Advanced Ruthenium-Catalyzed Synthesis of Polysubstituted Distyryl Indole Derivatives for Commercial Scale
Introduction to Next-Generation Indole Synthesis
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for greener, more efficient manufacturing processes. Patent CN109678862B introduces a groundbreaking methodology for the preparation of polysubstituted distyryl indole derivatives, a class of compounds critical to modern drug discovery. These molecular scaffolds are indispensable in the design of antineoplastic, anti-infective, and anti-inflammatory agents, yet their traditional synthesis has often been plagued by harsh conditions and environmental hazards. This new approach leverages a sophisticated Ruthenium-catalyzed system that operates effectively under mild, aqueous conditions, marking a substantial leap forward in sustainable chemical manufacturing. By enabling the direct functionalization of indole cores with high precision, this technology offers a robust pathway for producing complex pharmaceutical intermediates with exceptional purity profiles.
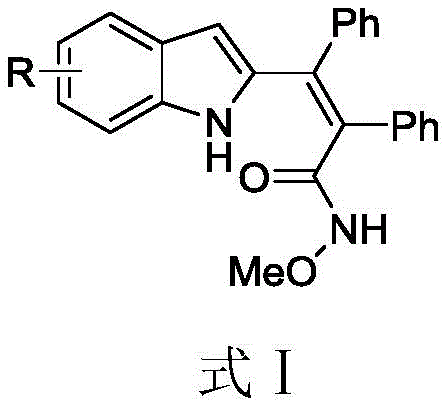
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of vinyl indole derivatives has relied heavily on transition metal catalysis involving Nickel or Cobalt systems, which present significant operational challenges for large-scale production. Previous methodologies, such as those reported by Nakao and Hiyama or the Yoshikai group, typically necessitate the use of toxic organic solvents like toluene and tetrahydrofuran, creating severe safety and disposal burdens for manufacturing facilities. Furthermore, these conventional routes often require elevated temperatures and extended reaction times, leading to increased energy consumption and potential thermal degradation of sensitive substrates. The reliance on strictly anhydrous conditions in many of these legacy processes also complicates the supply chain, requiring rigorous drying of reagents and solvents, which drives up both capital expenditure and operational costs. Consequently, there has been a persistent demand for a synthetic strategy that mitigates these risks while maintaining high yields and structural diversity.
The Novel Approach
The innovative protocol detailed in the patent data revolutionizes this synthetic landscape by utilizing a water-compatible Ruthenium(II) catalytic system that functions efficiently at room temperature. As illustrated in the reaction scheme below, the process couples substituted N-methoxyindole carboxamides with diphenylacetylene in a biphasic solvent system comprising water and dichloromethane in a 9:1 ratio. This unique solvent environment not only enhances the safety profile by minimizing volatile organic compound (VOC) emissions but also simplifies the workup procedure significantly. The reaction proceeds rapidly within just 3 hours, achieving yields as high as 99% for various substrates, which drastically reduces the residence time required in production reactors. This shift towards aqueous-phase chemistry represents a paradigm change, allowing manufacturers to access complex distyryl indole structures without the heavy environmental footprint associated with traditional organic synthesis.
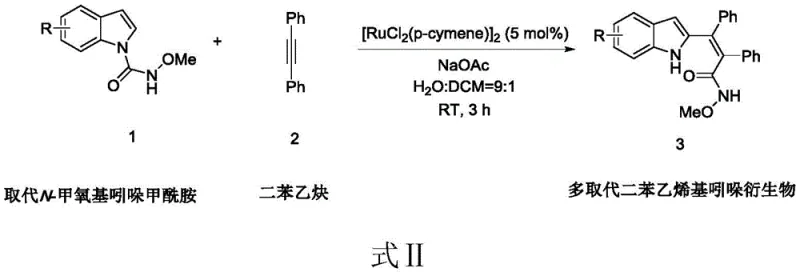
Mechanistic Insights into Ruthenium-Catalyzed C-H Activation
The success of this transformation lies in the precise orchestration of C-H activation facilitated by the N-methoxy amide directing group. The Ruthenium catalyst, specifically dichlorobis(p-cymene)ruthenium(II) dimer, coordinates with the oxygen atom of the amide moiety, positioning the metal center in close proximity to the C2 position of the indole ring. This coordination enables the selective cleavage of the C-H bond, generating a reactive organometallic intermediate that subsequently undergoes migratory insertion with the alkyne substrate. The presence of sodium acetate acts as a crucial base to facilitate the proton abstraction step, regenerating the active catalytic species and driving the cycle forward. This mechanism ensures high regioselectivity, preventing the formation of unwanted isomers that often complicate purification in less controlled systems. The robustness of this catalytic cycle allows it to tolerate a wide range of electronic environments on the indole ring, from electron-withdrawing halogens to electron-donating methoxy groups.
