Advanced Synthesis of Nitrile-Functionalized Adamantane Monomers for Next-Gen Lithography
Introduction to High-Performance Lithography Monomers
The relentless miniaturization of semiconductor elements has driven the industry towards shorter wavelength radiation sources, specifically KrF, ArF, and F2 excimer laser beams. To meet the stringent resolution requirements of modern photolithography, novel photoresist materials are essential. Patent CN1780812A introduces a groundbreaking class of adamantane derivatives characterized by a specific structure containing both a nitrile group and a (meth)acryloyloxy moiety. These compounds serve as critical monomers for functional resins, offering a unique balance of acid sensitivity, dry etching resistance, and substrate adhesion. The general structure, represented by Formula (I), allows for significant tunability in resin properties, addressing the limitations of earlier generations of photoresist materials.
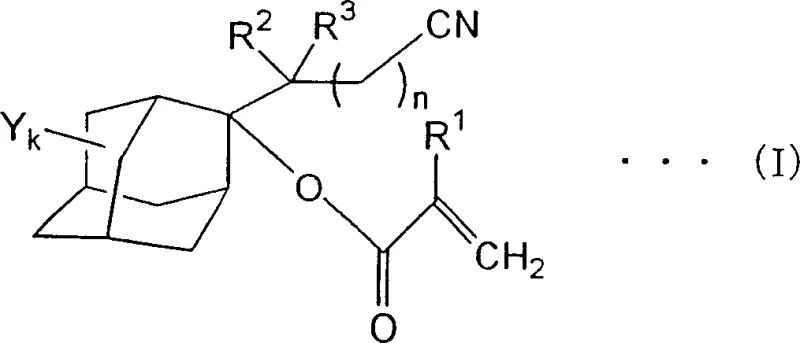
This technological advancement is particularly relevant for manufacturers seeking a reliable photoresist monomer supplier capable of delivering high-purity electronic chemical intermediates. The patent details a robust synthetic pathway that avoids the complexities of multi-step protection and deprotection sequences often found in traditional methods. By directly functionalizing the adamantane cage with a nitrile-containing side chain, the process streamlines production while enhancing the final polymer's performance in high-resolution patterning applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of photoresist resins relied heavily on simple adamantyl esters or cyclic hydrocarbon groups. While these materials provided basic dry etching resistance, they often suffered from poor adhesion to silicon substrates or insufficient transparency at deep UV wavelengths. Prior art, such as Patent Document 4, suggested introducing organic groups containing nitrile moieties to improve adhesion. However, these disclosures were largely theoretical regarding adamantane scaffolds, lacking specific embodiments that successfully combined a nitrile group with a (meth)acryloyloxy functionality on the same adamantane core. Conventional synthesis routes often struggled with regioselectivity, leading to complex mixtures of isomers that were difficult and costly to separate, thereby impacting the overall yield and purity required for semiconductor-grade materials.
The Novel Approach
The methodology disclosed in CN1780812A represents a paradigm shift in cost reduction in electronic chemical manufacturing by utilizing a direct nucleophilic addition strategy. Instead of building the carbon skeleton from scratch, the process leverages the readily available adamantanone as a starting material. By reacting this ketone with a nitrile compound in the presence of a strong base, the nitrile-containing side chain is installed with high precision. This is immediately followed by esterification with (meth)acrylic acid derivatives. This telescoped approach eliminates the need for isolating unstable intermediates and significantly reduces solvent usage and waste generation. The result is a streamlined process that delivers high-purity semiconductor process chemicals with superior consistency, directly addressing the supply chain bottlenecks associated with complex specialty chemical synthesis.
Mechanistic Insights into Base-Catalyzed Nucleophilic Addition
The core of this synthesis lies in the controlled generation of a carbanion at the alpha-position of the nitrile group. Strong bases such as n-butyllithium, lithium diisopropylamide (LDA), or sodium hydride are employed to abstract the acidic alpha-proton from the nitrile compound (e.g., acetonitrile or propionitrile). This generates a highly reactive nucleophile that attacks the electrophilic carbonyl carbon of the adamantanone. The steric environment of the adamantane cage dictates the approach of the nucleophile, ensuring that the addition occurs exclusively at the carbonyl position without affecting the rigid cage structure. The reaction is typically conducted at cryogenic temperatures, ranging from -80°C to 0°C, to suppress competing side reactions such as the self-polymerization of the nitrile or the formation of elimination by-products.
Following the formation of the lithium alkoxide intermediate, the second stage involves esterification. The alkoxide acts as a nucleophile attacking the carbonyl of a (meth)acrylic acid anhydride or acid halide. This step is crucial for installing the polymerizable vinyl group. The use of anhydrides is preferred over acid halides to minimize the formation of corrosive halide salts, which can complicate downstream purification. The mechanism ensures that the acid-labile nature of the resulting ester is preserved, which is vital for the chemical amplification mechanism in the final photoresist. Rigorous control of moisture and oxygen is maintained throughout to prevent hydrolysis of the sensitive intermediates, ensuring the final product meets the stringent purity specifications demanded by the semiconductor industry.
