Optimizing Carvedilol Production: A Technical Analysis of Patent CN102190613B for Commercial Scale-Up
Optimizing Carvedilol Production: A Technical Analysis of Patent CN102190613B for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes for critical cardiovascular medications, and Carvedilol remains a cornerstone therapy for hypertension and heart failure. A significant technological advancement in this domain is detailed in Chinese Patent CN102190613B, which discloses a novel preparation method that fundamentally alters the economic and operational landscape of producing this active pharmaceutical ingredient (API). Unlike traditional pathways that rely on cumbersome protection group strategies or suffer from low yields, this invention introduces a streamlined approach characterized by high operational simplicity and superior yield metrics. For a reliable carvedilol intermediate supplier, understanding the nuances of this patent is essential for optimizing supply chains and reducing manufacturing overheads. The core innovation lies in the specific sequence of coupling 4-epoxypropoxycarbazole with 2-methoxyphenethylamine under controlled conditions that mitigate side reactions.
This report provides a deep-dive technical analysis for R&D directors and procurement leaders, evaluating how this specific methodology addresses historical bottlenecks in cost reduction in API manufacturing. By eliminating the need for catalytic hydrogenation and complex purification sequences found in prior art, the process described in CN102190613B offers a compelling value proposition for large-scale production. The following sections will dissect the mechanistic advantages, compare them against legacy methods, and outline the commercial implications for global supply chains seeking high-purity carvedilol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
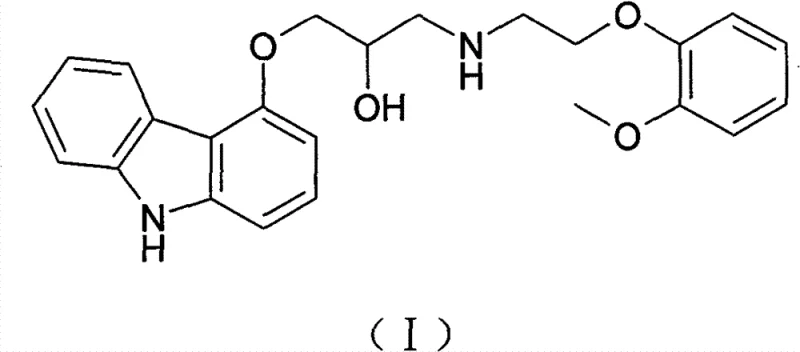
Historical synthesis routes for Carvedilol have been plagued by inefficiencies that drive up costs and complicate quality control. For instance, the method described in US Patent 4503067 utilizes a direct reaction between 4-glycidyloxycarbazole and O-methoxyphenethylamine but suffers from a disappointingly low yield of approximately 39.42%. Furthermore, this older technique generates significant amounts of by-products, necessitating extensive purification efforts that erode profit margins. Another prominent pathway, disclosed in PCT application WO2005113502, attempts to solve selectivity issues by introducing a benzyl protecting group.
While the benzyl protection strategy improves selectivity, it introduces severe logistical and economic drawbacks. This route requires the synthesis of a benzyl-protected intermediate, followed by a catalytic hydrogenation step to remove the protecting group. Catalytic hydrogenation typically requires expensive precious metal catalysts (such as palladium on carbon) and specialized high-pressure equipment, which increases capital expenditure and operational risk. Additionally, the removal of benzyl groups often leaves behind trace metal impurities that are difficult to purge to acceptable pharmaceutical standards, creating a bottleneck for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
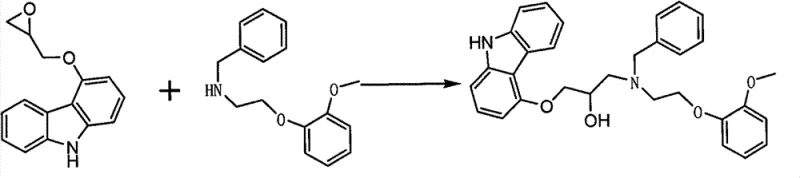
The method disclosed in CN102190613B circumvents these issues by employing a direct coupling strategy that avoids protection groups entirely. The process begins with the synthesis of 4-epoxypropoxycarbazole (Formula III) from 4-hydroxycarbazole and epichlorohydrin. Crucially, the subsequent coupling reaction is engineered to maximize efficiency: the epoxide intermediate is dissolved in tetrahydrofuran and added dropwise into a toluene solution of the amine reactant. This specific order of addition and solvent selection is not arbitrary; it is designed to maintain a kinetic environment where the desired nucleophilic attack is favored over side reactions. By operating without the need for hydrogenation, the process significantly simplifies the workflow, making it highly suitable for industrialized production where equipment versatility and throughput are paramount.
