Advanced Purification Technology for High-Purity Azilsartan: Scalable Solutions for Global API Manufacturing
Introduction to Advanced Azilsartan Refining Technologies
The pharmaceutical industry's demand for ultra-high purity angiotensin II receptor antagonists has driven significant innovation in downstream processing technologies. Patent CN103435604A introduces a breakthrough refining method for Azilsartan, a critical cardiovascular active pharmaceutical ingredient (API). This technology addresses the persistent challenges of removing structurally similar impurities that plague conventional recrystallization techniques. By employing a sophisticated two-step solvent crystallization strategy, the process achieves a final purity exceeding 99.85% while maintaining a robust total yield of over 80%. For R&D directors and procurement specialists, this represents a pivotal shift from complex chromatographic separations to scalable, cost-effective crystallization engineering. The method is particularly effective for treating crude products derived from recovered crystallization mother liquors, ensuring that waste streams are converted back into high-value assets without compromising quality standards.
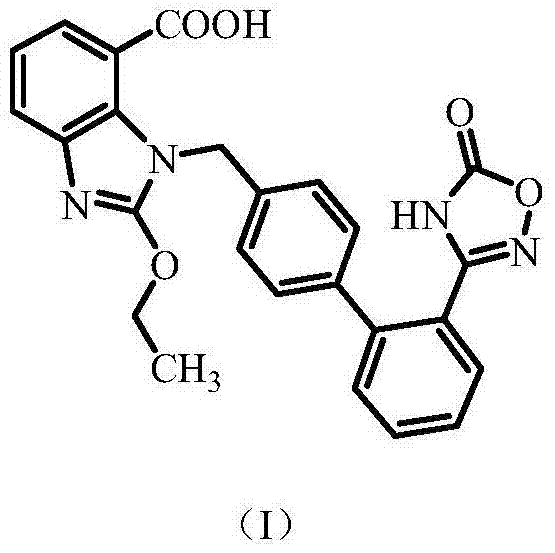
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Azilsartan has been hindered by the formation of stable solvates and difficult-to-remove tautomeric impurities. Prior art methods, such as those described in US 5243054 utilizing ethyl acetate, often result in the formation of unstable solvent compounds that degrade during storage, leading to a significant increase in Impurity B levels. Furthermore, alternative routes involving ethanol recrystallization frequently introduce esterification byproducts, known as Impurity G, due to the reactivity of the carboxylic acid group under acidic or thermal conditions. Another critical challenge arises from specific synthetic pathways, such as the one detailed in CN1067890A, which generates an imidazole tautomer impurity (Impurity I) that is notoriously difficult to separate via standard washing or single-solvent crystallization. These limitations necessitate complex, multi-stage purification protocols that drastically reduce overall yield and increase manufacturing costs.
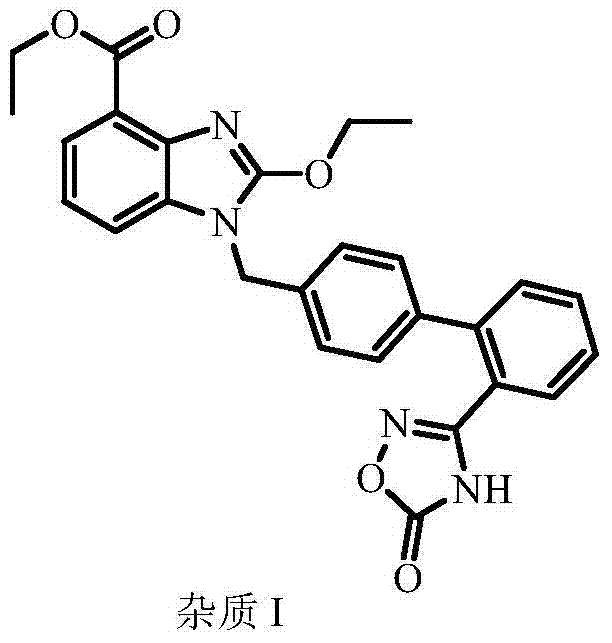
The Novel Approach
The novel approach disclosed in CN103435604A overcomes these thermodynamic and kinetic barriers through a sequential polarity-switching crystallization protocol. Instead of relying on a single solvent system, this method first utilizes a combination of polar aprotic solvents, such as 1,4-dioxane mixed with dimethyl sulfoxide (DMSO) or DMF, to dissolve the crude material at elevated temperatures (75-105°C). This initial step effectively solubilizes the target molecule while precipitating or retaining non-polar contaminants. Subsequently, the intermediate product is subjected to a second crystallization using polar protic solvents like alcohols or aqueous alcohol mixtures. This second stage is crucial for stripping away polar degradation products, specifically targeting the hydrolytically unstable oxadiazole ring derivatives and benzoglyoxaline breakdown products. This dual-solvent strategy ensures that both lipophilic and hydrophilic impurities are systematically eliminated, resulting in a chemically stable, high-purity final product.
Mechanistic Insights into Sequential Solvent Crystallization
The efficacy of this purification process lies in the precise manipulation of solubility parameters and hydrogen bonding interactions between the solvent system and the Azilsartan molecule. In the first stage, the use of polar aprotic solvents creates an environment where the zwitterionic character of the benzoglyoxaline moiety is stabilized, allowing for the selective crystallization of the desired polymorph while leaving highly non-polar organic byproducts in the mother liquor. The specific ratio of solvents, such as 1,4-dioxane to DMSO (approximately 9:1 to 12:1 v/v), is critical for controlling the supersaturation level during the cooling phase from 100°C down to 0-10°C. This controlled nucleation prevents the occlusion of mother liquor within the crystal lattice, which is a common source of persistent impurities. The filtration at this stage removes the bulk of the 'gummy' or oily impurities that typically interfere with downstream processing.
