Advanced Synthesis of Polysubstituted Distyryl Indole Derivatives for Commercial Pharmaceutical Applications
Advanced Synthesis of Polysubstituted Distyryl Indole Derivatives for Commercial Pharmaceutical Applications
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for greener, more efficient manufacturing processes. Patent CN109678862B introduces a groundbreaking preparation method for novel polysubstituted distyryl indole derivatives, addressing critical bottlenecks in the production of high-value pharmaceutical intermediates. This technology leverages a ruthenium-catalyzed C-H activation strategy that operates under remarkably mild conditions, specifically utilizing an aqueous phase at room temperature. For R&D directors and process chemists, this represents a paradigm shift away from harsh, energy-intensive protocols towards sustainable chemistry that does not compromise on yield or purity. The ability to synthesize complex vinyl indole scaffolds with such precision opens new avenues for drug discovery, particularly for antineoplastic and anti-inflammatory agents where these molecular fragments are indispensable.
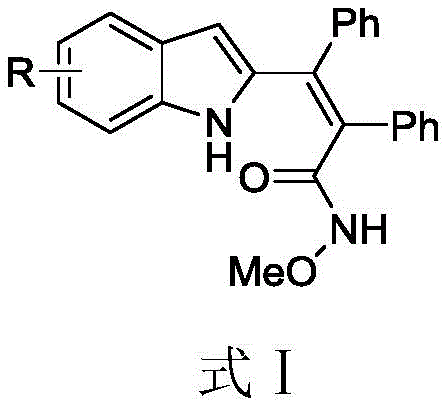
The core innovation lies in the specific structural versatility offered by this synthetic route. As illustrated in the general formula, the method accommodates a wide range of substituents (R) including chlorine, bromine, methoxy, and methyl groups on the indole ring. This flexibility is crucial for medicinal chemists who require diverse libraries of compounds for structure-activity relationship (SAR) studies. By enabling the direct functionalization of the indole core with diphenylacetylene, the process eliminates the need for pre-functionalized starting materials, thereby streamlining the supply chain and reducing the overall step count. This efficiency translates directly into cost savings and faster time-to-market for downstream API manufacturers seeking reliable sources of complex heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vinyl indole derivatives has been plagued by significant operational and environmental challenges that hinder large-scale adoption. Prior art, such as the nickel-catalyzed methods reported by Nakao and Hiyama in 2006, often necessitates the use of N-methyl or ester-based indoles and requires rigorous exclusion of moisture and oxygen. Furthermore, the cobalt-catalyzed approaches developed by the Yoshikai group in 2012, while effective, typically rely on N-pyrimidinyl indoles and demand the use of toxic organic solvents like toluene and tetrahydrofuran (THF). These solvents pose serious health and safety risks, require complex recovery systems, and generate substantial hazardous waste, driving up the cost of compliance and disposal. Additionally, these conventional routes frequently suffer from long reaction times and the need for elevated temperatures, which can lead to thermal degradation of sensitive intermediates and the formation of difficult-to-remove impurities.
The Novel Approach
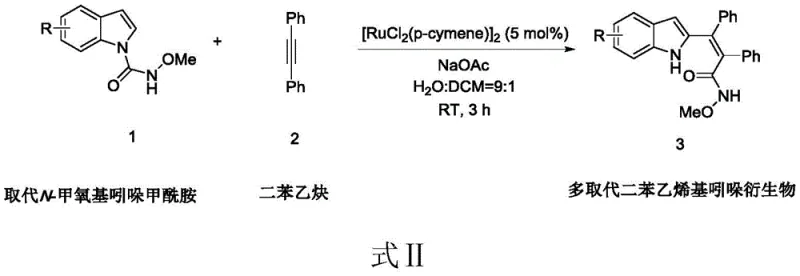
In stark contrast, the methodology disclosed in CN109678862B offers a robust and environmentally benign alternative that overcomes these historical limitations. The process utilizes a water-dichloromethane biphasic system (9:1 ratio), which not only reduces the reliance on volatile organic compounds but also facilitates easier product isolation. The reaction proceeds efficiently at room temperature (RT) within just 3 hours, demonstrating exceptional kinetic proficiency without the need for external heating. This mildness preserves the integrity of sensitive functional groups, ensuring high product purity and minimizing the formation of side products. The use of dichlorobis(4-methylisopropylphenyl)ruthenium(II) as a catalyst provides a highly active species that promotes the coupling of substituted N-methoxyindole formamide with diphenylacetylene with remarkable selectivity.
The operational simplicity of this novel approach cannot be overstated. The reaction mixture is simply stirred, and upon completion, the solvent is removed via rotary evaporation to yield a crude product that is readily purified by standard silica gel column chromatography. This straightforward workup procedure stands in sharp contrast to the multi-step quenching and extraction protocols often required for air-sensitive organometallic reactions. For procurement managers, this simplicity implies a reduction in processing time and labor costs, while for supply chain heads, it suggests a more robust process that is less prone to batch-to-batch variability caused by strict environmental controls. The high yields reported across various substrates further underscore the reliability of this method for commercial scale-up.
