Scalable Synthesis of High-Purity Proton Pump Inhibitors via Green Resin Catalysis
Scalable Synthesis of High-Purity Proton Pump Inhibitors via Green Resin Catalysis
The pharmaceutical industry constantly seeks robust, environmentally benign, and cost-effective pathways for synthesizing critical Active Pharmaceutical Ingredients (APIs) and their intermediates. A significant technological advancement in this domain is detailed in Chinese Patent CN103408532A, which discloses a novel preparation method for proton pump inhibitors (PPIs). This patent introduces a heterogeneous catalytic system that replaces traditional stoichiometric oxidants and homogeneous metal catalysts with an ion exchange resin and hydrogen peroxide. This shift represents a paradigm change in the manufacturing of benzimidazole sulfoxides, such as omeprazole, lansoprazole, and pantoprazole, offering a route that is not only chemically efficient but also aligns with modern green chemistry principles. For R&D directors and supply chain managers, understanding this technology is crucial for optimizing production lines and securing a reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of proton pump inhibitors involves the selective oxidation of the corresponding sulfide (thioether) precursors to the sulfoxide form. Conventional methodologies have relied heavily on the use of peracids, such as m-chloroperoxybenzoic acid (m-CPBA), or magnesium monoperoxyphthalate (MMPP). While effective, these reagents present significant drawbacks including high cost, potential explosiveness, and the generation of substantial organic waste that complicates downstream processing. Furthermore, alternative methods employing transition metal catalysts, such as vanadium pentoxide, sodium metavanadate, or zirconium complexes, introduce the risk of heavy metal contamination. Removing trace metals from the final API to meet regulatory limits often requires additional, costly purification steps like chromatography or specialized scavenging, which negatively impacts overall yield and extends production lead times. These legacy processes struggle to balance high selectivity with economic and environmental sustainability.
The Novel Approach
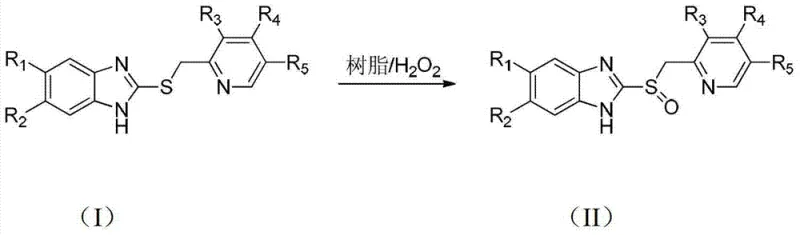
The methodology described in patent CN103408532A offers a transformative solution by utilizing an ion exchange resin as a heterogeneous catalyst in conjunction with hydrogen peroxide ($H_2O_2$) as the terminal oxidant. This approach fundamentally alters the reaction landscape by enabling the oxidation to proceed under mild conditions, typically between 10°C and 30°C, in common organic solvents like methanol or dichloromethane. The use of hydrogen peroxide is particularly advantageous as it is inexpensive, commercially available in high concentrations, and produces water as the only byproduct, thereby eliminating the toxic waste streams associated with peracids. Moreover, the heterogeneous nature of the resin catalyst allows for its facile removal from the reaction mixture via simple filtration, streamlining the workup process and facilitating catalyst recovery and reuse, which is a critical factor for long-term operational efficiency.
Mechanistic Insights into Resin-Catalyzed Selective Oxidation
The core chemical transformation involves the selective oxidation of the sulfur atom in the benzimidazole thioether structure to a sulfoxide, without over-oxidizing to the corresponding sulfone. In this system, the ion exchange resin, specifically strong acid cation exchange resins like the sodium-type D001 mentioned in the examples, acts as a solid support that likely modulates the local chemical environment around the active sites. While the bulk reaction medium is maintained at a basic pH of 9 to 10 using sodium hydroxide to facilitate the formation of the reactive perhydroxyl anion ($HOO^-$), the resin surface may provide a unique microenvironment that stabilizes the transition state. This stabilization is crucial for achieving high chemoselectivity, ensuring that the reaction stops at the sulfoxide stage rather than proceeding to the thermodynamically more stable sulfone. The precise interaction between the resin functional groups and the substrate prevents the uncontrolled radical pathways often seen in homogeneous metal catalysis.
From an impurity control perspective, this mechanism offers distinct advantages for pharmaceutical manufacturing. Traditional metal-catalyzed routes often suffer from batch-to-batch variability due to catalyst deactivation or aggregation, leading to inconsistent impurity profiles. In contrast, the solid resin catalyst described in the patent demonstrates remarkable stability. The patent data indicates that the resin can be reused for multiple cycles—up to 10 times in specific embodiments—while maintaining consistent catalytic activity and yield. This reproducibility is vital for maintaining a tight impurity spectrum, ensuring that the final product consistently meets the stringent purity specifications required for global regulatory compliance. The absence of heavy metals also simplifies the analytical burden, as there is no need for rigorous ICP-MS testing for residual vanadium or zirconium in every batch.