Mechanistic Insights into Epoxide Ring-Opening and Impurity Control
From a mechanistic perspective, the success of this synthesis hinges on the precise control of the nucleophilic ring-opening of the epoxide moiety. The reaction between the carbazole-derived epoxide and the phenethylamine is a classic SN2-type substitution, but it is susceptible to competitive side reactions if concentrations are not managed rigorously. The primary challenge in this chemistry is the potential for the newly formed secondary amine (the product) to act as a nucleophile itself, reacting with another molecule of the epoxide starting material. This leads to the formation of a tertiary amine by-product, identified in the patent as Impurity B (Formula VI).
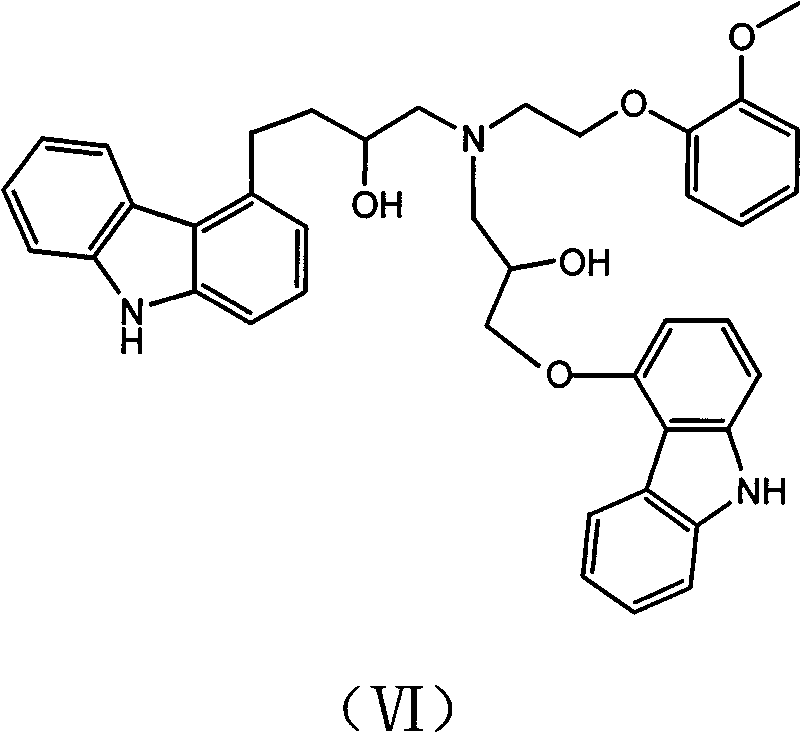
The patent elucidates a sophisticated strategy to suppress this bis-alkylation pathway. Experimental data indicates that if the concentration of the epoxide intermediate is too high relative to the amine, the probability of the product reacting further increases drastically. To counter this, the inventors specify that the 4-epoxypropoxycarbazole solution must be added dropwise to a solution containing a high density of O-methoxyphenethylamine. This ensures that any epoxide molecule entering the reaction vessel immediately encounters an excess of the primary amine reactant, kinetically favoring the formation of the desired secondary amine product. Furthermore, the choice of a methanol-water mixed solvent system (2:1 ratio) for the initial epoxide synthesis is critical; this specific polarity profile minimizes the solubility of the product while keeping impurities and raw materials in solution, facilitating a clean isolation via crystallization before the coupling step even begins.
