Advanced Manufacturing of Arotinolol Hydrochloride: A Novel One-Pot Coupling Strategy for Global Supply
Advanced Manufacturing of Arotinolol Hydrochloride: A Novel One-Pot Coupling Strategy for Global Supply
The pharmaceutical landscape for cardiovascular therapeutics demands increasingly efficient and environmentally sustainable manufacturing processes for key active ingredients. Arotinolol Hydrochloride, a potent selective beta-1 adrenoceptor antagonist used in the treatment of essential hypertension and angina, has historically faced synthesis challenges related to toxic reagent usage and low overall yields. The recent disclosure in patent CN112300149A introduces a transformative preparation method that addresses these critical bottlenecks. This innovative approach leverages a benzyl-protected side-chain intermediate and a sophisticated one-pot coupling strategy to deliver high-purity intermediates suitable for global supply chains. By fundamentally redesigning the connection between the thiophene core and the amino-alcohol side chain, this technology offers a robust pathway for reliable arotinolol hydrochloride supplier networks seeking to optimize their production capabilities.
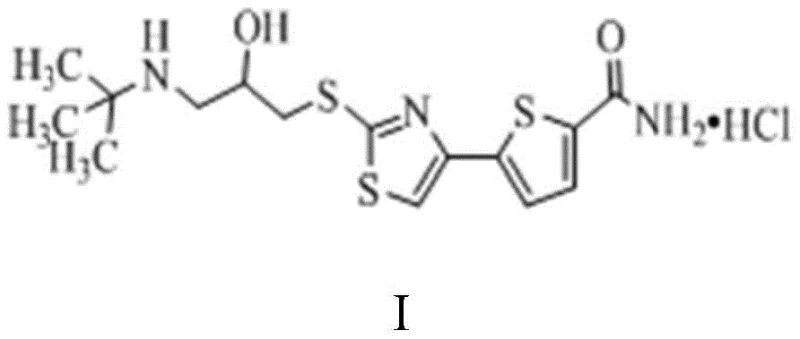
The significance of this patent lies in its ability to bypass the limitations of prior art, which often relied on hazardous halogenation agents and suffered from significant material loss during purification. The new methodology ensures that the final product meets rigorous quality specifications while drastically simplifying the operational workflow. For procurement and technical teams evaluating long-term sourcing strategies, understanding the mechanistic advantages of this route is essential for securing a stable supply of high-quality cardiovascular intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Arotinolol Hydrochloride has been plagued by two primary technical hurdles that impact both cost and environmental compliance. First, traditional routes frequently employ aggressive reagents such as elemental bromine and thionyl chloride to functionalize the thiophene ring, creating substantial pressure on environmental health and safety (EHS) protocols and waste treatment infrastructure. Second, and perhaps more critically, the construction of the side chain via the direct reaction of tert-butylamine with epichlorohydrin is chemically inefficient. As illustrated in the comparative analysis of prior art, this direct alkylation tends to produce significant quantities of dimer impurities, structurally identified as Compound IX in the patent literature.
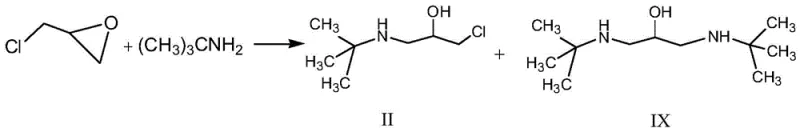
The formation of these dimeric byproducts not only consumes valuable raw materials but also complicates downstream purification, often necessitating energy-intensive column chromatography or repeated recrystallizations that erode the overall process yield. Furthermore, the intermediate 1-tert-butylamino-3-chloro-2-propanol generated in these older routes is inherently unstable, leading to decomposition and further yield losses during isolation. These factors combine to create a manufacturing process that is neither economically viable nor environmentally sustainable for modern large-scale production requirements.
The Novel Approach
The patented methodology presented in CN112300149A offers a decisive break from these inefficiencies by introducing a benzyl-protecting group strategy for the amine component. Instead of reacting tert-butylamine directly, the process utilizes benzyl tert-butylamine to react with epichlorohydrin, forming the stable protected intermediate (Compound III). This structural modification effectively sterically hinders the formation of the problematic dimer impurities, ensuring a much cleaner reaction profile. The true brilliance of this approach, however, is realized in the final coupling stage, where the protecting group is removed and the fragments are joined in a single operational sequence.
This novel route eliminates the need to isolate the unstable deprotected amine intermediate. By performing the catalytic deprotection and subsequent nucleophilic substitution in the same reactor without intermediate workup, the process minimizes material handling losses and solvent consumption. This "telescoped" operation not only accelerates the production cycle but also significantly reduces the generation of chemical waste, aligning perfectly with the principles of green chemistry and providing a clear pathway for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Catalytic Hydrogen Transfer and One-Pot Coupling
The core chemical innovation of this synthesis lies in the seamless integration of catalytic hydrogen transfer and nucleophilic substitution. In the final stage, Compound III undergoes deprotection in the presence of ammonium formate and a palladium on carbon (Pd/C) catalyst. Ammonium formate serves as a safe and convenient hydrogen donor, facilitating the cleavage of the benzyl group under mild conditions (20-30°C) to generate the reactive free amine in situ. This avoids the use of high-pressure hydrogen gas, thereby enhancing operational safety and reducing equipment complexity.
Once the free amine is generated, it immediately participates in a nucleophilic attack on the thiazole-thiophene core (Compound IV). The reaction is promoted by potassium carbonate and potassium iodide, where the iodide ion acts as a catalyst to facilitate the displacement of the leaving group on the side chain. This concerted mechanism ensures that the highly reactive free amine is consumed as soon as it is formed, preventing potential degradation or side reactions. The result is a highly efficient coupling that delivers the target molecule with exceptional purity, demonstrating a sophisticated control over reaction kinetics that is vital for producing high-purity beta-blocker intermediates.
