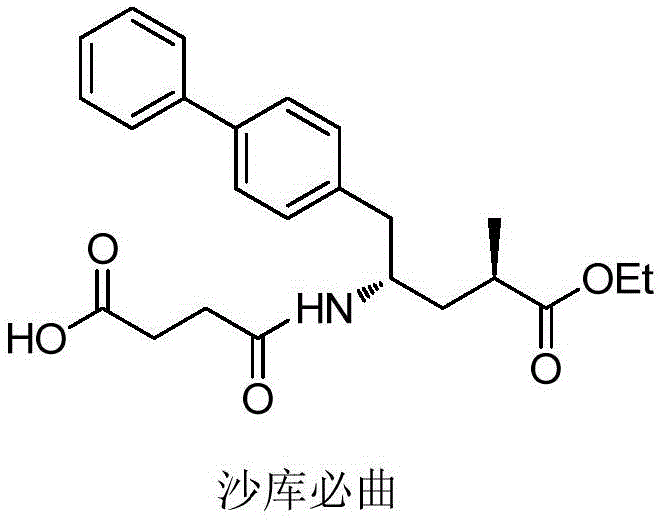
Advanced Synthesis of Sacubitril Intermediates via Enzymatic Catalysis for Commercial Scale-up
The pharmaceutical landscape for cardiovascular treatments has been significantly reshaped by the introduction of Sacubitril, a key component of the blockbuster drug Entresto. As detailed in the recent patent CN113121342A, a novel preparation method for Sacubitril intermediates has emerged, addressing critical bottlenecks in existing manufacturing protocols. This innovation leverages itaconic anhydride as a foundational raw material, subjecting it to a sophisticated sequence of manual reduction, esterification, selective hydrolysis, and carboxyl activation. The culmination of this process involves a coupling reaction with 4-biphenylacetic acid to yield the pivotal intermediate. This technical breakthrough is not merely an academic exercise but a strategic advancement for any entity seeking a reliable pharmaceutical intermediate supplier capable of delivering high-quality precursors for complex cardiovascular therapies.
The structural complexity of Sacubitril, chemically known as 4-(((2S,4R)-1-([1,1'-biphenyl]-4-yl)-5-ethoxy-4-methyl-5-oxopentan-2-yl)amino)-4-oxobutanoic acid, demands precise stereochemical control. The patented route offers a streamlined alternative to traditional syntheses, which often suffer from low atom economy and excessive step counts. By integrating biocatalytic elements with classical organic synthesis, this method ensures that the final product meets the rigorous purity standards required for API manufacturing. For procurement teams evaluating supply chain resilience, understanding the mechanistic underpinnings of this new route is essential for assessing long-term viability and cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sacubitril and its intermediates has been plagued by significant inefficiencies that hinder large-scale production. Prior art, such as the methods disclosed in WO2008031567A1 and CN104556700A, often relies on the use of expensive chiral induction reagents or complex metal-catalyzed reductions that require stringent conditions. These conventional routes frequently necessitate multiple protection and deprotection steps for carboxyl and amino groups, which drastically increases the number of unit operations and generates substantial chemical waste. Furthermore, the reliance on difficult-to-source chiral starting materials creates a fragile supply chain, where any disruption in the availability of specialized reagents can halt production entirely. The cumulative effect of these factors is a manufacturing process that is not only cost-prohibitive but also environmentally burdensome, failing to meet modern green chemistry standards.
The Novel Approach
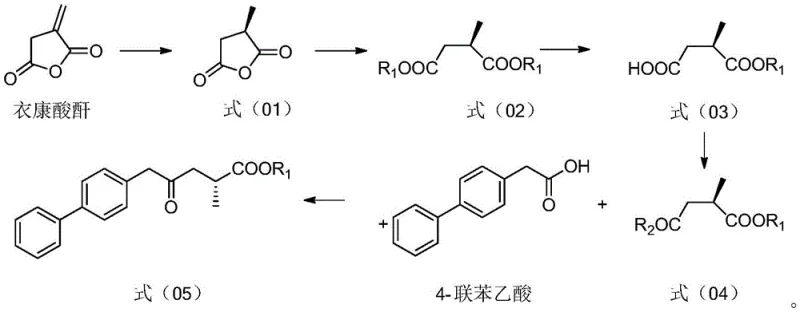
In stark contrast, the methodology presented in patent CN113121342A introduces a paradigm shift by utilizing itaconic anhydride, a commodity chemical, as the starting point. This novel approach bypasses the need for exotic chiral pools by installing chirality early in the synthesis through asymmetric hydrogenation. The process flow is remarkably linear, moving from reduction to esterification and then to a highly selective enzymatic hydrolysis. This strategy effectively eliminates the need for repetitive protection groups, thereby shortening the overall synthetic timeline. The integration of enzymatic steps, specifically lipase-catalyzed hydrolysis and transaminase-mediated amination, introduces a level of selectivity that is difficult to achieve with traditional chemical catalysts alone. This results in a cleaner reaction profile with fewer by-products, directly translating to simplified downstream processing and higher overall yields.
Mechanistic Insights into Asymmetric Hydrogenation and Enzymatic Resolution
The cornerstone of this synthesis is the initial asymmetric hydrogenation of itaconic anhydride, which sets the stereochemical foundation for the entire molecule. This transformation is catalyzed by a transition metal complex, preferably ruthenium, coordinated with a chiral ligand such as (S,S)-Me-Duphos. The interaction between the metal center and the chiral ligand creates a highly specific steric environment that directs the addition of hydrogen to the olefinic bond of the anhydride with exceptional enantioselectivity. Under optimized conditions, typically involving hydrogen pressures around 5 MPa and temperatures near 35°C, this step achieves an enantiomeric excess (ee) value greater than 99%. This high degree of stereocontrol is critical, as any racemic contamination at this stage would propagate through the synthesis, complicating purification and potentially compromising the safety profile of the final API.
