Advanced Aqueous Synthesis of Azilsartan: Technical Breakthroughs and Commercial Scalability
Advanced Aqueous Synthesis of Azilsartan: Technical Breakthroughs and Commercial Scalability
The global pharmaceutical landscape is continuously evolving to meet the demands of an aging population, particularly in the management of chronic conditions such as hypertension. Patent CN108752328B introduces a transformative approach to the synthesis of Azilsartan, a potent angiotensin II receptor antagonist, by shifting the paradigm from traditional organic solvent-based methods to a greener, aqueous-phase protocol. This technical disclosure is not merely an academic exercise but represents a significant leap forward in process chemistry, offering a streamlined two-step pathway that combines O-acylation with subsequent cyclization and hydrolysis. For R&D directors and process engineers, the implications are profound, as the method demonstrates high atom economy and operational simplicity while maintaining rigorous quality standards. The transition to water as a primary solvent addresses critical environmental and safety concerns inherent in legacy manufacturing processes, positioning this technology as a cornerstone for sustainable API production. By leveraging this patented methodology, manufacturers can achieve substantial improvements in yield and purity, directly impacting the bottom line and supply chain reliability for this high-value cardiovascular medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Azilsartan and its precursors has relied heavily on complex multi-step sequences that utilize volatile organic compounds (VOCs) and expensive reagents, creating significant bottlenecks in production efficiency. Prior art, such as that described in US Patent 5243054, often necessitates the use of solvents like 2,6-lutidine or dichloromethane, which pose severe environmental hazards and require costly waste treatment infrastructure. These conventional routes frequently suffer from low atom economy, where a significant portion of the starting materials ends up as waste rather than incorporated into the final product, driving up the cost of goods sold (COGS). Furthermore, the reliance on harsh reaction conditions and difficult-to-remove impurities often necessitates additional purification steps, such as column chromatography, which are impractical for large-scale commercial manufacturing. The cumulative effect of these inefficiencies is a supply chain that is vulnerable to regulatory scrutiny, raw material price volatility, and extended lead times, ultimately compromising the ability to meet global market demand for essential hypertension medications.
The Novel Approach
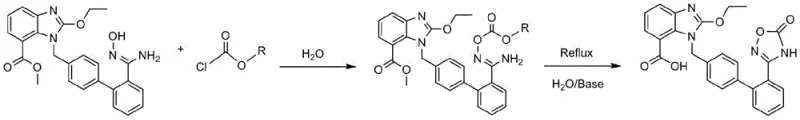
In stark contrast to these legacy methods, the novel approach detailed in the patent data utilizes a streamlined two-step process that operates primarily in an aqueous environment, drastically simplifying the operational workflow. The first step involves the formation of an O-acylated intermediate at a mild temperature of 25°C, eliminating the need for energy-intensive heating or cooling systems during the initial activation phase. The second step cleverly combines cyclization and hydrolysis into a single operation under reflux conditions at 100°C, using common inorganic bases such as sodium hydroxide or potassium carbonate. This consolidation of reaction steps not only reduces the total processing time but also minimizes the handling of intermediates, thereby reducing the risk of contamination and material loss. The use of water as a solvent fundamentally alters the economic model of production, replacing expensive organic solvents with a cheap, non-toxic, and non-flammable alternative. This shift enables a more robust and scalable manufacturing process that aligns perfectly with modern green chemistry principles and regulatory expectations for pharmaceutical intermediates.
Mechanistic Insights into Aqueous O-Acylation and Cyclization
The core of this technological advancement lies in the precise control of reaction kinetics within an aqueous medium, a feat that challenges traditional solubility assumptions in organic synthesis. The mechanism begins with the nucleophilic attack of the hydroxycarbamimidoyl group on the carbonylation reagent, such as phenyl chloroformate, to form the O-acylated intermediate. This step is critical as it activates the molecule for the subsequent ring closure, and the patent data indicates that maintaining a molar ratio of 1:2.5 between the substrate and the carbonylating agent optimizes the yield to approximately 93%. The use of water here is particularly ingenious; it facilitates the precipitation of the intermediate, allowing for simple filtration and washing without the need for complex extraction procedures. This physical separation method is far more scalable than liquid-liquid extraction, which often leads to emulsion issues and product loss at the ton-scale. The subsequent alkaline treatment triggers an intramolecular cyclization where the activated carbamate moiety reacts with the adjacent benzimidazole nitrogen, followed immediately by hydrolysis of the ester group to reveal the free carboxylic acid essential for biological activity.
Impurity control is inherently built into this mechanistic pathway, as the aqueous environment tends to suppress side reactions that are common in organic solvents, such as over-acylation or polymerization. The patent specifies that adjusting the pH of the filtrate to 3-4 using dilute hydrochloric acid after the reflux step ensures the precise precipitation of the final Azilsartan product while keeping soluble impurities in the aqueous phase. This pH-controlled crystallization is a powerful tool for purification, achieving a final purity of 99.5% as demonstrated in the experimental examples. The ability to reach such high purity levels through simple recrystallization from solvents like ethanol or acetonitrile, rather than through chromatography, is a major advantage for commercial production. It ensures that the impurity profile remains consistent and manageable, a key requirement for regulatory filings and quality assurance in the pharmaceutical industry. This mechanistic robustness provides R&D teams with a reliable platform for further process optimization and scale-up.
