substantial cost savings by reducing energy consumption and solvent usage per kilogram of product.
- Cost Reduction in Manufacturing: The transition to a normal-pressure process fundamentally alters the cost structure of producing this key intermediate by removing the need for expensive high-pressure rated vessels and the associated safety maintenance protocols. By utilizing readily available sodium amide and common organic solvents, the raw material costs remain stable and predictable, avoiding the volatility often seen with specialized reagents. The high conversion rates achieved through phase transfer catalysis mean that less starting material is wasted, directly improving the atom economy and reducing the cost of goods sold. Furthermore, the ability to recover and recycle solvents like xylene or decane adds another layer of economic efficiency, making the overall process highly competitive in the global market for agrochemical intermediates.
- Enhanced Supply Chain Reliability: Relying on a synthesis route that does not depend on high-pressure ammonia infrastructure mitigates the risk of production stoppages due to equipment failure or regulatory inspections of pressure systems. The robustness of the reaction conditions, which tolerate a wide temperature range and various solvent systems, ensures consistent output even when facing minor fluctuations in utility supplies or raw material quality. This reliability is crucial for downstream customers who require just-in-time delivery of intermediates for their own formulation processes, as it minimizes the risk of stockouts. Moreover, the simplicity of the workup procedure allows for shorter batch cycles, increasing the overall throughput of the manufacturing facility and ensuring a steady flow of product to meet market demand.
- Scalability and Environmental Compliance: The environmental footprint of this process is significantly lower than traditional methods, primarily due to the absence of venting large volumes of gaseous ammonia and the reduced energy intensity of the reaction. The solid byproduct, sodium halide, is easily separated by filtration and can often be disposed of or treated with minimal environmental impact compared to complex aqueous waste streams. The use of phase transfer catalysts in catalytic amounts rather than stoichiometric quantities further reduces the chemical load in the waste stream, aligning with green chemistry principles. This compliance with stringent environmental regulations facilitates smoother permitting processes for new production lines and enhances the sustainability profile of the supply chain, which is increasingly important for multinational agrochemical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity for stakeholders evaluating its feasibility. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industrial application.
Q: What are the safety advantages of using sodium amide over liquid ammonia for this synthesis?
A: The use of sodium amide allows the reaction to proceed at normal atmospheric pressure, eliminating the need for high-pressure autoclaves (3.5MPa) required by traditional liquid ammonia methods, thereby significantly reducing equipment costs and operational risks.
Q: How does the phase transfer catalyst improve the reaction efficiency?
A: Since sodium amide is insoluble in many organic solvents, the phase transfer catalyst facilitates the transport of the amide anion into the organic phase, enhancing the reaction rate and allowing for milder temperature conditions compared to uncatalyzed processes.
Q: What is the typical purity and yield achievable with this patented method?
A: According to the experimental data in patent CN101709051A, this method consistently achieves yields between 83% and 95% with product purity ranging from 95% to 98%, surpassing the 25% yield of older aqueous ammonia methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-Chloro-5-(Trifluoromethyl)Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of this intermediate in the global agrochemical supply chain and have positioned ourselves as a leader in its commercial production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of major fungicide manufacturers without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-amino-3-chloro-5-(trifluoromethyl)pyridine meets the exacting standards required for API and crop protection synthesis. Our commitment to process excellence means that we can consistently deliver material with the high purity profiles demonstrated in the patent examples, supporting your R&D and manufacturing goals with unwavering reliability.
We invite you to engage with our technical procurement team to discuss how our optimized synthesis capabilities can drive value for your organization. By partnering with us, you gain access to a Customized Cost-Saving Analysis that details how our efficient manufacturing processes can lower your total landed costs. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless integration of our high-quality intermediates into your supply chain.