Following esterification, the process employs a biocatalytic step that showcases the power of enzyme engineering in fine chemical synthesis. The diester intermediate is subjected to selective hydrolysis using a lipase, such as Lipozyme CALLB, in a phosphate buffer solution. Unlike chemical hydrolysis, which often lacks specificity and can lead to over-hydrolysis or racemization, the lipase selectively cleaves one ester bond while leaving the other intact, generating the mono-acid species required for the subsequent coupling. This enzymatic resolution acts as a second filter for optical purity, ensuring that only the desired enantiomer proceeds to the next stage. The mild aqueous conditions required for this step further underscore the environmental benefits of the process, avoiding the use of harsh acids or bases that are typical in traditional hydrolysis protocols.
How to Synthesize Sacubitril Intermediate Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly during the catalytic and enzymatic stages. The process begins with the charging of itaconic anhydride and a suitable solvent like ethyl acetate into a hydrogenation vessel, followed by the addition of the ruthenium catalyst and chiral ligand under an inert atmosphere. Once the chiral anhydride is formed, it is esterified using ethanol and an acid catalyst, after which the crude diester is subjected to the lipase treatment. The resulting mono-acid is then activated, typically using oxalyl chloride, to form an acid chloride which is immediately coupled with 4-biphenylacetic acid in the presence of a strong base like isopropyl magnesium chloride. The final transformation involves a transaminase-catalyzed amination to install the amine functionality, yielding the key intermediate ready for final amidation with succinic anhydride. Detailed standardized synthesis steps are provided in the guide below.
- Perform asymmetric hydrogenation of itaconic anhydride using a Ruthenium catalyst with (S,S)-Me-Duphos ligand to obtain chiral methyl succinic anhydride.
- Execute selective enzymatic hydrolysis of the diester intermediate using lipase to generate the mono-acid species with high optical purity.
- Couple the activated acid chloride with 4-biphenylacetic acid followed by transaminase-catalyzed amination to yield the final amino-ester intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling strategic advantages that extend beyond mere technical feasibility. The shift towards using itaconic anhydride as a primary feedstock represents a significant move towards cost stability, as this raw material is produced on a massive industrial scale for polymer applications, ensuring consistent availability and pricing. Unlike specialized chiral building blocks that are subject to volatile market dynamics and limited supplier bases, commodity chemicals provide a robust foundation for long-term supply contracts. Furthermore, the simplification of the synthetic route by removing protection and deprotection steps directly correlates to a reduction in raw material consumption and solvent usage, driving down the variable costs associated with each kilogram of produced intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive chiral induction reagents and the reduction in total step count fundamentally alter the cost structure of Sacubitril production. By avoiding the procurement of high-cost specialty ligands and minimizing the number of isolation and purification events, manufacturers can achieve substantial savings in both material and operational expenditures. The use of enzymatic catalysis also reduces the reliance on precious metals, which are not only expensive but also incur additional costs for recovery and disposal to meet environmental regulations. This leaner manufacturing model allows for a more competitive pricing strategy in the global API market without compromising on quality margins.
- Enhanced Supply Chain Reliability: Diversifying the raw material base to include widely available commodity chemicals significantly mitigates supply chain risks. The dependency on single-source suppliers for complex chiral starting materials is a known vulnerability in pharmaceutical manufacturing; this new route effectively decouples production from such bottlenecks. Additionally, the robustness of the enzymatic steps, which operate under mild conditions, reduces the likelihood of batch failures due to thermal runaways or sensitive reagent degradation. This reliability ensures a more predictable production schedule, enabling supply chain planners to maintain optimal inventory levels and meet delivery commitments with greater confidence.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that translate seamlessly from laboratory benchtop to multi-ton reactors. The avoidance of extreme temperatures and pressures simplifies the engineering requirements for production facilities, lowering capital expenditure barriers for scale-up. From an environmental perspective, the reduction in chemical waste and the use of biocatalysts align with increasingly stringent global sustainability mandates. This compliance not only avoids potential regulatory fines but also enhances the corporate social responsibility profile of the manufacturer, a factor that is becoming increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is crucial for R&D teams evaluating technology transfer and for quality assurance personnel establishing control strategies.
Q: What are the primary advantages of the itaconic anhydride route over traditional methods?
A: The primary advantage lies in the use of inexpensive, readily available raw materials like itaconic anhydride compared to costly chiral starting materials. Additionally, the process eliminates complex protection and deprotection steps, significantly simplifying the workflow and reducing waste generation.
Q: How is high optical purity maintained throughout the synthesis?
A: High optical purity is achieved through a dual-enantioselective strategy involving asymmetric hydrogenation with a specialized Ruthenium-(S,S)-Me-Duphos catalyst system and subsequent kinetic resolution via lipase-catalyzed hydrolysis, ensuring ee values exceeding 99%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is specifically designed for industrial scalability. It utilizes robust catalytic systems and mild enzymatic conditions that avoid extreme temperatures or pressures, making the process economically viable and environmentally compliant for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sacubitril Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis route for the cardiovascular therapeutic market. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of Sacubitril intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bottom line. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this superior manufacturing method into your portfolio. Let us be your partner in delivering high-quality, cost-effective solutions for the next generation of heart failure treatments.
