Advanced Phosgene Chlorination Technology for Commercial Scale 4-Chloro-3,5-Dimethylphenol Production
The global demand for high-efficiency broad-spectrum antimicrobial agents has intensified following regulatory restrictions on legacy biocides like triclosan. In this context, patent CN111517922B presents a transformative approach to synthesizing 4-chloro-3,5-dimethylphenol (PCMX), a critical active ingredient in personal care and industrial sterilization. This technology addresses long-standing challenges in regioselectivity and waste management by employing phosgene as a chlorinating reagent synergized with saturated nitrogen-oxygen heterocyclic promoters. For R&D Directors and Supply Chain Heads, this represents a pivotal shift towards cleaner, more efficient manufacturing protocols that align with modern environmental standards while ensuring consistent product quality. The method achieves conversion rates exceeding 97% and para-selectivity above 96%, establishing a new benchmark for reliability in the supply of functional active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 4-chloro-3,5-dimethylphenol has relied heavily on chlorination using chlorine gas or sulfuryl chloride, both of which suffer from significant technical and environmental drawbacks. When chlorine gas is employed, the para-to-ortho substitution ratio (P/O) is notoriously low, often hovering around 1.5, necessitating complex and costly separation processes to isolate the desired para-isomer. Alternatively, while sulfuryl chloride can improve selectivity to a P/O ratio greater than 6, it generates sulfur dioxide as a hazardous by-product, imposing severe burdens on waste treatment infrastructure and increasing operational compliance costs. Furthermore, existing catalytic systems involving organic sulfides and metal chlorides often fail to achieve sufficient para-selectivity, with P/O ratios stagnating around 14.4, leaving substantial room for improvement in yield purity and process sustainability for reliable functional active ingredients supplier networks.
The Novel Approach
The innovative methodology disclosed in the patent data overcomes these historical bottlenecks by introducing a synergistic catalytic system involving phosgene and specific saturated nitrogen-oxygen heterocyclic compounds. This combination fundamentally alters the reaction environment, enabling a dramatic enhancement in para-selectivity without the generation of noxious sulfur-containing gases. By utilizing promoters such as N-methylmorpholine or 4-cyclohexylmorpholine, the process achieves P/O ratios exceeding 40, effectively eliminating the need for difficult ortho-isomer separation. This leap in selectivity translates directly into cost reduction in personal care chemical manufacturing by minimizing raw material loss and simplifying downstream purification. The operational simplicity, conducted preferably at atmospheric pressure and moderate temperatures, further underscores the viability of this route for large-scale commercial adoption.
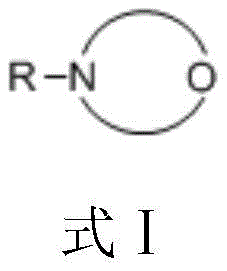
Mechanistic Insights into Phosgene-Morpholine Synergistic Chlorination
The core scientific breakthrough of this technology lies in the intricate molecular interaction between the phenolic substrate and the nitrogen-oxygen heterocyclic promoter. As illustrated by the general structure in the patent data, the promoter's nitrogen or oxygen atoms form specific intermolecular hydrogen bonds with the hydroxyl group of 3,5-dimethylphenol. This interaction serves a dual purpose: firstly, it prevents the carbonyl group of phosgene from engaging in unproductive hydrogen bonding with the phenol, thereby preserving the electrophilic reactivity of the phosgene for chlorination. Secondly, and more critically, the bulky nature of the promoter complex creates significant steric hindrance at the ortho positions of the phenolic ring. This steric shield effectively blocks chlorination at the ortho sites, directing the electrophilic attack exclusively to the para position, which results in the observed high-purity antibacterial agents with minimal isomeric contamination.
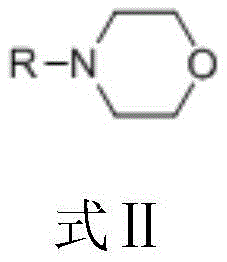
Further analysis of the impurity control mechanism reveals why this system outperforms traditional Lewis acid catalysts. In conventional methods, non-specific activation often leads to poly-chlorination or ortho-substitution, creating a complex impurity profile that requires extensive recrystallization or chromatography to resolve. In contrast, the phosgene-promoter system maintains a highly specific reaction pathway where the conversion of 3,5-dimethylphenol consistently reaches above 97%, with the desired 4-chloro-3,5-dimethylphenol selectivity remaining above 96%. This level of precision reduces the load on quality control laboratories and ensures that the final product meets stringent purity specifications required for pharmaceutical and personal care applications. The ability to tune the promoter structure, such as selecting specific morpholine derivatives, allows for further optimization of the reaction kinetics to suit different commercial scale-up of complex phenolic derivatives requirements.