How to Synthesize Arotinolol Hydrochloride Efficiently
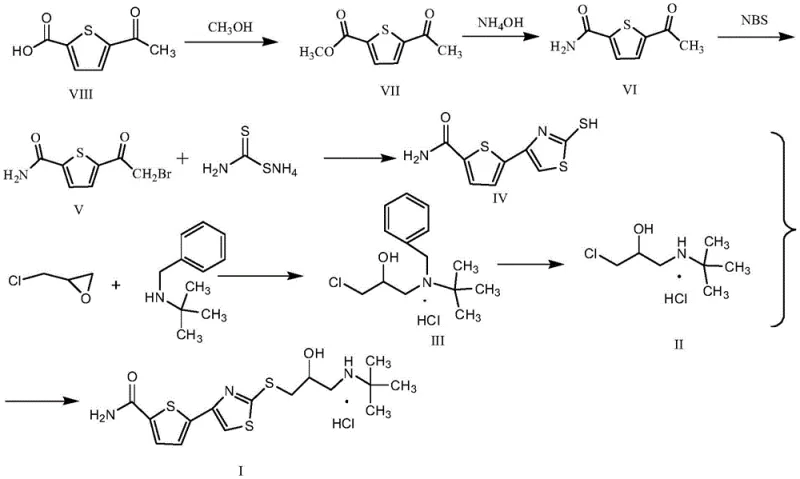
The synthesis of Arotinolol Hydrochloride via this patented route involves a logical sequence of transformations designed for maximum efficiency. The process begins with the conversion of 5-acetyl-2-thiophenecarboxylic acid into the corresponding amide core, followed by the separate preparation of the protected amino-alcohol side chain. The critical innovation occurs in the final step, where these two advanced intermediates are united. The detailed standardized operating procedures, including specific molar ratios, temperature controls, and workup protocols required to replicate this high-yield process, are outlined below for technical reference.
- Preparation of the thiophene core (Compound IV) starting from 5-acetyl-2-thiophenecarboxylic acid via esterification, ammonolysis, alpha-bromination, and cyclization with ammonium dithiocarbamate.
- Synthesis of the protected side chain (Compound III) by reacting benzyl tert-butylamine with epichlorohydrin in the presence of a phase transfer catalyst.
- One-pot coupling: Catalytic deprotection of Compound III using ammonium formate/Pd-C, followed immediately by nucleophilic substitution with Compound IV to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits beyond mere chemical elegance. The elimination of toxic reagents like bromine and thionyl chloride simplifies regulatory compliance and reduces the costs associated with hazardous waste disposal. Moreover, the avoidance of column chromatography—a technique notoriously difficult to scale and expensive to operate—means that production can be executed using standard stainless steel reactors, significantly lowering capital expenditure barriers for scale-up.
- Cost Reduction in Manufacturing: The implementation of a one-pot deprotection and coupling strategy fundamentally alters the cost structure of production. By removing the isolation and purification steps for the unstable intermediate, the process saves substantial amounts of solvents, filtration media, and labor hours. The higher overall yield resulting from the suppression of dimer impurities means that less raw material is required to produce the same amount of finished API, driving down the unit cost of goods sold without compromising quality standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as benzyl tert-butylamine and ammonium dithiocarbamate, mitigates the risk of raw material shortages. The robustness of the reaction conditions, which tolerate mild temperatures and atmospheric pressure, ensures consistent batch-to-batch reproducibility. This stability is crucial for maintaining continuous supply lines to downstream formulation partners, reducing the lead time for high-purity pharmaceutical intermediates and preventing stockouts.
- Scalability and Environmental Compliance: The process is inherently designed for the commercial scale-up of complex pharmaceutical intermediates. The use of ammonium formate as a hydrogen source eliminates the need for specialized high-pressure hydrogenation equipment, allowing for easier technology transfer between manufacturing sites. Furthermore, the significant reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, future-proofing the supply chain against potential regulatory crackdowns on chemical emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Arotinolol Hydrochloride using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: How does this novel route improve upon traditional Arotinolol synthesis methods?
A: Traditional methods often utilize toxic reagents like bromine and thionyl chloride, and suffer from low yields due to dimer impurity formation during side-chain synthesis. This patented route employs a benzyl-protected intermediate to prevent dimerization and utilizes a one-pot deprotection-coupling strategy to eliminate isolation steps, significantly enhancing overall yield and environmental safety.
Q: What is the purity profile of the Arotinolol Hydrochloride produced via this method?
A: The process is designed to meet stringent regulatory standards. Following a specific recrystallization protocol involving ethyl acetate, methanol, and water, the final product achieves a purity exceeding 99.9% as determined by HPLC, with maximum single impurities controlled below 0.05%.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the route is explicitly optimized for industrial application. It avoids complex purification techniques like column chromatography, relies on commercially available raw materials, and consolidates multiple reaction steps into a single vessel, making it highly suitable for large-scale manufacturing from 100 kgs to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arotinolol Hydrochloride Supplier
The technological advancements detailed in patent CN112300149A represent a significant leap forward in the manufacturing of cardiovascular therapeutics, offering a cleaner, faster, and more economical route to market. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative chemistry to life. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of Arotinolol Hydrochloride we produce meets the highest international standards for safety and efficacy.
We invite global pharmaceutical partners to collaborate with us to leverage this superior synthesis route for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your next project benefits from the most advanced and reliable manufacturing technologies available in the industry.
