Scalable Synthesis of 2-Amino-9,9-Diphenylfluorene for Next-Gen OLED Displays
The rapid evolution of the organic light-emitting diode (OLED) industry has placed immense pressure on the supply chain for high-performance blue light emitting materials, specifically demanding intermediates that offer superior stability and charge transport properties. Patent CN113582823B introduces a groundbreaking preparation method for 2-amino-9,9-diphenylfluorene, a critical electron-rich conjugated pi-system that serves as a foundational building block for next-generation display technologies. This patent details a robust four-step synthetic route that begins with the bromination of 9-fluorenone and culminates in a sophisticated liquid ammonia amination reaction, effectively addressing the longstanding challenges of high turn-on voltage and efficiency roll-off in traditional OLED devices. By leveraging a combination of electrophilic substitution, Grignard chemistry, and transition metal catalysis, this methodology not only enhances the photophysical properties of the final material but also streamlines the manufacturing workflow to support the massive scale-up required by the global flat panel display market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amino-substituted fluorene derivatives has relied heavily on noble metal catalysis systems, such as the magnetic palladium carbon methods disclosed in earlier patents like CN 106831443A. While these traditional approaches demonstrated the feasibility of nitration and reduction pathways, they suffer from significant economic and operational drawbacks that hinder true mass production capabilities. The primary bottleneck lies in the post-reaction processing, where the separation of fine magnetic catalyst particles from the viscous reaction mixture becomes increasingly difficult and costly as batch sizes grow, often leading to product contamination with trace heavy metals. Furthermore, the reliance on expensive noble metal catalysts that require complex regeneration cycles inflates the raw material costs substantially, making the final electronic chemical less competitive in a price-sensitive market. The multi-step nature of older routes often involves harsh nitration conditions that generate hazardous waste streams, complicating environmental compliance and increasing the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the novel approach outlined in CN113582823B utilizes a streamlined sequence that replaces problematic nitration steps with a highly selective bromination followed by a efficient liquid ammonia amination. This strategy fundamentally shifts the process economics by employing a copper-palladium co-catalytic system that operates effectively in liquid ammonia, a medium that offers superior solubility for ammonia gas and enhances reaction kinetics at elevated pressures. The elimination of magnetic catalyst separation issues allows for a much cleaner workup procedure, significantly reducing the time and resources dedicated to purification while ensuring the stringent purity specifications required for electronic grade materials are met consistently. By integrating a Friedel-Crafts alkylation step that utilizes trifluoromethanesulfonic acid, the process achieves high regioselectivity and yield, minimizing the formation of isomeric impurities that could otherwise degrade the performance of the final OLED device. This holistic redesign of the synthetic pathway demonstrates a clear commitment to process intensification, enabling manufacturers to achieve higher throughput with lower operational expenditures.
Mechanistic Insights into Liquid Ammonia Amination and Friedel-Crafts Alkylation
The core of this technological advancement lies in the precise orchestration of four distinct chemical transformations, beginning with the electrophilic aromatic substitution of 9-fluorenone. In the initial stage, 9-fluorenone reacts with liquid bromine in an acidic medium comprising acetic acid and concentrated sulfuric acid, where the protonation of the carbonyl oxygen activates the ring system for selective bromination at the 2-position. 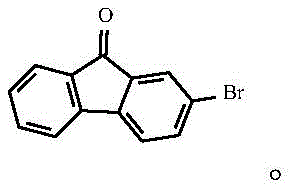 This step is critical as it installs the necessary leaving group for subsequent cross-coupling reactions while maintaining the integrity of the fluorenone core, which is essential for the optical properties of the final molecule. Following isolation, the 2-bromo-9-fluorenone undergoes a nucleophilic addition with a phenylmagnesium bromide Grignard reagent in tetrahydrofuran, where the low temperature control prevents side reactions and ensures the formation of the tertiary alcohol intermediate.
This step is critical as it installs the necessary leaving group for subsequent cross-coupling reactions while maintaining the integrity of the fluorenone core, which is essential for the optical properties of the final molecule. Following isolation, the 2-bromo-9-fluorenone undergoes a nucleophilic addition with a phenylmagnesium bromide Grignard reagent in tetrahydrofuran, where the low temperature control prevents side reactions and ensures the formation of the tertiary alcohol intermediate. 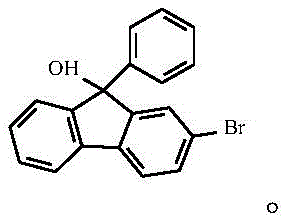 The resulting 2-bromo-9-phenyl-9-hydroxy-fluorene is then subjected to a Friedel-Crafts alkylation with benzene, catalyzed by trifluoromethanesulfonic acid, which facilitates the dehydration and subsequent attachment of the second phenyl ring to form the sterically crowded 9,9-diphenyl scaffold.
