Advanced Photoresist Monomer Synthesis for High-Resolution Semiconductor Manufacturing
The semiconductor industry continuously demands higher precision in lithography processes, driving the need for advanced photoresist materials that can resolve increasingly smaller feature sizes. Patent CN111662267B introduces a groundbreaking photoresist acid-producing resin monomer containing a dioxobicyclo[2.2.2]octane dicarboxylic acid ester structure, representing a significant leap forward in electronic chemical engineering. This innovative molecular architecture integrates the photoacid generator directly into the resin backbone, fundamentally addressing the critical issue of acid diffusion that plagues conventional chemically amplified resists. By combining an acetal structure with a robust polycyclic framework, this technology not only enhances the mechanical stability of the resist film but also optimizes the solubility switch mechanism required for high-contrast pattern development. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, understanding the nuances of this synthesis is crucial for securing next-generation manufacturing capabilities. The patent details a sophisticated yet practical approach to monomer design that balances high performance with synthetic feasibility, offering a compelling value proposition for companies aiming to reduce lead time for high-purity photoresist monomers in their supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoresist formulations typically rely on the physical blending of a separate photoacid generator (PAG) with a base resin matrix, a method that inherently suffers from thermodynamic instability and molecular mobility issues during the exposure and post-exposure bake processes. In these conventional systems, the generated acid molecules are free to diffuse laterally within the resist film, leading to blurred pattern edges, increased line width roughness, and a consequent loss of resolution that limits the minimum feature size achievable in integrated circuit fabrication. Furthermore, the lack of covalent bonding between the acid source and the polymer backbone often results in inconsistent acid distribution, causing variability in dissolution rates that complicates the development process and reduces overall yield. From a supply chain perspective, sourcing high-purity separate components and ensuring their compatibility adds layers of complexity and cost to the manufacturing workflow, often requiring extensive purification steps to remove unreacted PAG residues that could contaminate the final device. These limitations highlight the urgent need for a molecularly integrated solution that locks the acid generating capability directly into the structural framework of the resist material itself.
The Novel Approach
The novel approach described in the patent overcomes these historical barriers by chemically tethering the photoacid generator to the resin monomer through a carefully designed ester linkage, effectively immobilizing the acid source until the precise moment of activation. This structural integration ensures that the generated acid is produced exactly where it is needed within the polymer matrix, drastically reducing lateral diffusion and enabling the creation of sharper, more defined patterns with superior edge roughness characteristics. The incorporation of the dioxobicyclo[2.2.2]octane moiety adds significant rigidity and thermal stability to the molecule, enhancing the etching resistance of the final resist film and allowing it to withstand the harsh plasma environments used in downstream patterning steps. Additionally, the presence of acetal groups introduces a pH-sensitive cleavage point that facilitates rapid solubility changes upon exposure, improving the contrast ratio and ensuring clean development without scumming or residue formation. This holistic design strategy not only elevates the technical performance of the photoresist but also streamlines the formulation process, offering a clear pathway for cost reduction in display & optoelectronic materials manufacturing by simplifying the raw material inventory.
