Advanced Nickel-Catalyzed Synthesis of Difluoroalkyl-Substituted Aromatics for Commercial Scale-Up
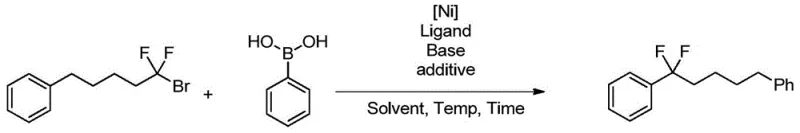
The pharmaceutical and agrochemical industries continuously seek efficient methodologies to incorporate fluorine atoms into organic scaffolds, given the profound impact of fluorination on metabolic stability and bioavailability. Patent CN105669365B discloses a groundbreaking preparation method for aryl or heteroaryl compounds containing difluoroalkyl substitution, addressing critical limitations in current synthetic strategies. This technology leverages a nickel-catalyzed Suzuki coupling reaction between arylboronic acids and functionalized difluoroalkyl halides, offering a robust alternative to traditional deoxofluorination techniques. By utilizing inexpensive nickel salts and nitrogen-containing ligands, the process achieves high conversion rates and exceptional yields under mild conditions. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this innovation represents a significant leap forward in accessing complex fluorinated building blocks essential for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of difluoromethylene groups into aromatic systems has relied heavily on deoxofluorination of carbonyl or aldehyde precursors using aggressive reagents such as DAST (diethylaminosulfur trifluoride) or nucleophilic fluorinating agents like HF-Py. These conventional pathways are fraught with significant drawbacks that hinder their utility in complex molecule synthesis. The fluorinating reagents employed are often highly reactive and incompatible with a multitude of important functional groups, leading to side reactions and decomposition of sensitive substrates. Furthermore, these methods frequently require harsh reaction conditions and generate toxic byproducts, posing safety risks and environmental challenges during scale-up. The inability to tolerate diverse functional groups severely limits the application of these methods for synthesizing advanced difluoromethylene-containing compounds required in cutting-edge medicinal chemistry programs.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a transition metal-catalyzed cross-coupling strategy that circumvents the pitfalls of direct fluorination. By employing a nickel catalyst system in the presence of a base and specific ligands, the method facilitates the coupling of readily available arylboronic acids with difluoroalkyl bromides. This Suzuki-type coupling is remarkably versatile, demonstrating excellent functional group compatibility across a wide range of substrates including those with electron-withdrawing and electron-donating groups. The reaction proceeds efficiently even with non-activated difluoroalkyl halides, a class of substrates that has historically been challenging to activate. This breakthrough enables the synthesis of diverse difluoroalkyl-substituted aromatics with high purity and yield, providing a safer and more sustainable route for manufacturing high-purity OLED material precursors and active pharmaceutical ingredients.
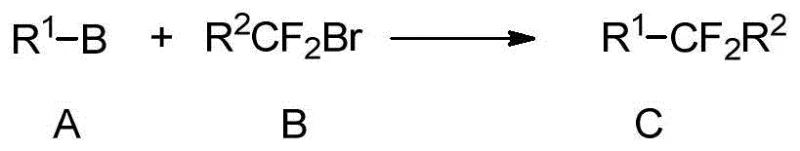
Mechanistic Insights into Nickel-Catalyzed Cross-Coupling
The success of this methodology hinges on the unique ability of the nickel catalyst to activate the carbon-bromine bond in difluoroalkyl halides, which are typically inert due to the lack of conjugation. The catalytic cycle likely initiates with the oxidative addition of the nickel(0) species into the C-Br bond of the difluoroalkyl substrate, forming an organonickel(II) intermediate. This step is facilitated by the presence of bidentate nitrogen ligands such as bipyridine or phenanthroline derivatives, which stabilize the low-valent nickel center and modulate its electronic properties. Subsequent transmetallation with the arylboronic acid, activated by the base, transfers the aryl group to the nickel center. The final reductive elimination step releases the desired difluoroalkyl-substituted product and regenerates the active nickel(0) catalyst. This mechanism allows for the coupling of sterically hindered and electronically diverse partners, ensuring broad applicability in the commercial scale-up of complex polymer additives and fine chemicals.