How to Synthesize Proton Pump Inhibitor Intermediates Efficiently
The synthetic protocol outlined in the patent provides a clear, step-by-step guide for implementing this technology in a pilot or production setting. The process begins with the suspension of the sulfide precursor and the resin catalyst in a suitable organic solvent. Critical process parameters include strict temperature control (10-30°C) and pH maintenance (9-10) during the slow addition of hydrogen peroxide. This controlled addition is key to managing the exotherm and preventing runaway oxidation. Following the reaction period of 18 to 30 hours, the mixture is filtered to recover the catalyst, and the filtrate undergoes a straightforward quenching and extraction sequence involving sodium thiosulfate and pH adjustments to isolate the pure sulfoxide product. For detailed standard operating procedures and specific parameter optimization, please refer to the technical guide below.
- Mix the proton pump precursor sulfide compound with an ion exchange resin catalyst (e.g., Sodium-type D001) in an organic solvent such as methanol or dichloromethane.
- Maintain the reaction temperature between 10°C and 30°C, adjust the pH to 9-10 using sodium hydroxide, and slowly add hydrogen peroxide dropwise over 18 to 30 hours.
- Filter to remove the resin catalyst, treat the filtrate with sodium thiosulfate, adjust pH to alkaline conditions, and extract the final high-purity sulfoxide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resin-catalyzed oxidation technology translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing expensive, hazardous peracids and scarce transition metal catalysts with commodity chemicals like hydrogen peroxide and reusable ion exchange resins, manufacturers can significantly reduce their exposure to volatile raw material markets. This shift not only lowers the direct cost of goods sold (COGS) but also mitigates the risk of supply disruptions caused by the limited availability of specialized reagents. The ability to recycle the catalyst further amplifies these savings, turning a consumable cost center into a durable asset that depreciates over many production cycles.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of costly stoichiometric oxidants and the reduction of waste disposal fees. Hydrogen peroxide is one of the most cost-effective oxidants available globally, and its use avoids the generation of chlorinated organic waste associated with m-CPBA. Additionally, the simple filtration step required to remove the resin eliminates the need for complex aqueous workups or column chromatography often needed to remove metal residues. This streamlined downstream processing reduces solvent consumption, energy usage for distillation, and labor hours, collectively driving down the manufacturing cost per kilogram of the final API intermediate without compromising quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the reaction conditions. The mild temperature range (10-30°C) means that the process does not require specialized heating or cryogenic cooling infrastructure, making it adaptable to a wider range of manufacturing facilities. Furthermore, the reusability of the resin catalyst ensures that production capacity is not bottlenecked by the lead time for catalyst procurement. Since the resin can be regenerated and reused multiple times while maintaining activity, facilities can maintain continuous production schedules with minimal interruption for catalyst replenishment, ensuring a steady flow of high-purity intermediates to downstream formulation units.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces safety and environmental challenges, but this method is inherently designed for scale. The use of hydrogen peroxide generates water as a byproduct, significantly reducing the E-factor (mass of waste per mass of product) of the process. This aligns perfectly with increasingly stringent global environmental regulations regarding solvent discharge and heavy metal pollution. The absence of toxic metal catalysts simplifies the environmental permitting process for new manufacturing lines and reduces the liability associated with hazardous waste handling. Consequently, this technology facilitates faster scale-up from pilot plant to commercial tonnage, allowing companies to respond rapidly to market demand for generic or branded proton pump inhibitors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resin-catalyzed oxidation technology. These insights are derived directly from the experimental data and claims presented in patent CN103408532A, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this green chemistry approach into their existing portfolios.
Q: What are the advantages of using ion exchange resin over metal catalysts for PPI synthesis?
A: Unlike traditional vanadium or zirconium catalysts which leave toxic heavy metal residues requiring complex removal steps, ion exchange resin is a heterogeneous catalyst that can be simply removed by filtration. Furthermore, the resin can be reused multiple times while maintaining catalytic activity, significantly reducing material costs and environmental waste.
Q: How does this method ensure high chemical purity for pharmaceutical intermediates?
A: The method utilizes hydrogen peroxide as a clean oxygen source under mild conditions (10-30°C), which minimizes side reactions and over-oxidation to sulfones. The patent data indicates that this selective oxidation process consistently achieves chemical purity levels reaching 99%, meeting stringent pharmaceutical standards without extensive purification.
Q: Is this oxidation process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its mild reaction conditions and simple workup procedure. The elimination of expensive peracids and the ability to recycle the solid catalyst make it economically viable for industrial manufacturing, addressing both cost-efficiency and supply chain stability for bulk API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Proton Pump Inhibitor Supplier
The technological potential of resin-catalyzed oxidation for proton pump inhibitors represents a significant opportunity for optimizing pharmaceutical supply chains. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against international pharmacopeia standards. We understand that consistency is key in the pharma industry, and our advanced manufacturing capabilities allow us to deliver high-purity intermediates with reliable impurity profiles.
We invite you to collaborate with us to leverage these advanced synthetic routes for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green oxidation method can improve your margins. Please contact our technical procurement team today to request specific COA data for our proton pump inhibitor intermediates and to discuss route feasibility assessments for your upcoming campaigns.