The resulting 2-bromo-9-phenyl-9-hydroxy-fluorene is then subjected to a Friedel-Crafts alkylation with benzene, catalyzed by trifluoromethanesulfonic acid, which facilitates the dehydration and subsequent attachment of the second phenyl ring to form the sterically crowded 9,9-diphenyl scaffold. 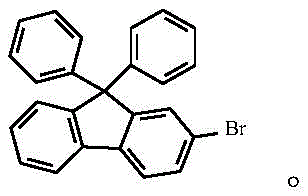 The final and most innovative step involves the conversion of the aryl bromide to the primary amine using cuprous oxide and palladium acetate in liquid ammonia, a reaction that proceeds via a catalytic cycle involving oxidative addition, ligand exchange with ammonia, and reductive elimination to yield the target 2-amino-9,9-diphenylfluorene.
The final and most innovative step involves the conversion of the aryl bromide to the primary amine using cuprous oxide and palladium acetate in liquid ammonia, a reaction that proceeds via a catalytic cycle involving oxidative addition, ligand exchange with ammonia, and reductive elimination to yield the target 2-amino-9,9-diphenylfluorene. 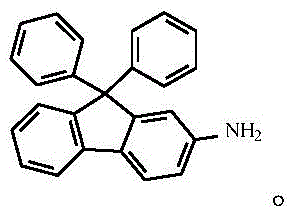 This mechanistic pathway ensures that the electron-donating amino group is introduced with high fidelity, preserving the conjugated system necessary for efficient hole injection in OLED applications.
This mechanistic pathway ensures that the electron-donating amino group is introduced with high fidelity, preserving the conjugated system necessary for efficient hole injection in OLED applications.
Impurity control is meticulously managed throughout this synthesis through specific solvent choices and crystallization techniques that exploit the solubility differences between the target molecule and potential byproducts. For instance, the use of a dichloromethane-petroleum ether mixture during the chromatographic purification of intermediates effectively removes unreacted starting materials and homocoupling products that could arise from the Grignard or amination steps. The final recrystallization from an ethanol-toluene system is particularly effective at excluding trace metal residues from the palladium and copper catalysts, which is a non-negotiable requirement for materials intended for use in thin-film transistor backplanes. By understanding the specific interaction of the fluorene backbone with these solvent systems, process chemists can fine-tune the isolation parameters to maximize recovery yields without compromising the electronic purity of the substance. This level of control over the impurity profile is what distinguishes a laboratory curiosity from a commercially viable electronic chemical, ensuring that every batch meets the rigorous standards demanded by display panel manufacturers.
How to Synthesize 2-Amino-9,9-Diphenylfluorene Efficiently
The practical execution of this synthesis requires careful attention to reaction conditions, particularly the handling of liquid ammonia and the control of exothermic events during the Grignard addition. The patent outlines a standardized protocol that begins with the preparation of the brominated ketone, followed by the sequential buildup of molecular complexity through carbon-carbon and carbon-nitrogen bond forming reactions. Operators must ensure that the pressure reactor used for the final amination step is rated for the specific pressures generated by liquid ammonia at elevated temperatures, typically around 10 atmospheric pressures, to maintain safety and reaction efficiency. Detailed standard operating procedures for each transformation, including precise molar ratios and temperature ramps, are essential for reproducing the high yields reported in the patent examples.
- Brominate 9-fluorenone using liquid bromine in acetic acid and sulfuric acid to form 2-bromo-9-fluorenone.
- React 2-bromo-9-fluorenone with phenylmagnesium bromide in THF to yield 2-bromo-9-phenyl-9-hydroxy-fluorene.
- Perform Friedel-Crafts alkylation with benzene and trifluoromethanesulfonic acid to produce 2-bromo-9,9-diphenylfluorene.