How to Synthesize 4-Chloro-3,5-Dimethylphenol Efficiently
Implementing this synthesis route requires careful attention to reagent addition rates and temperature control to maximize the benefits of the catalytic system. The process begins by dissolving the starting material, 3,5-dimethylphenol, along with the selected promoter in a chlorinated solvent such as tetrachloroethylene, ensuring a homogeneous reaction medium. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating the high yields reported in the patent examples. Adhering to the specified molar ratios, particularly maintaining a phosgene to phenol ratio between 2:1 and 3:1, is crucial for driving the reaction to completion while minimizing excess reagent waste. Proper management of the exothermic chlorination phase ensures safety and consistency across batches.
- Dissolve 3,5-dimethylphenol and a saturated nitrogen-oxygen heterocyclic promoter (e.g., N-methylmorpholine) in a chlorinated solvent like tetrachloroethylene.
- Slowly introduce phosgene gas and the phenol solution into a reactor maintained at 80-100°C under an inert atmosphere over 0.5 to 2 hours.
- Maintain reaction temperature for 2 to 8 hours, then cool the mixture to crystallize the product, followed by filtration to isolate high-purity 4-chloro-3,5-dimethylphenol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers substantial strategic advantages beyond mere technical performance. The elimination of sulfur dioxide by-products removes a major bottleneck in waste disposal, significantly reducing the environmental compliance burden and associated fees that often inflate the cost of goods sold. Moreover, the high selectivity of the process means that less raw material is wasted on unwanted isomers, leading to a more efficient utilization of 3,5-dimethylphenol feedstock. This efficiency contributes to substantial cost savings and stabilizes pricing structures, making the supply of high-purity disinfectant intermediates more predictable and resilient against market fluctuations. The simplified workup procedure, involving basic crystallization and filtration, also reduces energy consumption and processing time.
- Cost Reduction in Manufacturing: The removal of expensive heavy metal catalysts and the avoidance of complex sulfide waste treatment systems drastically simplify the production infrastructure. By eliminating the need for specialized scrubbing equipment required for sulfur dioxide neutralization, capital expenditure is reduced, and operational overhead is minimized. The high yield and selectivity ensure that the maximum amount of input material is converted into saleable product, optimizing the overall cost structure without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of readily available solvents like tetrachloroethylene and common promoters such as morpholine derivatives ensures that the supply chain is not dependent on exotic or scarce reagents. This accessibility reduces the risk of production delays caused by raw material shortages. Furthermore, the robustness of the reaction conditions, which tolerate standard industrial equipment and atmospheric pressure, facilitates easier technology transfer between manufacturing sites, ensuring reducing lead time for high-purity disinfectant intermediates across global facilities.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, operating efficiently at temperatures between 80°C and 100°C without requiring high-pressure vessels. This makes it ideal for expansion from pilot plants to multi-ton production scales. Additionally, the absence of gaseous sulfide emissions aligns perfectly with increasingly strict global environmental regulations, future-proofing the manufacturing asset against potential regulatory crackdowns and ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chlorination technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this method into their existing production portfolios for optimal resource allocation.
Q: How does this method improve para-selectivity compared to traditional chlorination?
A: By utilizing saturated nitrogen-oxygen heterocyclic promoters, the method forms intermolecular hydrogen bonds with the phenolic hydroxyl group. This steric hindrance blocks ortho-substitution, achieving a P/O ratio exceeding 40, significantly higher than the 1.5 ratio seen with chlorine gas.
Q: What are the environmental advantages of using phosgene over sulfuryl chloride?
A: Traditional methods using sulfuryl chloride generate sulfur dioxide gas as a by-product, complicating waste treatment. This phosgene-based route eliminates gaseous sulfide emissions, simplifying tail gas handling and reducing environmental compliance costs.
Q: Can this process be scaled for industrial production of disinfectant intermediates?
A: Yes, the patent demonstrates operation at atmospheric pressure with common solvents like tetrachloroethylene. The high conversion rates (>97%) and simple crystallization workup indicate strong feasibility for commercial scale-up of complex phenolic derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-3,5-Dimethylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the fine chemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the phosgene-morpholine chlorination method are translated into reliable industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that verify every batch against the highest industry standards. Our capability to manage complex chemistries allows us to offer a stable supply of high-quality intermediates essential for the personal care and pharmaceutical industries.
We invite you to collaborate with us to leverage these technological advancements for your product lines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can enhance your supply chain efficiency and product quality.
