Advanced Nickel-Catalyzed Synthesis of Difluoroalkyl Arenes for Commercial Scale-Up
The chemical industry is constantly seeking more efficient pathways to incorporate fluorine motifs into complex organic scaffolds, particularly for life science applications. Patent CN105669365B introduces a transformative approach to synthesizing aryl or heteroaryl compounds containing difluoroalkyl substitutions. This technology leverages a nickel-catalyzed Suzuki-type cross-coupling reaction, bridging the gap between readily available aryl boronic acids and functionalized difluoroalkyl halides. The significance of this invention lies in its ability to access structural motifs that serve as critical bioisosteres for carbonyl groups, thereby enhancing metabolic stability and binding affinity in drug candidates. By utilizing a robust catalytic system involving nickel salts, nitrogen-containing ligands, and specific additives, the process achieves high conversion rates under relatively mild thermal conditions. This represents a substantial leap forward from classical fluorination techniques, offering a reliable difluoroalkyl intermediate supplier pathway that is both scalable and economically viable for industrial applications.
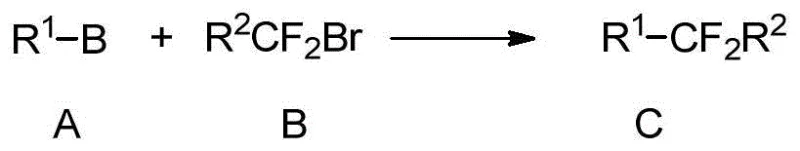
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of difluoromethylene groups into aromatic systems has been fraught with significant synthetic challenges and safety hazards. Traditional methodologies often rely on deoxofluorination of carbonyl or aldehyde precursors using highly reactive and toxic reagents such as DAST (diethylaminosulfur trifluoride) or HF-Py complexes. These reagents are not only dangerous to handle on a large scale due to their potential for generating hydrogen fluoride but also exhibit poor functional group tolerance, often leading to decomposition of sensitive substrates. Furthermore, existing transition metal-catalyzed methods have frequently been restricted to activated difluoroalkyl halides, failing to react efficiently with non-activated alkyl chains where the carbon-bromine bond is inert. This limitation severely restricts the chemical space accessible to medicinal chemists, forcing reliance on multi-step sequences that drive up production costs and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in the patent overcomes these barriers through a cleverly designed nickel-catalyzed cross-coupling protocol. By employing specific ligand systems, such as substituted bipyridines or phenanthrolines, alongside pyridine-based additives like DMAP, the catalyst effectively activates the challenging C-Br bond of non-activated difluoroalkyl bromides. This novel approach allows for the direct coupling of a wide array of aryl and heteroaryl boronic acids with diverse difluoroalkyl fragments. The reaction proceeds smoothly in ether solvents like triglyme at moderate temperatures, typically around 80°C, eliminating the need for cryogenic conditions or excessive heating. This shift in paradigm enables cost reduction in pharmaceutical manufacturing by simplifying the synthetic route, reducing the number of purification steps, and utilizing cheaper base metals instead of precious palladium catalysts, thus providing a versatile platform for generating complex fluorinated building blocks.
Mechanistic Insights into Nickel-Catalyzed Cross-Coupling
The success of this transformation hinges on the unique electronic properties of the nickel catalyst and the synergistic effect of the ligand-additive combination. Mechanistically, the reaction is believed to proceed through a radical-mediated pathway typical of nickel-catalyzed alkyl cross-couplings. The nickel center undergoes oxidative addition into the carbon-bromine bond of the difluoroalkyl halide, potentially generating an alkyl radical intermediate that is stabilized by the adjacent fluorine atoms. The presence of the nitrogen-containing ligand is crucial for maintaining the catalyst in the active oxidation state and preventing premature decomposition or aggregation into inactive nickel black. Additionally, the inclusion of additives such as DMAP appears to facilitate the transmetallation step with the boronic acid species, ensuring rapid turnover of the catalytic cycle. This mechanistic understanding allows for fine-tuning of the reaction parameters to accommodate sterically hindered substrates or electronically diverse aryl rings, ensuring consistent quality and yield across different batches.
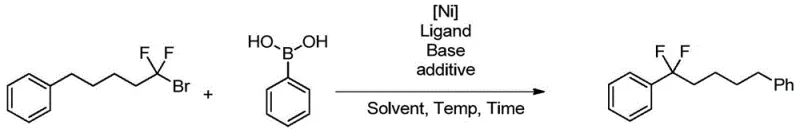
Impurity control is another critical aspect addressed by this catalytic system. In conventional fluorination, side reactions such as elimination to form olefins or hydrolysis of the fluorinating reagent often generate difficult-to-remove impurities. In contrast, the nickel-catalyzed coupling described here demonstrates high chemoselectivity, tolerating functional groups like esters, ketones, nitriles, and even free hydroxyl groups when protected. The mild basic conditions employed, typically using potassium carbonate, minimize the risk of base-sensitive side reactions such as epimerization or elimination. Furthermore, the use of well-defined nickel salts ensures that heavy metal residues can be effectively managed through standard scavenging techniques, meeting the stringent purity specifications required for API production. This high level of control over the reaction profile translates directly into reduced downstream processing costs and higher overall process efficiency.