Impurity control is a critical aspect of this process, particularly given the sensitivity of fluorinated intermediates. The use of mild bases like potassium carbonate and controlled reaction temperatures between 60°C and 80°C minimizes the risk of beta-fluorine elimination, a common side reaction that leads to defluorinated byproducts. Additionally, the choice of solvent, preferably ether-based solvents like triglyme or THF, plays a pivotal role in solubilizing the inorganic base and stabilizing the catalytic species. The protocol's robustness is evidenced by its tolerance to moisture and air to some extent, although inert conditions are preferred for optimal results. By carefully optimizing the ligand-to-catalyst ratio and additive concentration, the process effectively suppresses homocoupling of the boronic acid, ensuring a clean impurity profile that simplifies downstream purification and reduces overall production costs.
How to Synthesize Difluoroalkyl-Substituted Aryl Compounds Efficiently
The synthesis protocol outlined in the patent provides a straightforward procedure for generating these valuable intermediates. The reaction typically involves charging a vessel with the arylboronic acid, nickel catalyst precursor, ligand, and base in a suitable solvent, followed by the addition of the difluoroalkyl bromide. The mixture is then heated to facilitate the coupling. Detailed standardized synthesis steps see the guide below.
- Combine arylboronic acid, nickel catalyst (e.g., NiCl2·DME), nitrogen-containing ligand, and base in a suitable ether solvent.
- Add the functionalized difluoroalkyl bromide substrate to the reaction mixture under inert atmosphere.
- Heat the mixture to 60-80°C for 8-24 hours, then monitor conversion and isolate the product via standard workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this nickel-catalyzed process offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs associated with the catalyst system. Unlike palladium-catalyzed processes which rely on precious metals with volatile pricing, nickel is abundant and significantly cheaper, leading to substantial cost savings in fine chemical manufacturing. Furthermore, the avoidance of hazardous fluorinating agents like DAST eliminates the need for specialized containment equipment and expensive waste disposal protocols, thereby reducing the total cost of ownership for the production facility. The simplicity of the workup procedure, often requiring only standard extraction and chromatography, further enhances operational efficiency and throughput.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with inexpensive nickel salts directly impacts the bill of materials, offering a clear path to margin improvement. Additionally, the high atom economy of the Suzuki coupling minimizes waste generation, aligning with green chemistry principles and reducing environmental compliance costs. The ability to use commercially available starting materials without complex pre-functionalization steps streamlines the supply chain, reducing lead times and inventory holding costs. This economic efficiency makes the process highly attractive for the cost reduction in electronic chemical manufacturing where margin pressures are intense.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as arylboronic acids and simple alkyl bromides ensures a stable and resilient supply chain. These starting materials are produced by multiple global suppliers, mitigating the risk of single-source dependency that often plagues specialized fluorinating reagents. The robustness of the reaction conditions means that production can be maintained consistently without frequent batch failures due to reagent sensitivity. This reliability is crucial for maintaining continuous supply of reliable agrochemical intermediate supplier networks, ensuring that downstream customers receive their materials on schedule without disruption.
- Scalability and Environmental Compliance: The mild reaction temperatures and use of standard ether solvents make this process inherently scalable from gram to multi-ton quantities. The absence of highly toxic reagents simplifies the safety profile of the plant, allowing for easier regulatory approval and lower insurance premiums. Waste streams are easier to treat compared to those generated by sulfur-based fluorination methods, facilitating compliance with increasingly stringent environmental regulations. This scalability supports the commercial scale-up of complex polymer additives and pharmaceutical intermediates, enabling manufacturers to respond quickly to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed difluoroalkylation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for potential adopters of the technology.
Q: What are the advantages of this nickel-catalyzed method over traditional fluorination?
A: Unlike traditional methods using harsh reagents like DAST which have poor functional group compatibility, this nickel-catalyzed Suzuki coupling operates under mild conditions with excellent tolerance for sensitive groups like aldehydes, ketones, and esters.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the method utilizes inexpensive nickel salts instead of precious palladium catalysts and employs common solvents like triglyme or THF, making it highly scalable and cost-effective for industrial production.
Q: What types of substrates are compatible with this reaction?
A: The protocol demonstrates broad substrate scope, successfully coupling various aryl and heteroaryl boronic acids with non-activated difluoroalkyl bromides, including complex molecules like steroid derivatives and protected amino acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoroalkyl Aryl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed synthesis route for producing high-value fluorinated intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of difluoroalkyl-substituted compounds meets the highest industry standards. We understand the critical nature of supply continuity in the pharmaceutical sector and are committed to delivering consistent quality and reliability.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can optimize your supply chain and reduce your overall manufacturing costs. Let us be your partner in bringing innovative fluorinated solutions to market efficiently and sustainably.