Industrial Scale Synthesis of Shakubiqu Intermediate via Chiral Hydrogenation and Enzymatic Resolution
Industrial Scale Synthesis of Shakubiqu Intermediate via Chiral Hydrogenation and Enzymatic Resolution
The pharmaceutical landscape for heart failure treatment was significantly altered by the approval of Entresto, a combination drug containing Valsartan and Shakubiqu (Sacubitril). As demand for this life-saving medication grows, the efficiency of its supply chain becomes paramount. Patent CN113121342B discloses a groundbreaking preparation method for a key Shakubiqu intermediate, offering a robust alternative to legacy synthesis routes. This technology leverages inexpensive raw materials like Itaconic anhydride and employs advanced catalytic asymmetric hydrogenation alongside biocatalytic resolution. By shifting the chiral induction to the very beginning of the synthesis, this method drastically simplifies the downstream processing. The structural complexity of the final active pharmaceutical ingredient requires precise control over stereochemistry, which this new route addresses with exceptional fidelity.
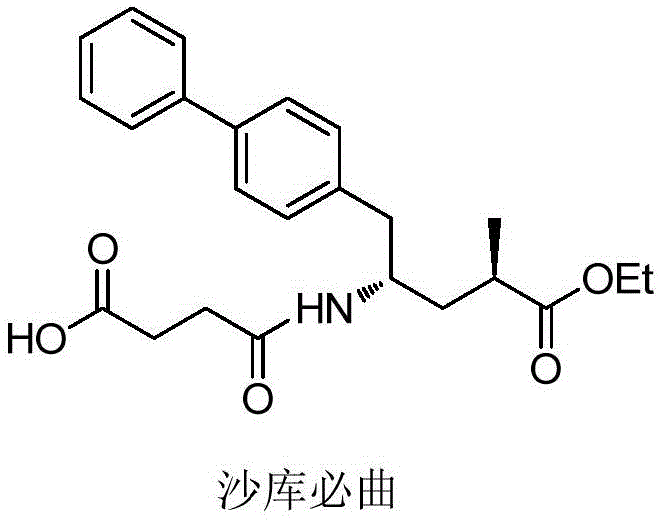
This innovative approach not only enhances the purity profile of the resulting intermediate but also aligns with green chemistry principles by utilizing enzymatic steps. For global procurement teams, understanding the nuances of this synthesis is critical for securing a reliable pharmaceutical intermediates supplier. The patent details a pathway that minimizes waste and maximizes yield, directly impacting the cost of goods sold (COGS) for the final API. As we delve deeper into the technical specifics, it becomes evident that this methodology represents a significant leap forward in the commercial scale-up of complex pharmaceutical intermediates, ensuring a stable supply for the growing market of cardiovascular therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing Shakubitril and its precursors have historically been plagued by inefficiencies that hinder large-scale production. For instance, earlier patents such as WO2008031567A1 describe routes relying on the oxidation of amino-substituted biphenylpropanol followed by Wittig reactions and subsequent metal-catalyzed chiral resolution. These traditional pathways often necessitate the use of expensive chiral ligands and precious metal catalysts at late stages of the synthesis, which complicates purification and increases the risk of metal contamination in the final product. Furthermore, these routes frequently involve multiple protection and deprotection steps for carboxyl and amino groups, leading to a proliferation of unit operations and a significant reduction in overall atom economy. The reliance on difficult-to-source chiral starting materials also introduces supply chain vulnerabilities, making cost reduction in API manufacturing challenging.
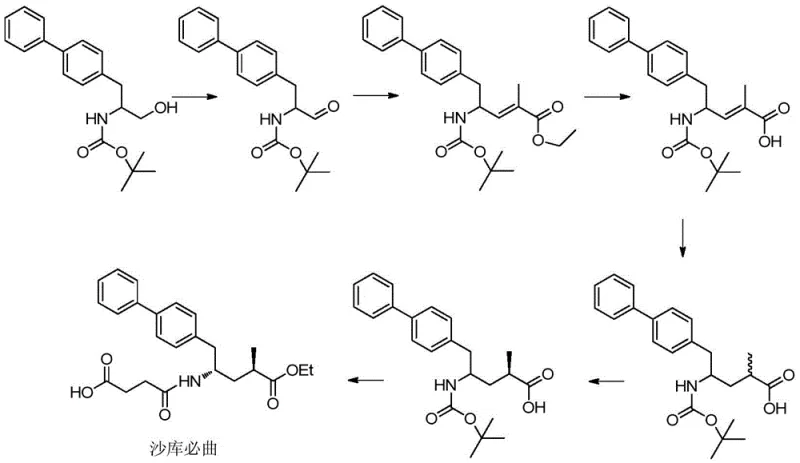
The Novel Approach
In stark contrast, the method disclosed in CN113121342B initiates the synthesis from Itaconic anhydride, a commodity chemical that is both economically viable and abundantly available. This novel strategy employs a chiral metal catalyst, specifically a Ruthenium complex with (S,S)-Me-Duphos ligand, to perform an asymmetric hydrogenation right at the outset. This early introduction of chirality sets the stereochemical foundation for the entire molecule, eliminating the need for difficult resolutions later in the sequence. The process continues with a highly selective enzymatic hydrolysis using lipase, which operates under mild conditions to differentiate between ester groups without the need for harsh chemical reagents. This streamlined approach significantly reduces the number of reaction steps and avoids the cumbersome protection strategies seen in older methodologies, thereby facilitating a more direct and cost-effective path to the key ketone intermediate.