How to Synthesize Difluoroalkyl Substituted Aryl Compounds Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the catalyst and ligand to ensure maximum turnover. The standard protocol involves charging a reaction vessel with the aryl boronic acid, the difluoroalkyl bromide, and the nickel catalyst precursor in a dry ether solvent. It is essential to maintain an inert atmosphere to prevent oxidation of the low-valent nickel species. The addition of the base and the pyridine additive should be timed to coincide with the heating phase to initiate the catalytic cycle effectively. While the general procedure is robust, specific substrates may require optimization of the ligand structure or solvent choice to achieve optimal yields. For detailed operational parameters and safety guidelines regarding the handling of fluorinated alkyl halides, operators should refer to the specific experimental sections of the patent documentation.
- Combine aryl boronic acid, nickel catalyst (e.g., NiCl2·DME), bipyridine ligand, and base in a glyme solvent.
- Add the functionalized difluoroalkyl bromide substrate and an additive such as DMAP to the reaction mixture.
- Heat the mixture to 60-80°C for 8-24 hours to achieve high conversion and isolate the difluoroalkylated product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this technology offers compelling advantages by decoupling the production of fluorinated intermediates from the supply constraints of exotic fluorinating reagents. The reliance on commodity chemicals such as aryl boronic acids and simple nickel salts creates a more resilient supply chain that is less susceptible to market volatility. Since the starting materials are widely available from multiple global vendors, procurement teams can negotiate better pricing and secure long-term contracts without fear of single-source bottlenecks. Moreover, the operational simplicity of the reaction, which does not require specialized high-pressure equipment or extreme temperature control, allows for manufacturing in standard multipurpose reactors. This flexibility enhances supply chain reliability and reduces the capital expenditure required for facility upgrades, making it an attractive option for contract manufacturing organizations looking to expand their fluorination capabilities.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive palladium catalysts with abundant nickel salts and the elimination of costly, hazardous fluorinating agents. By avoiding the use of reagents like DAST, facilities save significantly on waste disposal costs and safety infrastructure requirements. The high atom economy of the coupling reaction further contributes to cost efficiency, as fewer raw materials are wasted in side products. Additionally, the simplified workup procedures reduce solvent consumption and energy usage during isolation, leading to substantial cost savings in the overall production budget without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: The use of stable, shelf-stable starting materials ensures that production schedules can be maintained consistently. Unlike moisture-sensitive fluorinating reagents that require cold chain logistics and special handling, the boronic acids and nickel catalysts used here are robust and easy to store. This stability minimizes the risk of batch failures due to reagent degradation, thereby improving on-time delivery performance. The broad substrate scope also means that a single manufacturing line can be adapted to produce a wide variety of difluoroalkylated compounds, allowing for agile responses to changing demand patterns in the pharmaceutical pipeline and reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: Scaling this reaction to commercial volumes is straightforward due to the absence of exothermic hazards associated with traditional fluorination. The mild reaction conditions allow for safe operation in large-scale reactors, facilitating the transition from kilogram to ton-scale production. From an environmental standpoint, the process generates less hazardous waste, aligning with green chemistry principles and reducing the regulatory burden on manufacturing sites. The avoidance of persistent fluorinated byproducts and the use of recyclable solvents further enhance the sustainability profile of the manufacturing process, supporting corporate ESG goals and ensuring compliance with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its adoption. Common inquiries often revolve around the compatibility of specific functional groups and the scalability of the nickel catalyst system. The following answers address these concerns based on the data provided in the patent literature, offering clarity on the practical implementation of this technology. These insights are intended to assist technical teams in assessing the feasibility of integrating this method into their existing workflows.
Q: What are the advantages of this nickel-catalyzed method over traditional fluorination?
A: Unlike traditional deoxofluorination using hazardous reagents like DAST, this nickel-catalyzed cross-coupling operates under mild conditions with excellent functional group compatibility, avoiding toxic byproducts and harsh temperatures.
Q: Is this method suitable for non-activated difluoroalkyl halides?
A: Yes, a key innovation of this patent is the ability to couple non-activated difluoroalkyl bromides efficiently, overcoming the inertness of the C-Br bond that limits many other transition metal catalyzed methods.
Q: What represents the primary cost driver in this synthesis?
A: The primary cost drivers are the specialized difluoroalkyl bromide starting materials; however, the use of inexpensive nickel salts and common ligands significantly reduces catalyst costs compared to palladium-based alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoroalkyl Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced fluorination technologies in modern drug discovery. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN105669365B can be successfully translated into robust manufacturing processes. We are committed to delivering high-purity difluoroalkyl intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in nickel catalysis and cross-coupling reactions positions us as a trusted partner for developing complex fluorinated building blocks.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your development timeline and optimize your supply chain efficiency.