- Conduct amination in liquid ammonia using cuprous oxide and palladium acetate to finalize 2-amino-9,9-diphenylfluorene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple yield improvements, fundamentally altering the cost structure of producing high-value OLED intermediates. The shift away from magnetic palladium carbon catalysts eliminates the need for specialized filtration equipment and the associated downtime required for catalyst recovery, leading to a drastic simplification of the production line infrastructure. This simplification translates directly into lower capital expenditure requirements for new manufacturing facilities and reduced maintenance costs for existing plants, allowing companies to allocate resources more efficiently across their broader portfolio of electronic chemicals. Furthermore, the use of commodity chemicals like benzene and liquid ammonia as primary reagents ensures a stable and resilient supply chain that is less susceptible to the volatility often seen with specialized proprietary catalysts. The ability to source raw materials from multiple global suppliers mitigates the risk of production stoppages due to single-source dependencies, providing a strategic advantage in a market where continuity of supply is paramount for meeting the just-in-time delivery schedules of major display manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive noble metal catalysts with a more economical copper-palladium system that requires significantly lower loading levels to achieve comparable conversion rates. By removing the complex separation steps associated with magnetic catalysts, the process reduces the consumption of solvents and energy required for purification, resulting in substantial operational cost savings per kilogram of finished product. The higher overall yield of the four-step sequence means that less raw material is wasted, further driving down the cost of goods sold and improving the gross margin profile for manufacturers adopting this technology. Additionally, the simplified waste stream, devoid of magnetic particulates, lowers the costs associated with hazardous waste disposal and environmental compliance, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as 9-fluorenone, bromobenzene, and benzene ensures that the supply chain remains robust even during periods of global logistical disruption. Unlike specialized catalysts that may have long lead times or limited production capacity, these foundational reagents can be sourced from a diverse network of chemical suppliers, reducing the risk of bottlenecks that could delay production campaigns. The scalability of the liquid ammonia amination step, which utilizes standard high-pressure reactor technology common in the fine chemical industry, means that production capacity can be ramped up quickly to meet surging demand without the need for bespoke equipment fabrication. This flexibility allows supply chain planners to respond agilely to market fluctuations, ensuring that customers receive their orders on time and fostering long-term partnerships based on reliability and trust.
- Scalability and Environmental Compliance: From an environmental perspective, this method offers a cleaner alternative to traditional nitration-based routes by avoiding the generation of large volumes of acidic wastewater and nitrogen oxide emissions. The use of liquid ammonia as a solvent minimizes the need for volatile organic compounds, aligning the process with increasingly stringent global regulations regarding air quality and worker safety. The ability to recycle solvents like tetrahydrofuran and dichloromethane through established distillation protocols further reduces the environmental footprint of the manufacturing process, supporting corporate sustainability goals. Moreover, the inherent safety of the process, when managed with appropriate engineering controls for pressure and temperature, ensures that large-scale production can be conducted without compromising the well-being of the workforce or the surrounding community, making it an ideal candidate for long-term industrial investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, providing clarity for stakeholders evaluating its potential for integration into their supply chains. These answers are derived directly from the experimental data and process descriptions found within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is critical for assessing the feasibility of scaling this technology from pilot plant to full commercial production.
Q: Why is liquid ammonia used in the final amination step?
A: Liquid ammonia serves as both the solvent and the nitrogen source, facilitating the nucleophilic substitution under high pressure and temperature conditions with improved solubility for the reactants.
Q: How does this method improve upon magnetic palladium carbon catalysis?
A: This method avoids the complex separation processes associated with magnetic catalysts and utilizes a more cost-effective copper-palladium co-catalytic system that is easier to scale industrially.
Q: What are the key purity controls in this synthesis?
A: Purity is maintained through specific recrystallization steps using ethanol-toluene mixtures and rigorous washing protocols during the intermediate isolation phases.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-9,9-Diphenylfluorene Supplier
As the demand for high-efficiency blue OLED materials continues to surge, partnering with a technically proficient manufacturer like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies that deliver both performance and value. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to maintain stringent purity specifications and rigorous QC labs that guarantee batch-to-batch consistency. We understand that the transition from laboratory innovation to industrial reality requires more than just a patent; it demands a partner who can navigate the complexities of process safety, regulatory compliance, and cost optimization to bring your vision to market successfully. Our commitment to excellence means that every gram of 2-amino-9,9-diphenylfluorene we produce is backed by comprehensive analytical data and a dedication to continuous improvement that keeps us at the forefront of the electronic materials sector.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific volume requirements and quality standards. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this optimized process, along with specific COA data and route feasibility assessments that demonstrate our capability to meet your exact needs. Let us help you secure a stable supply of this critical intermediate, empowering your R&D efforts to push the boundaries of what is possible in next-generation display technology.