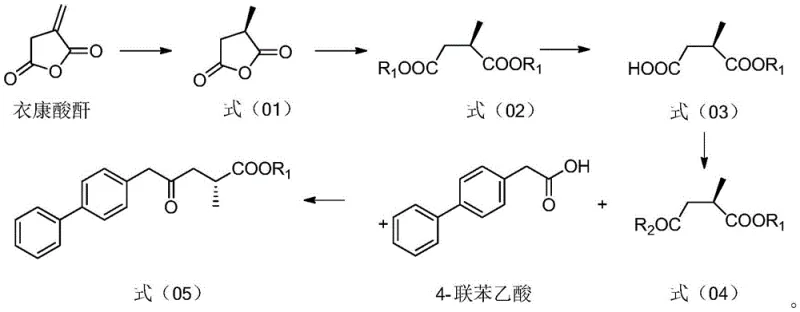
Mechanistic Insights into Chiral Hydrogenation and Enzymatic Resolution
The core of this technological advancement lies in the precise control of stereochemistry through transition metal catalysis and biocatalysis. The initial step involves the hydrogenation of Itaconic anhydride under a hydrogen pressure of 0.1-10 MPa, preferably 5 MPa, using a catalyst system comprising a Group 8 transition metal like Ruthenium and the chiral ligand (S,S)-Me-Duphos. This specific ligand-metal combination creates a chiral environment that favors the formation of the R-enantiomer of methyl succinic anhydride with an enantiomeric excess (ee) value exceeding 99%. Following this, the anhydride is esterified to a diester, which then undergoes a kinetic resolution mediated by lipase (such as LipozymeCALB). The enzyme selectively hydrolyzes one of the ester bonds in a phosphate buffer solution at a controlled pH of roughly 7, yielding a mono-acid with high optical purity. This dual-catalyst strategy ensures that the chiral integrity is maintained and enhanced throughout the early stages of the synthesis.
Subsequent steps involve the activation of the carboxylic acid to an acid chloride using oxalyl chloride, followed by a coupling reaction with 4-biphenylacetic acid in the presence of a strong base like isopropyl magnesium chloride. This coupling forms the carbon-carbon bond necessary to construct the biphenyl backbone of the Shakubitril molecule. The final transformation involves a transaminase-catalyzed reaction to introduce the amino group, followed by amidation with succinic anhydride. The transaminase step is particularly noteworthy as it replaces traditional reductive amination which might require high-pressure hydrogenation or stoichiometric reducing agents. The final amidation condenses the amine intermediate with succinic anhydride to form the target succinimide-like structure found in Shakubitril, completing the synthesis with high fidelity.
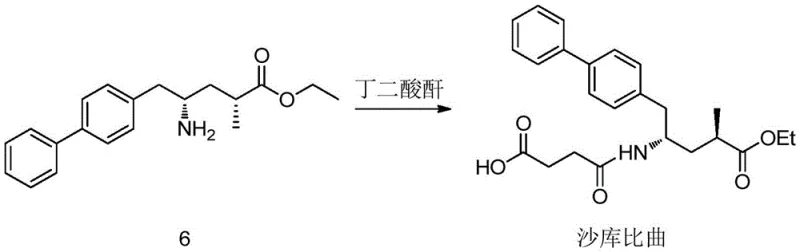
How to Synthesize Shakubiqu Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing high-purity intermediates suitable for pharmaceutical applications. The process begins with the charging of Itaconic anhydride and ethyl acetate into a hydrogenation vessel, followed by the addition of the chiral catalyst system. After the hydrogenation is complete, the crude product is concentrated and purified to yield the chiral anhydride. This intermediate is then subjected to esterification with ethanol and sulfuric acid, followed by the critical enzymatic hydrolysis step where pH and temperature control are vital for selectivity. The resulting mono-acid is activated and coupled with the biphenyl fragment, and the final ketone intermediate is converted to the amine via transamination before the final amidation. For detailed operational parameters and safety considerations, please refer to the standardized guide below.