In the second mechanistic stage, the transition to polar protic solvents leverages hydrogen bonding capabilities to disrupt the aggregation of polar impurities such as Impurity B and Impurity H. Impurity B, a degradation product resulting from the rupture of ether bonds in the benzoglyoxaline side group under thermal stress, exhibits higher solubility in alcoholic media compared to the parent Azilsartan molecule at low temperatures. By heating the intermediate product in alcohols (methanol, ethanol, or isopropanol) to 65-80°C and then slowly cooling, the process exploits the differential solubility curves of the API versus its degradation products. This ensures that even trace amounts of open-ring degraded species (Impurity A, Impurity F) and complex derivatives (Impurity H) remain in solution, while the pure Azilsartan crystallizes out with a purity profile where single impurities are reduced from 0.5% to below 0.1%.
How to Synthesize High-Purity Azilsartan Efficiently
Implementing this refining protocol requires strict adherence to temperature gradients and solvent ratios to maximize the rejection of impurities. The process is designed to be operationally simple, relying on standard unit operations found in any GMP-compliant facility, such as heated dissolution tanks, filtration units, and jacketed crystallizers. The key to success lies in the complete dissolution of the crude material in the aprotic phase before cooling, ensuring no seed crystals of impure material remain to propagate defects. Following the initial crystallization and drying, the secondary dissolution in protic solvents must be performed with care to avoid thermal degradation, maintaining temperatures between 65°C and 80°C. Detailed standardized operating procedures regarding stirring speeds (e.g., 100-150 rpm) and cooling rates are essential to reproduce the high yields and purity levels reported in the patent data.
- Dissolve crude Azilsartan in a heated mixture of polar aprotic solvents (e.g., 1,4-dioxane and DMSO), filter, and cool to crystallize the primary refined product.
- Redissolve the primary product in heated polar protic solvents (e.g., alcohol/water mixtures), filter hot to remove insolubles, and cool to induce final crystallization.
- Filter the final crystals and dry under reduced pressure at controlled temperatures (40-70°C) to obtain sterile Azilsartan with purity exceeding 99.85%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology offers substantial strategic advantages beyond mere technical specifications. The elimination of complex chromatographic columns or exotic reagents translates directly into a streamlined supply chain with fewer points of failure. By utilizing commodity solvents like 1,4-dioxane, DMSO, and ethanol, manufacturers can secure raw materials easily without relying on specialized chemical vendors, thereby reducing lead times and mitigating supply risk. Furthermore, the ability to process recovered mother liquor back into the main production stream significantly enhances material efficiency. This closed-loop capability means that yield losses are minimized, and the overall cost of goods sold (COGS) is drastically reduced without the need for expensive recycling infrastructure.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal scavengers or preparative HPLC purification steps often required to remove trace tautomers. By relying solely on crystallization, the operational expenditure is significantly lowered, as energy consumption is limited to standard heating and cooling cycles. The high total yield of over 80% ensures that raw material input is maximized, providing a clear economic advantage over lower-yielding conventional methods that require multiple re-processing steps to achieve similar purity levels.
- Enhanced Supply Chain Reliability: The robustness of this solvent system against variations in crude feedstock quality ensures consistent output. Since the method is effective even on crude products recovered from mother liquors, it provides a safety net for production batches that might otherwise be scrapped due to marginal purity failures. This flexibility allows for a more resilient manufacturing schedule, reducing the risk of stockouts and ensuring a continuous supply of high-purity Azilsartan to downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the use of recoverable organic solvents aligns well with green chemistry initiatives. The solvents used are amenable to standard distillation recovery processes, minimizing hazardous waste generation. The simplicity of the equipment requirements—essentially reactors and filters—means that scaling from pilot plant (100 kgs) to full commercial production (multi-ton) involves minimal engineering changes, facilitating rapid technology transfer and faster time-to-market for generic formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on impurity control and process viability. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this method into existing manufacturing lines.
Q: How does this purification method handle Impurity H and Impurity B?
A: The process utilizes a dual-solvent strategy where polar aprotic solvents initially remove non-polar impurities, followed by polar protic solvents that specifically target and eliminate polar degradation products like Impurity B and complex derivatives like Impurity H, reducing them to below 0.1%.
Q: Can mother liquor from this process be recycled effectively?
A: Yes, the patent explicitly demonstrates that crude product recovered from crystallization mother liquor can be reprocessed using the same method to achieve quality consistent with the primary product, significantly reducing waste and raw material costs.
Q: What is the expected purity and yield of the final Azilsartan product?
A: Industrial implementation of this refining method consistently yields total recovery rates over 80%, with final HPLC purity exceeding 99.85% and single impurity peaks controlled below 0.1%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azilsartan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of purifying molecules like Azilsartan requires more than just standard chemical processing; it demands deep expertise in crystal engineering and impurity profiling. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate solvent switching protocols described in CN103435604A are executed with precision. Our facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch meets stringent purity specifications exceeding 99.85% as required by global pharmacopoeias.
We invite potential partners to engage with our technical procurement team to discuss how this advanced refining method can be tailored to your specific supply chain needs. By leveraging our optimized processes, we can provide a Customized Cost-Saving Analysis that demonstrates the tangible economic benefits of switching to this high-efficiency purification route. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective supply of high-purity Azilsartan for your cardiovascular drug portfolio.