How to Synthesize Azilsartan Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and reaction conditions outlined in the patent to ensure maximum efficiency and reproducibility. The process begins with the preparation of the O-acylated intermediate, where precise temperature control at 25°C is maintained to prevent degradation of the sensitive hydroxycarbamimidoyl functionality. Following the isolation of this intermediate, the second step involves a reflux operation that must be monitored to ensure complete conversion without excessive thermal stress on the product. The detailed standardized synthesis steps provided below offer a comprehensive guide for process chemists to replicate these results in a pilot or production setting, ensuring that the theoretical benefits of the aqueous method are realized in practice. Adhering to these protocols allows manufacturers to leverage the full potential of this green chemistry approach.
- Mix 1-[(2'-(hydroxycarbamimidoyl)[1,1'-biphenyl]-4-yl)methyl]-2-ethoxy-1H-benzo[d]imidazole-7-carboxylate with a carbonylation reagent in water at 25°C to form the O-acylated intermediate.
- React the O-acylated intermediate with an alkaline reagent in water under reflux at 100°C to induce cyclization and hydrolysis.
- Adjust the pH of the filtrate to 3-4 using dilute HCl, filter the solid, and recrystallize to obtain high-purity Azilsartan.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this aqueous synthesis method offers compelling advantages that extend far beyond simple technical metrics. The elimination of expensive organic solvents and the reduction in processing steps translate directly into a significantly reduced cost of manufacturing, allowing for more competitive pricing strategies in the global API market. The reliance on water and common inorganic bases mitigates the risk associated with the supply volatility of specialized organic reagents, ensuring a more stable and predictable production schedule. Furthermore, the simplified waste profile, characterized primarily by aqueous streams rather than hazardous organic waste, drastically lowers the environmental compliance costs and logistical burdens associated with waste disposal. These factors combine to create a supply chain that is not only more cost-effective but also more resilient to external shocks and regulatory changes.
- Cost Reduction in Manufacturing: The shift to water as a solvent eliminates the substantial recurring expense of purchasing, recovering, and disposing of volatile organic compounds, leading to substantial cost savings in raw material procurement. Additionally, the consolidation of the cyclization and hydrolysis steps reduces energy consumption and labor hours, further driving down the operational expenditure per kilogram of product. By removing the need for complex purification techniques like chromatography, the process also reduces the consumption of silica gel and elution solvents, which are significant cost drivers in traditional API synthesis. These cumulative efficiencies result in a leaner manufacturing process that maximizes margin potential without compromising on product quality.
- Enhanced Supply Chain Reliability: The use of readily available and commodity-grade reagents such as sodium hydroxide and hydrochloric acid ensures that production is not held hostage by the supply constraints of exotic catalysts or specialized solvents. This accessibility enhances the reliability of the supply chain, allowing for continuous production runs and faster response times to market demand fluctuations. The robustness of the aqueous process also reduces the likelihood of batch failures due to solvent quality issues or moisture sensitivity, which are common pain points in organic synthesis. Consequently, procurement managers can secure more consistent delivery schedules and reduce the need for safety stock, optimizing working capital.
- Scalability and Environmental Compliance: The inherent safety of using water as a primary solvent simplifies the scale-up process, as it removes the fire and explosion hazards associated with large volumes of flammable organic liquids. This safety profile facilitates easier regulatory approval for new manufacturing sites and reduces the insurance and infrastructure costs associated with hazardous material handling. Moreover, the reduced environmental footprint aligns with the increasing corporate sustainability goals of major pharmaceutical buyers, making this supply source more attractive for long-term partnerships. The ability to scale from laboratory to commercial production with minimal process modification ensures a smooth transition and rapid time-to-market for new generic or branded formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous synthesis technology for Azilsartan production. These insights are derived directly from the patent data and are intended to clarify the operational benefits and feasibility of adopting this method for large-scale manufacturing. Understanding these details is crucial for stakeholders evaluating the potential integration of this process into their existing supply chains.
Q: How does the aqueous synthesis method improve atom economy compared to traditional organic solvent methods?
A: The patented method utilizes water as the primary solvent for both O-acylation and cyclization steps, eliminating the need for large volumes of expensive and hazardous organic solvents like DMP or dichloromethane, thereby significantly improving atom economy and reducing waste disposal costs.
Q: What are the critical purity specifications achieved in the final Azilsartan product using this route?
A: Experimental data from the patent indicates that the final recrystallized product achieves a purity of 99.5%, meeting stringent pharmaceutical standards required for antihypertensive API manufacturing without requiring complex chromatographic purification.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process operates under mild conditions (25°C for step 1, 100°C reflux for step 2) and uses readily available inorganic bases and water, making it highly scalable, safe, and economically viable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azilsartan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the aqueous synthesis route for Azilsartan and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity Azilsartan that meets stringent purity specifications through our rigorous QC labs, ensuring that every batch conforms to the highest international standards. Our infrastructure is designed to support the green chemistry principles outlined in the patent, allowing us to offer a sustainable and cost-effective supply solution for this vital antihypertensive agent.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of our manufacturing capabilities. Let us help you optimize your supply chain with a reliable partner dedicated to quality, innovation, and long-term value creation in the pharmaceutical intermediates sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