- Perform chiral reduction of Itaconic anhydride using a Ruthenium catalyst and (S,S)-Me-Duphos ligand under hydrogen pressure to obtain R-methyl succinic anhydride.
- Execute esterification followed by selective enzymatic hydrolysis using lipase to generate the mono-acid chiral building block.
- Activate the carboxyl group and couple with 4-biphenylacetic acid, followed by transamination and final amidation with succinic anhydride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere technical elegance. The primary advantage stems from the utilization of Itaconic anhydride, a fermentation-derived feedstock that is significantly cheaper and more accessible than the specialized chiral building blocks required by conventional methods. This shift in raw material sourcing mitigates the risk of supply disruptions and price volatility associated with niche fine chemicals. Furthermore, the integration of enzymatic steps reduces the dependency on stoichiometric chiral reagents and heavy metal catalysts, which not only lowers material costs but also simplifies the waste treatment process. The elimination of multiple protection and deprotection sequences translates directly into fewer unit operations, reduced solvent consumption, and shorter cycle times, all of which contribute to a leaner and more responsive manufacturing operation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive chiral resolution agents with catalytic systems. By establishing chirality early using a reusable metal-ligand complex and a biocatalyst, the process avoids the 50% theoretical yield loss inherent in classical resolution techniques. Additionally, the use of commodity chemicals like Itaconic anhydride and 4-biphenylacetic acid ensures that the raw material cost base remains low and stable. The simplified workflow reduces labor and utility costs associated with extended reaction times and complex workups, leading to a structurally lower cost of production that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the chemical transformations involved. Hydrogenation and enzymatic hydrolysis are well-understood, scalable unit operations that are less prone to the variability seen in cryogenic or highly sensitive organometallic reactions. The ability to source key starting materials from multiple global suppliers reduces single-source dependency risks. Moreover, the high selectivity of the enzymatic steps minimizes the formation of difficult-to-remove impurities, ensuring consistent quality batch after batch. This consistency is crucial for maintaining regulatory compliance and avoiding costly production delays due to out-of-specification results.
- Scalability and Environmental Compliance: From an environmental perspective, this route aligns with modern sustainability goals by reducing the E-factor (mass of waste per mass of product). The avoidance of chlorinated solvents in certain steps and the use of aqueous buffers for enzymatic reactions demonstrate a commitment to greener chemistry. The process is designed for scalability, with reaction conditions such as temperature and pressure that are easily manageable in standard stainless steel reactors. This ease of scale-up facilitates a smoother transition from pilot plant to commercial production, ensuring that supply can meet market demand without the need for specialized or exotic equipment.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial viability of this synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these aspects helps stakeholders evaluate the potential for integrating this technology into their existing supply chains.
Q: What are the key advantages of using Itaconic Anhydride as a starting material?
A: Itaconic anhydride is a readily available, low-cost commodity chemical derived from fermentation. Using it avoids the need for expensive chiral pool starting materials or complex resolution steps required in older patents like WO2008031567A1.
Q: How is high stereochemical purity achieved in this process?
A: The process utilizes a highly enantioselective hydrogenation step with a Ru/(S,S)-Me-Duphos catalyst system achieving >99% ee, followed by a kinetic resolution via lipase hydrolysis which further ensures optical purity.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method eliminates repeated protection and deprotection steps and uses robust catalytic systems (hydrogenation and enzymatic hydrolysis) that are well-suited for kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Shakubiqu Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value pharmaceutical intermediates like Shakubitril. Our team of expert chemists has extensively analyzed the methodology disclosed in CN113121342B and possesses the technical capability to implement this advanced chiral hydrogenation and enzymatic resolution strategy. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with state-of-the-art hydrogenation reactors and biocatalysis suites, allowing us to execute these sensitive reactions under stringent purity specifications. With our rigorous QC labs, we guarantee that every batch meets the highest standards of optical purity and chemical integrity required for API synthesis.
We invite you to collaborate with us to leverage this cutting-edge technology for your Shakubitril supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this novel route. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project requirements. Let us help you optimize your manufacturing process and secure a competitive advantage in the global cardiovascular drug market.