How to Synthesize Carvedilol Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric and thermal parameters defined in the patent to ensure reproducibility and safety. The process is divided into two distinct stages: the formation of the epoxide intermediate and the final coupling. The first stage involves reacting 4-hydroxycarbazole with epichlorohydrin in the presence of aqueous sodium hydroxide, where temperature control is vital to prevent hydrolysis of the epoxide ring. The second stage demands precise dropwise addition rates to manage the exotherm and maintain the concentration gradient necessary for impurity suppression.
- React 4-hydroxycarbazole with epichlorohydrin in a methanol/water solvent system using aqueous sodium hydroxide to form 4-epoxypropoxycarbazole.
- Dissolve the resulting epoxide intermediate in tetrahydrofuran and add it dropwise to a toluene solution of 2-methoxyphenethylamine.
- Control reaction temperature and concentration carefully to minimize bis-alkylation impurities, followed by crystallization and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to the methodology described in CN102190613B represents a strategic opportunity to optimize the cost structure of Carvedilol production. The elimination of the benzyl protection and subsequent hydrogenation steps removes a significant portion of the variable costs associated with the legacy routes. Without the need for precious metal catalysts, the bill of materials is reduced, and the dependency on volatile commodity markets for metals like palladium is severed. This structural change in the synthesis route translates directly to improved margin stability and cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the removal of the catalytic hydrogenation step. In traditional processes, this step requires expensive catalysts, high-pressure reactors, and rigorous safety protocols, all of which contribute to high overheads. By adopting the direct coupling method, manufacturers eliminate these capital-intensive requirements. Furthermore, the simplified purification process—relying primarily on crystallization rather than complex chromatographic separations—reduces solvent consumption and waste disposal costs, leading to substantial overall savings in the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The raw materials required for this novel route, specifically 4-hydroxycarbazole, epichlorohydrin, and 2-methoxyphenethylamine, are commodity chemicals with well-established global supply chains. Unlike specialized protecting group reagents which may have limited suppliers, these feedstocks are readily available from multiple sources. This diversity in sourcing mitigates the risk of supply disruptions and allows procurement teams to negotiate more favorable terms. Additionally, the robustness of the reaction conditions reduces the likelihood of batch failures, ensuring a more consistent and reliable output of high-purity carvedilol intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process offers distinct advantages. The avoidance of heavy metal catalysts simplifies the regulatory burden regarding residual metal limits in the final API, a critical factor for FDA and EMA compliance. The solvent systems employed (toluene, THF, methanol, water) are standard in the industry and can be efficiently recovered and recycled. The high yield reported in the patent embodiments (over 45% total yield with >99% purity) implies that less raw material is wasted per kilogram of product, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within CN102190613B, providing a factual basis for decision-making.
Q: How does the new method avoid the formation of Impurity B (VI)?
A: The method controls the concentration of 4-epoxypropoxycarbazole by adding it dropwise to a high concentration of the amine reactant. This ensures the epoxide reacts rapidly with the amine rather than reacting with the already formed product, which prevents the formation of the bis-alkylated impurity.
Q: What are the advantages over the benzyl protection route?
A: Unlike the benzyl protection route which requires expensive catalytic hydrogenation and additional protection/deprotection steps, this novel method utilizes a direct coupling strategy. This eliminates the need for precious metal catalysts and reduces the total number of unit operations.
Q: What purity levels can be achieved with this process?
A: According to the patent data, the final product achieves a purity greater than 99% (HPLC) after recrystallization, with a total two-step yield exceeding 45%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carvedilol Intermediate Supplier
The technical elegance of the route described in CN102190613B underscores the importance of partnering with a manufacturer who possesses both the chemical expertise and the infrastructure to execute it flawlessly. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific solvent systems and thermal profiles required for this epoxide coupling, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that consistency is key in the pharmaceutical supply chain, and our processes are validated to deliver the high-quality intermediates necessary for downstream API synthesis.
We invite global partners to engage with us to explore how this optimized synthesis route can benefit their specific supply chain needs. By collaborating with our technical team, you can receive a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential clients to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your transition to this superior manufacturing method is seamless and commercially viable.