From an impurity control perspective, the mild reaction conditions play a pivotal role in ensuring the integrity of the final product. Operating at room temperature minimizes the risk of thermal decomposition or polymerization of the reactive alkene intermediates, which are common side reactions in high-temperature processes. The use of a biphasic system also aids in sequestering polar byproducts and inorganic salts into the aqueous layer, leaving the organic phase relatively clean prior to final purification. This inherent selectivity reduces the burden on downstream processing, meaning that the crude product obtained after solvent removal is already of high quality. For R&D teams focused on scaling, this translates to a more predictable impurity profile and a streamlined path to meeting stringent regulatory specifications for pharmaceutical intermediates.
How to Synthesize Polysubstituted Distyryl Indole Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and mixing efficiency to maximize the benefits of the biphasic system. The standard protocol involves charging the reactor with the indole substrate, diphenylacetylene, the ruthenium catalyst at 5 mol% loading, and two equivalents of sodium acetate. The reaction is then initiated by adding the water-dichloromethane solvent mixture, followed by vigorous stirring to ensure adequate interfacial contact between the phases. Detailed standardized operating procedures for this synthesis, including specific mixing rates and quenching protocols, are outlined in the structured guide below.
- Charge the reactor with substituted N-methoxyindole carboxamide, diphenylacetylene, dichlorobis(p-cymene)ruthenium(II) dimer, and sodium acetate.
- Add the solvent mixture of water and dichloromethane (9: 1 ratio) and stir the reaction mixture at room temperature for 3 hours.
- Concentrate the filtrate using a rotary evaporator and purify the crude product via silica gel column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aqueous-phase synthesis offers tangible strategic advantages that extend beyond mere chemical efficiency. The elimination of toxic solvents like toluene and THF significantly reduces the regulatory compliance burden and lowers the costs associated with hazardous waste disposal and solvent recovery systems. By shifting to a water-rich solvent system, facilities can leverage existing infrastructure more safely, reducing the need for specialized explosion-proof equipment often required for large volumes of flammable organics. This transition directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering the total cost of ownership for the production line. Furthermore, the short reaction time of 3 hours increases the throughput capacity of existing reactors, allowing for greater production volume without additional capital investment in new hardware.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive, strictly anhydrous solvents and reduces energy consumption by operating at ambient temperature. This qualitative shift removes the costly steps of solvent drying and heating, leading to substantial operational savings. Additionally, the high yields observed across diverse substrates minimize raw material waste, ensuring that expensive starting materials like substituted indoles are utilized with maximum efficiency. The simplified workup, involving basic rotary evaporation and standard chromatography, further reduces labor and processing time compared to multi-step traditional syntheses.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents, such as diphenylacetylene and common ruthenium salts, ensures a robust supply chain that is less susceptible to disruptions. The tolerance for various substituents means that a single platform technology can be used to produce a wide library of derivatives, reducing the need for multiple specialized synthesis lines. This flexibility allows suppliers to respond more rapidly to changing demands from drug developers who may require different analogs during the lead optimization phase. The stability of the reagents also simplifies storage and logistics, reducing the risk of spoilage during transport.
- Scalability and Environmental Compliance: The aqueous nature of the reaction aligns perfectly with modern green chemistry principles, making it easier to obtain environmental permits for scale-up operations. The reduction in volatile organic compounds (VOCs) improves workplace safety and reduces the environmental impact of the manufacturing site. Scaling this process from gram to kilogram or ton scale is facilitated by the exotherm management inherent in room-temperature reactions, removing the need for complex cooling systems. This environmental friendliness enhances the corporate sustainability profile of the manufacturer, a key metric for many global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this Ruthenium-catalyzed synthesis. These answers are derived directly from the experimental data and scope analysis provided in the patent documentation, ensuring accuracy for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of this route for specific project requirements.
Q: What are the specific reaction conditions for this synthesis?
A: The reaction utilizes a biphasic solvent system of water and dichloromethane (9:1) at room temperature for 3 hours, employing 5 mol% of a Ruthenium catalyst.
Q: Does this method support diverse substituent groups on the indole ring?
A: Yes, the protocol demonstrates excellent tolerance for various substituents including chlorine, bromine, methoxy, and methyl groups at different positions on the indole scaffold.
Q: How is the product purified after the reaction?
A: Following the reaction, the solvent is removed via rotary evaporation, and the resulting crude product is purified using standard silica gel column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Distyryl Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the acceleration of drug development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of distyryl indole derivatives meets the exacting standards required for clinical and commercial applications. Our commitment to technical excellence allows us to navigate the complexities of C-H activation chemistry with precision and reliability.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and reduce your overall time to market.