Mechanistic Insights into Acid-Generating Resin Polymerization
The core innovation lies in the unique molecular architecture that combines a sulfonium cation with a specialized anion derived from hydroxyl-containing sulfonates, all anchored to a polymerizable acrylate backbone via a multi-step esterification sequence. Upon exposure to radiation, the sulfonium moiety undergoes photolysis to release a strong sulfonic acid, which then catalyzes the cleavage of the adjacent acetal protecting groups within the same molecule or neighboring chains. This intramolecular or near-neighbor acid catalysis ensures a highly localized deprotection reaction, converting the hydrophobic polymer segments into hydrophilic carboxylic acid or hydroxyl groups that are readily soluble in aqueous alkaline developers. The dioxobicyclo structure serves as a rigid spacer that prevents the collapse of the resist pattern during development, maintaining the vertical profile of the features even at high aspect ratios. 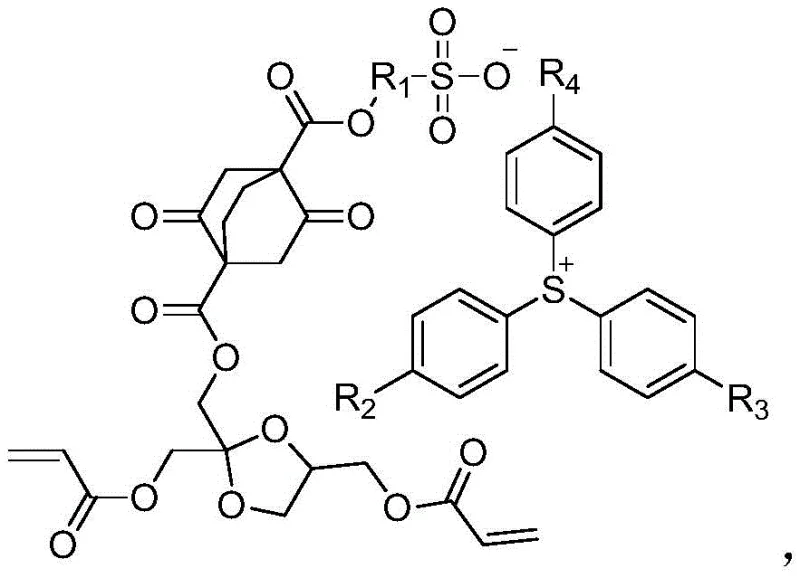
Furthermore, the mechanism leverages the specific electronic properties of the fluorinated alkyl groups attached to the sulfonate anion to tune the acidity strength, ensuring that the generated acid is potent enough to drive the deprotection reaction efficiently without causing excessive background fogging. The polymerization capability is preserved through the terminal acrylate double bonds, allowing this monomer to be copolymerized with other functional monomers to fine-tune the glass transition temperature and mechanical properties of the final resist resin. This level of molecular control allows manufacturers to tailor the resist performance for specific lithography wavelengths, whether it be deep ultraviolet or extreme ultraviolet applications. The precise arrangement of the ester linkages also contributes to the overall solubility profile, ensuring that the unexposed regions remain insoluble while the exposed regions dissolve rapidly, a critical factor for achieving high-fidelity pattern transfer in commercial scale-up of complex polymer additives.
How to Synthesize Photoresist Monomer Efficiently
The synthesis of this advanced monomer follows a logical four-step sequence that prioritizes yield and purity, beginning with the formation of a cyclic hemiketal intermediate through the condensation of polyhydroxy acrylates. This initial step establishes the acid-labile core of the molecule, setting the stage for subsequent functionalization with the rigid bicyclic acid and the photoactive sulfonate group. The process utilizes common organic solvents like toluene and dichloromethane, along with standard acid catalysts such as p-toluenesulfonic acid, making it accessible for facilities equipped with standard fine chemical reactors. Careful control of reaction temperatures and stoichiometry is essential to minimize side reactions and ensure the integrity of the sensitive acetal and ester functionalities throughout the synthesis. 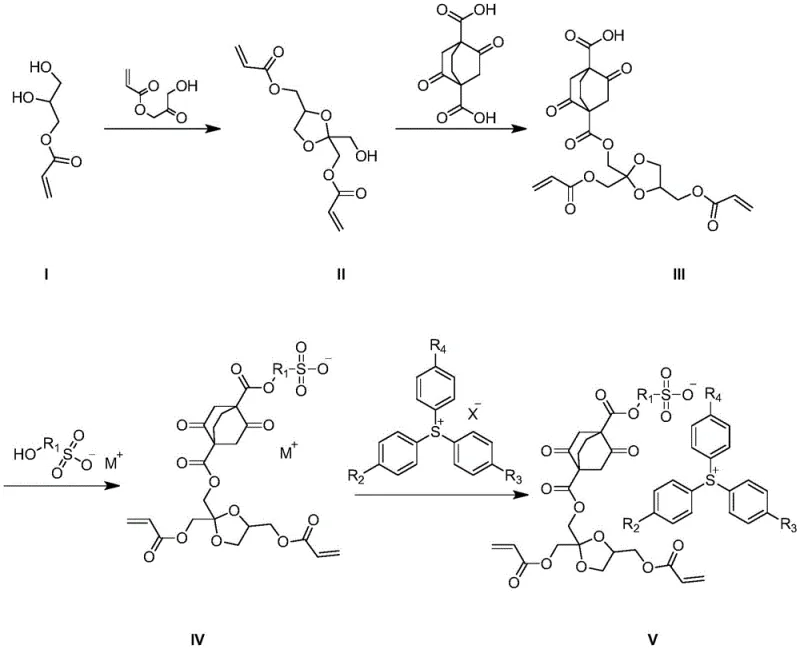
- Perform acetal reaction between 2,3-dihydroxypropyl acrylate and 3-hydroxy-2-carbonyl acrylate to form the cyclic hemiketal intermediate.