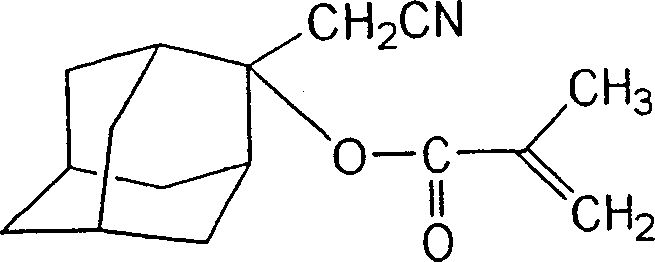
How to Synthesize 2-Cyanomethyl-2-adamantyl Methacrylate Efficiently
The practical implementation of this chemistry is best illustrated by the synthesis of 2-cyanomethyl-2-adamantyl methacrylate, a preferred embodiment of the invention. This specific compound demonstrates the efficacy of using acetonitrile and adamantanone to create a monomer with exceptional thermal and chemical stability. The process begins with the preparation of the nitrile anion in dry tetrahydrofuran (THF) under an inert atmosphere. Precise temperature control is maintained using a dry ice/isopropanol bath to keep the reaction mixture at -78°C during the addition of the base. Once the adamantanone is introduced, the mixture is allowed to warm slowly to 0°C to complete the addition, achieving conversion rates exceeding 99.5% as confirmed by gas chromatography.
- Deprotonate acetonitrile using n-BuLi at -78°C in THF to form the nucleophile.
- Add adamantanone solution dropwise to the nitrile anion mixture and warm to 0°C to form the lithium alkoxide intermediate.
- React the alkoxide intermediate with methacrylic anhydride and triethylamine at 0°C, followed by aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits beyond mere technical performance. The reliance on commodity chemicals like adamantanone, acetonitrile, and methacrylic anhydride ensures a stable and resilient supply chain, reducing the risk of raw material shortages that often plague specialty chemical production. Furthermore, the elimination of transition metal catalysts removes the need for expensive and time-consuming metal scavenging steps, which are typically required to meet the ultra-low metal content specifications of the electronics industry. This simplification of the purification train translates directly into substantial cost savings and reduced lead times for high-purity photoresist monomers.
- Cost Reduction in Manufacturing: The process avoids the use of precious metal catalysts and complex protecting group strategies. By utilizing a direct addition-esterification sequence, the number of unit operations is minimized. This reduction in processing steps lowers energy consumption and solvent waste disposal costs. Additionally, the high selectivity of the reaction reduces the burden on purification columns, allowing for higher throughput and better overall equipment effectiveness in commercial plants.
- Enhanced Supply Chain Reliability: The starting materials are bulk chemicals with established global supply networks, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of pressures and temperatures within safe operating limits, ensures consistent batch-to-batch quality. This reliability is critical for maintaining continuous production schedules in the fast-paced semiconductor manufacturing sector, where delays can have cascading effects on downstream device fabrication.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, having been demonstrated effectively from laboratory to pilot scales without loss of yield or selectivity. The absence of heavy metals and the use of recyclable solvents like THF and hexane align with modern green chemistry principles. Waste streams are primarily organic and saline, which are easier to treat compared to heavy metal-contaminated waste, facilitating compliance with increasingly strict environmental regulations governing chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel adamantane derivatives. The answers are derived directly from the experimental data and mechanistic understanding provided in the patent literature, ensuring accuracy and relevance for technical decision-makers evaluating this technology for integration into their supply chains.
Q: Why are nitrile-containing adamantane derivatives preferred for ArF lithography?
A: These derivatives offer superior dry etching resistance and UV transmittance compared to traditional alicyclic esters. The nitrile group enhances adhesion to substrates without compromising the acid sensitivity required for chemically amplified resists.
Q: How does the patented synthesis method control impurities?
A: The process utilizes low-temperature conditions (-78°C to 0°C) and specific strong bases like n-BuLi or LDA to ensure high regioselectivity. This minimizes side reactions such as polymerization of the methacrylate group or over-alkylation, achieving selectivity up to 99.5%.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the reaction uses commercially available starting materials like adamantanone and acetonitrile. The exothermic nature is manageable with standard cooling systems, and the workup involves simple washing and precipitation, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyanomethyl-2-adamantyl Methacrylate Supplier
The synthesis of nitrile-functionalized adamantane monomers represents a significant opportunity for advancing lithography capabilities, but translating this patent chemistry into commercial reality requires expert process engineering. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art cryogenic reactors and rigorous QC labs capable of meeting the stringent purity specifications required for electronic grade chemicals. We understand the critical nature of impurity profiles in photoresist monomers and employ advanced analytical techniques to ensure every batch performs consistently in your formulations.
We invite you to engage with our technical team to explore how this technology can optimize your current resin portfolio. By requesting a Customized Cost-Saving Analysis, you can gain insights into potential efficiency gains specific to your volume requirements. Our technical procurement team is prepared to provide specific COA data and route feasibility assessments to help you make informed sourcing decisions. Contact us today to secure a stable supply of high-performance monomers for your next-generation lithography projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