Mechanistic Insights into Ruthenium-Catalyzed C-H Activation
The success of this synthesis hinges on the unique reactivity of the ruthenium(II) catalyst in facilitating directed C-H activation. The N-methoxyamide moiety serves as a powerful directing group, coordinating to the ruthenium center and positioning it proximal to the C2-H bond of the indole ring. This coordination lowers the activation energy for C-H bond cleavage, allowing the metal to insert into the bond under mild conditions. Following C-H activation, the coordinatively unsaturated ruthenium species undergoes migratory insertion with the diphenylacetylene substrate. This step is critical as it forms the new carbon-carbon bond that extends the conjugation of the indole system, creating the distyryl motif. The subsequent reductive elimination or protonolysis releases the final product and regenerates the active catalytic species, closing the catalytic cycle.
From an impurity control perspective, the mechanism offers distinct advantages. The high regioselectivity imposed by the directing group ensures that functionalization occurs exclusively at the desired position, preventing the formation of regioisomers that are notoriously difficult to separate. Furthermore, the mild reaction conditions prevent the polymerization of the alkyne or the decomposition of the indole core, which are common side reactions in harsher thermal processes. The aqueous environment may also play a role in stabilizing transition states through hydrophobic effects, enhancing the reaction rate and selectivity. This mechanistic understanding allows process chemists to fine-tune reaction parameters, such as catalyst loading and stoichiometry, to optimize the impurity profile for GMP manufacturing.
How to Synthesize Polysubstituted Distyryl Indole Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol begins with the precise weighing of substituted N-methoxyindole carboxamide and diphenylacetylene in a 1:1 molar ratio, ensuring complete consumption of the valuable alkyne. The catalyst, [RuCl2(p-cymene)]2, is added at a loading of 5 mol%, which strikes an optimal balance between catalytic activity and cost efficiency. Sodium acetate acts as a base to facilitate the C-H activation step by neutralizing the acid generated during the cycle. The choice of solvent is pivotal; a 9:1 mixture of water and dichloromethane creates a heterogeneous system that maximizes the solubility of organic reactants while leveraging the green properties of water. Detailed standardized synthesis steps follow below.
- Charge a reactor with substituted N-methoxyindole carboxamide, diphenylacetylene, dichlorobis(4-methylisopropylphenyl)ruthenium(II) catalyst, and sodium acetate.
- Add a solvent mixture of water and dichloromethane (9: 1 ratio) and stir the reaction mixture at room temperature for 3 hours.
- Concentrate the filtrate using a rotary evaporator to obtain the crude product, then purify via silica gel column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this patented process offers compelling economic and logistical benefits. The shift to a room-temperature aqueous process fundamentally alters the cost structure of manufacturing these complex intermediates. By eliminating the need for energy-intensive heating and specialized anhydrous solvents, the utility costs associated with production are significantly reduced. Moreover, the high atom economy of the coupling reaction ensures that raw materials are converted efficiently into the desired product, minimizing waste generation and the associated disposal fees. This aligns perfectly with modern green chemistry initiatives, potentially qualifying the process for regulatory incentives or preferred supplier status with environmentally conscious pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive, air-sensitive ligands and the use of a commercially available ruthenium dimer catalyst drastically lowers the raw material costs. Furthermore, the simplified workup procedure, which avoids complex extractions and extensive drying steps, reduces labor hours and solvent consumption. The high yields observed across a broad substrate scope mean that less starting material is required to produce the same amount of API intermediate, directly improving the margin profile for procurement teams negotiating supply contracts.
- Enhanced Supply Chain Reliability: The reagents used in this process, such as diphenylacetylene and various substituted indoles, are commodity chemicals with robust global supply chains. Unlike specialized organometallic reagents that may have long lead times or single-source dependencies, these materials are readily available from multiple vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Additionally, the stability of the reaction conditions reduces the likelihood of batch failures, ensuring a consistent and reliable flow of material to downstream customers.
- Scalability and Environmental Compliance: The use of water as the primary solvent significantly reduces the environmental footprint of the manufacturing process, simplifying compliance with increasingly stringent environmental regulations. The biphasic nature of the reaction mixture facilitates easy separation of the organic product, reducing the load on wastewater treatment facilities. This scalability is crucial for meeting the demands of commercial API production, where tonnage quantities are often required. The process is inherently safer, operating at ambient pressure and temperature, which lowers insurance premiums and reduces the need for specialized high-pressure reactor infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What are the optimal reaction conditions for synthesizing distyryl indole derivatives?
A: The optimal conditions involve using a water/dichloromethane (9:1) solvent system at room temperature for 3 hours, with a catalyst loading of 5 mol% [RuCl2(p-cymene)]2.
Q: Does this method tolerate various substituents on the indole ring?
A: Yes, the method demonstrates excellent functional group tolerance, successfully accommodating electron-withdrawing groups like bromine and chlorine, as well as electron-donating groups like methoxy and methyl.
Q: How does this aqueous phase method compare to traditional organic synthesis routes?
A: Unlike traditional methods requiring toxic solvents like toluene or THF and elevated temperatures, this protocol operates at room temperature in an environmentally friendly aqueous phase, significantly simplifying workup and reducing environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Distyryl Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and scalable synthetic routes for next-generation pharmaceutical intermediates. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN109678862B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are designed to handle complex organometallic chemistry safely and efficiently, adhering to the highest standards of quality and safety.
We invite you to collaborate with us to leverage this advanced synthesis method for your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this greener route can improve your margins. Please contact us today to request specific COA data for our catalog of indole derivatives or to discuss route feasibility assessments for your proprietary molecules. Let us be your partner in accelerating your drug development timeline with high-quality, cost-effective chemical solutions.