- Conduct esterification with 2,5-dioxobicyclo[2.2.2]octane-1,4-dicarboxylic acid using p-toluenesulfonic acid catalyst.
- React the resulting intermediate with hydroxyl-containing sulfonate and subsequently with triphenyl sulfur halide to finalize the resin monomer structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement standpoint, the adoption of this integrated monomer technology offers substantial strategic benefits by consolidating multiple functional requirements into a single raw material, thereby simplifying the supply chain and reducing the risk of component mismatch. The synthetic route relies on commercially available starting materials such as acrylic acid derivatives and sulfonium salts, which mitigates the risk of supply disruptions associated with exotic or proprietary reagents often found in niche electronic chemical markets. By eliminating the need for separate PAG blending and the associated quality control checks for mixture homogeneity, manufacturers can significantly streamline their production workflows and reduce the overall operational overhead. This consolidation of functionality translates directly into enhanced supply chain reliability, as fewer distinct chemical entities need to be sourced, qualified, and stored, allowing for more agile response times to fluctuating market demands.
- Cost Reduction in Manufacturing: The streamlined synthesis pathway eliminates the need for expensive transition metal catalysts or complex purification protocols often required to remove residual metal contaminants from electronic grade chemicals. By utilizing organic acid catalysts and standard workup procedures like extraction and recrystallization, the process inherently lowers the cost of goods sold while maintaining the high purity standards required for semiconductor applications. The high yields reported in the patent examples suggest that raw material utilization is efficient, minimizing waste generation and further contributing to overall economic viability. Additionally, the improved performance of the resist means that less material may be required per wafer to achieve the same patterning results, driving down the effective cost per unit for the end user.
- Enhanced Supply Chain Reliability: The reliance on robust chemical transformations such as esterification and nucleophilic substitution ensures that the manufacturing process is resilient to minor variations in operating conditions, leading to consistent batch-to-batch quality. This predictability is vital for supply chain heads who must guarantee uninterrupted delivery of critical materials to fabrication plants operating on tight schedules. The use of stable intermediates that can be isolated and characterized provides additional checkpoints for quality assurance, reducing the likelihood of off-spec product reaching the customer. Furthermore, the scalability of the reaction conditions means that production volumes can be ramped up quickly without the need for specialized high-pressure or cryogenic equipment, facilitating a more responsive supply network.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods that might involve heavy metals or volatile organic compounds in larger quantities. The solvents used, such as toluene and ethyl acetate, are well-understood in industrial hygiene contexts and can be effectively recovered and recycled, aligning with modern green chemistry initiatives and environmental regulations. The solid nature of the final monomer product simplifies packaging and transportation, reducing the carbon footprint associated with logistics. This environmental compatibility not only meets regulatory requirements but also appeals to downstream customers who are increasingly scrutinizing the sustainability credentials of their supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photoresist monomer technology in industrial settings. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical properties for potential partners. Understanding these details is essential for evaluating the fit of this material within existing manufacturing lines and for planning future procurement strategies effectively.
Q: How does this monomer structure improve photoresist resolution?
A: The monomer covalently bonds the photoacid generator to the resin backbone, preventing acid diffusion during post-exposure baking, which significantly reduces line edge roughness and improves pattern fidelity.
Q: What are the solubility characteristics of this new resin?
A: The inclusion of polyester-based structures and specific alkyl groups enhances solubility in fat-soluble solvents, ensuring uniform spin-coating and consistent film formation on substrates.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the synthetic route utilizes standard esterification and substitution reactions with readily available catalysts like p-toluenesulfonic acid, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoresist Monomer Supplier
As the semiconductor and display industries continue to push the boundaries of miniaturization, the demand for high-performance photoresist materials has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring cutting-edge innovations like the dioxobicyclo-based monomer to the global market. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs ensuring that every batch meets the exacting standards required for advanced lithography applications. We understand that consistency is key in electronic chemical manufacturing, and our state-of-the-art facilities are designed to deliver the reliability and precision that top-tier technology companies demand.
We invite you to collaborate with us to explore how this advanced monomer technology can enhance your product portfolio and optimize your manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating the tangible economic benefits of switching to this integrated resin system. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to drive the next generation of electronic material innovation forward.
