Revolutionizing Tadalafil Production: A High-Yield Two-Step Strategy for Global Pharmaceutical Supply Chains
Revolutionizing Tadalafil Production: A High-Yield Two-Step Strategy for Global Pharmaceutical Supply Chains
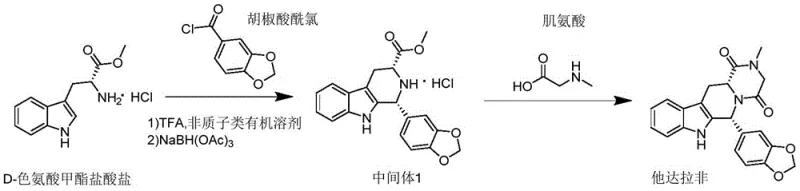
The pharmaceutical landscape for PDE5 inhibitors is constantly evolving, driven by the need for more efficient, safer, and scalable manufacturing processes. Patent CN110684025A introduces a groundbreaking preparation method for tadalafil that fundamentally alters the synthetic paradigm by reducing the entire sequence to merely two distinct steps. This innovation addresses long-standing industry pain points regarding the use of toxic, controlled, and malodorous reagents such as thionyl chloride and Lawson's reagent, which have historically plagued production lines. By utilizing easily obtainable raw materials like piperic acid acyl chloride and D-tryptophan methyl ester hydrochloride, the disclosed method achieves a remarkable yield of 90.54% in the final step, demonstrating exceptional efficiency. For global stakeholders, this represents a significant leap forward in process chemistry, offering a robust pathway for the commercial scale-up of complex heterocycles while adhering to stricter environmental and safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of tadalafil has been fraught with operational hazards and inefficiencies that complicate supply chain management and increase production costs. Prior art methods, such as those disclosed in US2009312548 or the route developed by Wupulhua et al., typically rely on a cascade of dangerous reagents including thionyl chloride, chloroacetyl chloride, and methylamine. These substances are not only highly toxic and flammable but also pose significant risks of explosion and severe environmental pollution, necessitating expensive containment and waste treatment protocols. Furthermore, alternative routes utilizing Lawson's reagent introduce severe olfactory challenges and generate unpleasant byproducts, making them unsuitable for large-scale facilities located near populated areas. The cumulative effect of these multi-step processes often results in a dismal total yield of approximately 25.9%, leading to substantial material waste and inflated raw material consumption.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in CN110684025A streamlines the synthesis into a highly efficient two-step sequence that eliminates the need for hazardous acylating agents and smelly sulfur-based reagents. The novel approach leverages a direct condensation between D-tryptophan methyl ester hydrochloride and piperic acid acyl chloride, facilitated by a mild protonic acid catalyst rather than harsh Lewis acids. This strategic shift allows for the formation of the critical tetrahydro-beta-carboline intermediate in a single pot without the need for isolating unstable imine species. By bypassing the traditional chloroacetylation and subsequent methylation steps, the new route drastically reduces the number of unit operations, thereby minimizing solvent usage and energy consumption. This simplification is pivotal for any reliable pharmaceutical intermediates supplier aiming to enhance throughput while maintaining rigorous safety compliance.
Mechanistic Insights into TFA-Catalyzed Reductive Amination
The core chemical innovation of this patent lies in the sophisticated manipulation of the Friedel-Crafts acylation and Schiff base reaction mechanisms within a unified reaction vessel. Traditionally, Friedel-Crafts reactions require stoichiometric amounts of metal halides like aluminum trichloride, which generate voluminous hydroxide colloids upon quenching, leading to filtration nightmares and product entrapment. The inventors surprisingly discovered that a catalytic amount of trifluoroacetic acid (TFA) is sufficient to drive both the initial acylation and the subsequent condensation to form an imine intermediate. This protonic acid catalyst avoids the formation of gelatinous precipitates, ensuring a homogeneous reaction mixture that is easy to process. Following the imine formation, the addition of sodium triacetoxyborohydride effects a selective reductive amination, stabilizing the structure into Intermediate 1 with high stereochemical integrity. This mechanistic elegance ensures that the reaction proceeds smoothly at mild temperatures (30-45°C), preserving the delicate indole moiety from degradation.
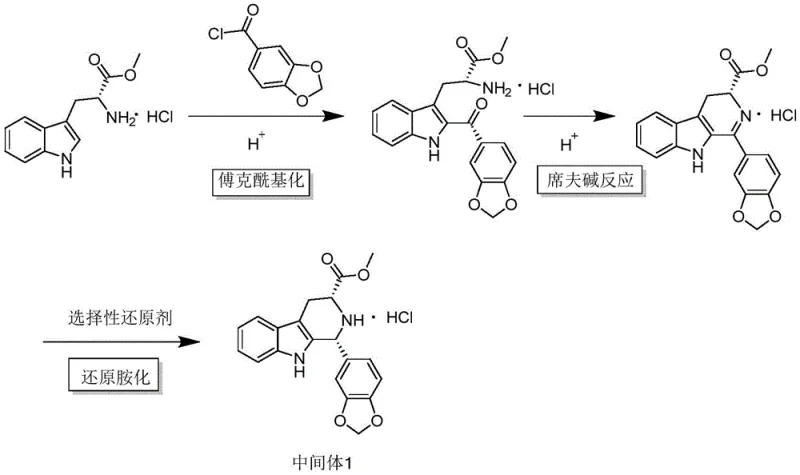
Controlling the impurity profile in such complex heterocyclic syntheses is paramount for meeting pharmacopeial standards, and this method offers inherent advantages in purity control. The use of sodium triacetoxyborohydride as the reducing agent is particularly advantageous because it is selective for imines over ketones or aldehydes, preventing over-reduction or side reactions that could generate difficult-to-remove impurities. Furthermore, the one-pot nature of the first step means that the unstable imine intermediate is never isolated, thereby preventing its decomposition into polymeric byproducts which often occur during work-up procedures in conventional routes. The subsequent cyclization step utilizes sarcosine and magnesium methoxide, which promotes a clean aminolysis reaction. The specific optimization of the mass ratio between the intermediate and the alkaline reagent (1:1.5) ensures complete conversion while minimizing the formation of open-ring byproducts, resulting in a final product with purity levels reaching 100.00% as confirmed by HPLC analysis.
