Advanced Copper-Catalyzed Etherification for High-Purity Carbazole and Fluorene OLED Intermediates
Advanced Copper-Catalyzed Etherification for High-Purity Carbazole and Fluorene OLED Intermediates
The rapid evolution of the organic light-emitting diode (OLED) industry demands intermediates of exceptional purity and structural precision, particularly for host materials based on carbazole and fluorene scaffolds. Patent CN113105384B introduces a transformative preparation method for these critical organic electroluminescent intermediates, addressing long-standing inefficiencies in etherification processes. This technology leverages a novel LiBr/Cu-(BTC)(SIPr) catalyst system to facilitate the coupling of bromocarbazole or fluorene derivatives with various alcohol or phenol reagents. Unlike traditional methods that rely on costly noble metals or harsh conditions, this innovation achieves high conversion rates under mild alkaline environments, utilizing inexpensive bases such as potassium carbonate or sodium hydroxide. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the synthesis of high-value display materials while mitigating supply chain risks associated with precious metal dependency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl ether intermediates for OLED applications has been plagued by economic and technical inefficiencies. Traditional routes often employ palladium catalysts, such as palladium acetate, which necessitate the use of expensive electron-rich phosphine ligands in stoichiometric excess to achieve acceptable yields. Even when utilizing conventional copper-based systems, the catalyst loading is typically high, often exceeding 5 mol%, and requires equivalent amounts of specialized ligands to stabilize the active species. Furthermore, these legacy processes frequently depend on cesium carbonate as the base, a reagent that is not only costly but also presents handling challenges on a large scale. The cumulative effect of these factors is a process with a high cost of goods sold (COGS) and a complex impurity profile due to residual heavy metals and ligand byproducts, which are detrimental to the performance of final electronic devices.
The Novel Approach
The methodology disclosed in the patent fundamentally restructures this synthetic landscape by introducing a robust copper-metal-organic framework-inspired catalyst. This system operates effectively at loadings as low as 3-5% of the substrate mass, eliminating the need for additional external ligands. By enabling the use of cheap, readily available inorganic bases like sodium hydroxide or potassium carbonate, the process drastically simplifies the reaction matrix. The reaction proceeds smoothly at moderate temperatures ranging from 60°C to 110°C, yielding target ether products with efficiencies consistently exceeding 80%, and in many optimized instances reaching up to 90%. This shift not only lowers the direct material costs but also streamlines the downstream purification process, as the absence of complex ligands and noble metals reduces the burden on metal scavenging and chromatographic separation steps.
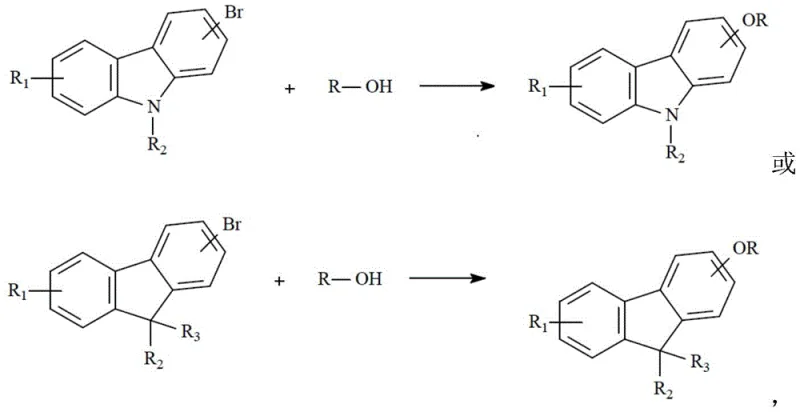
Mechanistic Insights into LiBr/Cu-(BTC)(SIPr) Catalyzed Etherification
The core of this technological advancement lies in the unique structure and stability of the LiBr/Cu-(BTC)(SIPr) catalyst. Prepared via a solvothermal method involving copper nitrate, trimesic acid (BTC), and a bulky N-heterocyclic carbene precursor (SIPr), this catalyst forms a highly active species capable of facilitating the C-O bond formation efficiently. The presence of lithium bromide appears to play a crucial role in modulating the electronic environment of the copper center, enhancing its oxidative addition capability towards the aryl bromide substrate. This mechanistic efficiency allows the reaction to proceed with lower activation energy, thereby permitting the use of milder bases and temperatures. For the R&D team, understanding this mechanism is vital for troubleshooting and further optimization, as the catalyst's stability ensures consistent performance across multiple batches without significant degradation of activity.
From an impurity control perspective, this catalytic system offers distinct advantages over palladium-mediated couplings. Palladium residues are notoriously difficult to remove completely and can act as quenching sites in OLED devices, reducing luminous efficiency and device lifetime. The copper-based system described here, while still requiring removal, generally presents fewer challenges in terms of trace metal specification compliance compared to noble metals. Additionally, the mild reaction conditions minimize thermal degradation of the sensitive carbazole and fluorene cores, preventing the formation of polymeric byproducts or decomposition species that often complicate the purification of electronic-grade chemicals. The result is a cleaner reaction profile that aligns perfectly with the stringent quality requirements of the display industry.