How to Synthesize Tadalafil Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize the yield of the key Intermediate 1 and the final tadalafil product. The process begins with the dissolution of the starting materials in an aprotic solvent, preferably tetrahydrofuran, which has been shown to provide superior solubility and reaction kinetics compared to polar aprotic solvents like DMF or acetonitrile. The addition of trifluoroacetic acid must be carefully metered to maintain a mass ratio of roughly 1:0.05 relative to the tryptophan ester, as deviations can significantly impact the yield of the intermediate. Following the formation of Intermediate 1, the reaction mixture is directly subjected to amidation with sarcosine, activated by DCC and DMAP, before undergoing a critical reflux step. Detailed standard operating procedures and specific stoichiometric ratios for scaling this reaction are provided in the structured guide below.
- Dissolve D-tryptophan methyl ester hydrochloride and piperic acid acyl chloride in tetrahydrofuran, add trifluoroacetic acid catalyst, and react at 30°C before adding sodium triacetoxyborohydride for reductive amination to form Intermediate 1.
- Add sarcosine and triethylamine to the Intermediate 1 solution, activate with DCC and DMAP, then perform reflux cyclization at 80°C using magnesium methoxide as the alkaline reagent.
- Cool the reaction mixture, filter off solids, extract the organic phase with ethyl acetate, and purify the crude product via recrystallization to obtain high-purity tadalafil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple yield improvements. By eliminating the reliance on controlled substances like piperonal and toxic reagents such as thionyl chloride, manufacturers can significantly reduce the regulatory burden and insurance costs associated with handling hazardous materials. The removal of Lawson's reagent, known for its pervasive and difficult-to-contain odor, further mitigates community relations risks and allows for production in a wider range of industrial zones. This streamlined process inherently supports cost reduction in API manufacturing by decreasing the number of isolation and purification steps, which in turn reduces solvent consumption, labor hours, and waste disposal fees. The ability to produce high-purity material in fewer steps also shortens the overall production cycle time, enhancing responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the synthetic route, which removes the need for expensive and hazardous reagents that require specialized storage and handling infrastructure. By replacing multi-step sequences involving chloroacetyl chloride and methylamine with a direct condensation and cyclization, the process eliminates several unit operations that traditionally contribute to high variable costs. The high yield of over 90% in the final step ensures that raw material utilization is maximized, reducing the cost of goods sold (COGS) per kilogram of active pharmaceutical ingredient. Additionally, the avoidance of metal halide catalysts removes the necessity for costly metal scavenging resins or complex aqueous work-ups designed to remove heavy metal residues.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available starting materials such as piperic acid acyl chloride and D-tryptophan methyl ester hydrochloride, which are commodity chemicals with stable global supply networks. Unlike routes dependent on niche or strictly controlled precursors, this method reduces the risk of supply disruptions caused by regulatory changes or vendor shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities without significant yield loss, further ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules and meeting the stringent delivery timelines required by downstream pharmaceutical partners.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with green chemistry principles by minimizing waste generation and avoiding the release of noxious gases. The absence of sulfur-containing reagents and chlorinated acylating agents simplifies effluent treatment, lowering the environmental compliance costs associated with wastewater processing. The process is inherently scalable, as demonstrated by the successful transition from laboratory to pilot scales without the exothermic runaway risks often associated with traditional Friedel-Crafts reactions using Lewis acids. This scalability ensures that the technology can support commercial scale-up of complex heterocycles from hundreds of kilograms to multi-ton annual production capacities without requiring massive capital investment in new reactor types.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel tadalafil synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in patent CN110684025A, providing clarity on the operational advantages over legacy methods. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply source.
Q: How does this new method improve upon traditional tadalafil synthesis routes?
A: Traditional routes often utilize toxic reagents like thionyl chloride and chloroacetyl chloride, or smelly reagents like Lawson's reagent, resulting in complex multi-step processes with low total yields (around 25.9%). This patented method simplifies the process to just two steps with a one-pot boiling mode for the first intermediate, achieving significantly higher yields (over 90%) and eliminating hazardous controlled substances.
Q: What represents the critical innovation in the catalytic system for Intermediate 1?
A: The critical innovation is the replacement of traditional Lewis acid catalysts (like aluminum trichloride) with a catalytic amount of protonic acid, specifically trifluoroacetic acid (TFA). This avoids the formation of hydroxide colloids that block filtration and allows the Friedel-Crafts acylation and Schiff base reaction to occur simultaneously, followed by efficient reductive amination using sodium triacetoxyborohydride.
Q: Why is magnesium methoxide preferred as the alkaline reagent in the cyclization step?
A: Experimental data within the patent indicates that among various alkaline reagents tested, including triethylamine and sodium hydroxide, magnesium methoxide provides the highest yield of tadalafil during the reflux aminolysis reaction. It effectively facilitates the ring closure with sarcosine under optimized temperature conditions of 80°C in tetrahydrofuran.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tadalafil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature profiles required for this two-step synthesis, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch. We are committed to delivering high-purity tadalafil intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical team to explore how this innovative route can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemistry available.