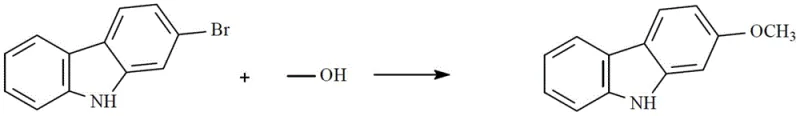
How to Synthesize Carbazole and Fluorene Intermediates Efficiently
Implementing this synthesis route requires precise adherence to the catalyst preparation and reaction protocols outlined in the patent to ensure reproducibility and high yield. The process begins with the in-situ or ex-situ generation of the active catalyst, followed by the introduction of the halogenated substrate and the nucleophilic alcohol or phenol. The reaction is conducted under an inert atmosphere to prevent oxidation of the catalyst or substrates, ensuring maximum turnover. While the general principles are straightforward, the specific ratios of solvent to substrate and the activation temperature of the catalyst are critical parameters that influence the final outcome. For detailed operational procedures, please refer to the standardized synthesis guide below which breaks down the exact experimental conditions.
- Prepare the LiBr/Cu-(BTC)(SIPr) catalyst by refluxing copper nitrate, imidazole ligand, lithium bromide, and trimesic acid in ethanol/DMF, followed by vacuum drying and activation.
- Combine bromocarbazole or fluorene substrate with alkyl alcohol or phenol reagent and a cheap inorganic base such as potassium carbonate or sodium hydroxide in a suitable solvent.
- Add 3-5% of the prepared catalyst, heat the mixture to 60-110°C under nitrogen protection for 6-10 hours, then filter and crystallize to obtain the high-purity ether product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere technical feasibility. The primary advantage is the substantial reduction in raw material costs driven by the substitution of expensive palladium and cesium carbonate with commodity chemicals. This shift insulates the manufacturing process from the volatile pricing of precious metals and specialized reagents, leading to a more predictable and stable cost structure. Furthermore, the simplified work-up procedure, which often involves basic filtration and crystallization rather than complex column chromatography, reduces solvent consumption and waste generation, contributing to both economic and environmental sustainability goals.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts and expensive ligands directly lowers the bill of materials. By utilizing a copper-based system with low loading and cheap inorganic bases, the overall production cost is significantly reduced. This cost efficiency is compounded by the higher yields achieved, which maximize the output per unit of starting material, thereby improving the overall process economics and margin potential for high-volume OLED intermediate manufacturing.
- Enhanced Supply Chain Reliability: Reliance on palladium and specialized phosphine ligands often introduces supply chain bottlenecks due to their limited global availability and geopolitical sourcing risks. In contrast, the reagents required for this novel method—copper salts, trimesic acid, and common alkali bases—are widely available from multiple global suppliers. This diversification of the supply base enhances resilience against disruptions, ensuring continuous production capability and reliable delivery schedules for downstream OLED panel manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of common solvents like toluene or dioxane make this process highly amenable to scale-up from pilot plant to commercial tonnage production. The reduced toxicity profile compared to heavy palladium systems simplifies waste treatment and regulatory compliance. The ability to produce high-purity intermediates with a smaller environmental footprint aligns with the increasing corporate sustainability mandates of major electronics conglomerates, adding value to the supply partnership.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this etherification technology. These insights are derived directly from the patent data to provide clarity on process capabilities and limitations. Understanding these details is essential for evaluating the fit of this technology within your existing manufacturing infrastructure and quality assurance frameworks.
Q: What are the primary advantages of the LiBr/Cu-(BTC)(SIPr) catalyst over traditional palladium systems?
A: The novel copper-based catalyst eliminates the need for expensive noble metals like palladium and complex phosphine ligands. It operates at significantly lower loadings (3-5%) compared to conventional copper systems and allows for the use of inexpensive inorganic bases like potassium carbonate instead of costly cesium carbonate, drastically reducing raw material costs while maintaining yields above 80%.
Q: How does this process improve impurity profiles for OLED applications?
A: By avoiding palladium catalysts, the process inherently removes the risk of trace heavy metal contamination which is critical for electronic grade materials. Furthermore, the catalyst system is heterogeneous or easily separable, and the mild reaction conditions minimize side reactions, resulting in a cleaner crude product that requires less intensive purification to meet stringent electronic specifications.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method is designed for scalability. It utilizes common solvents like toluene or dioxane and operates at moderate temperatures (60-110°C) under standard nitrogen protection. The catalyst is robust, and the work-up involves simple filtration and crystallization, making it highly suitable for commercial scale-up from kilogram to multi-ton production without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole and Fluorene Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of next-generation display technologies. Our technical team has extensively analyzed the pathway described in CN113105384B and possesses the expertise to adapt and optimize this copper-catalyzed etherification for your specific product requirements. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis essential for electronic grade materials.
We invite you to collaborate with us to leverage this cost-effective and robust synthetic route for your OLED material supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can enhance your supply security and reduce your total cost of ownership for these critical electroluminescent intermediates.
