Advanced Catalytic Synthesis of Arotinolol Hydrochloride for Commercial Pharmaceutical Production
Introduction to the Novel Preparation Method
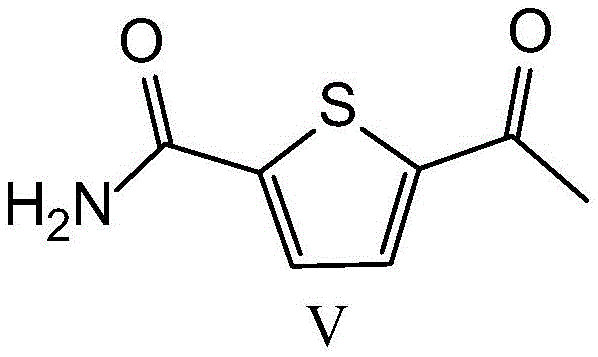
The pharmaceutical industry constantly seeks more efficient and environmentally benign pathways for producing critical cardiovascular medications. Patent CN115124522A introduces a groundbreaking preparation method for arotinolol hydrochloride, a selective beta-1 adrenoceptor antagonist widely used for treating essential hypertension and angina pectoris. This innovation fundamentally shifts the synthetic paradigm by replacing traditional stoichiometric brominating agents with a catalytic amount of copper bromide. By utilizing 5-acetylthiophene-2-formamide as the primary raw material, the process achieves a one-pot bromination-cyclization to generate the key intermediate 5-(2-mercapto-4-thiazolyl)-2-thiophenecarboxamide. This technical leap not only streamlines the production workflow but also aligns with modern green chemistry principles, offering a robust solution for manufacturers aiming to optimize their supply chains for high-purity beta-blocker intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of arotinolol hydrochloride has been plagued by significant operational and environmental hurdles. Early methodologies, such as those disclosed in patent EP 0021840, relied heavily on the use of toxic and hazardous liquid bromine for the bromination step. This approach necessitates rigorous safety protocols, specialized corrosion-resistant equipment, and complex purification procedures to remove bromine residues, all of which drive up operational expenditures. Furthermore, alternative routes described in Chinese patent CN104370900A utilized high-boiling point solvents like dimethyl sulfoxide (DMSO) in the final salting-out stages. The presence of such solvents poses a severe risk of excessive residue in the final active pharmaceutical ingredient (API), complicating quality control and potentially failing stringent regulatory limits. Additionally, some prior art routes involved the construction of side chains requiring protection and deprotection steps using noble metal catalysts, resulting in poor atom economy and inflated production costs due to the length of the synthetic sequence.
The Novel Approach
The methodology presented in CN115124522A offers a decisive break from these inefficiencies by implementing a catalytic bromocyclization strategy. Instead of using hazardous liquid bromine or expensive N-bromosuccinimide (NBS) in stoichiometric quantities, the process employs a catalytic amount of copper bromide (CuBr2). This modification allows for the direct conversion of 5-acetylthiophene-2-formamide into the thiazole-containing intermediate in a single pot, effectively shortening the synthetic route by eliminating unnecessary isolation steps. The convergence of the mother nucleus and the side chain is achieved through a streamlined condensation reaction that avoids the need for high-boiling solvents in the final purification. By shifting to lower boiling solvents like methanol and ethanol for crystallization, the new method ensures easier solvent removal and superior product purity. This approach not only enhances the economic viability of the process but also significantly mitigates the environmental footprint associated with traditional halogenation chemistries.
Mechanistic Insights into Copper-Catalyzed Bromocyclization
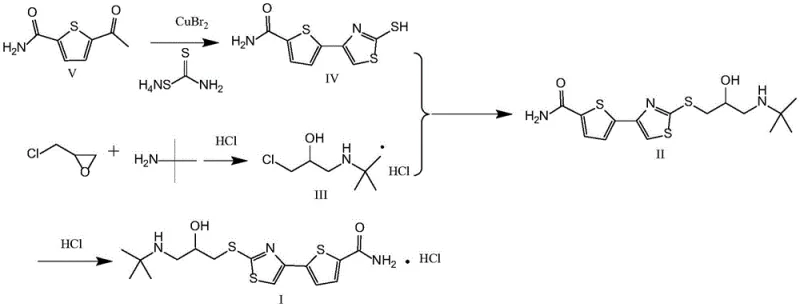
The core of this technological advancement lies in the mechanistic efficiency of the copper-catalyzed alpha-bromination and subsequent cyclization. In the first critical step, 5-acetylthiophene-2-formamide undergoes alpha-bromination mediated by copper(II) bromide. Unlike free-radical bromination which can lead to poly-brominated byproducts and regio-isomers, the copper-mediated pathway offers superior selectivity for the alpha-position of the acetyl group. Following the in-situ generation of the alpha-bromo ketone, the reaction mixture is treated with ammonium dithiocarbamate. This reagent acts as both a sulfur source and a nitrogen source, facilitating a rapid cyclization to form the thiazole ring. The synergy between the copper catalyst and the dithiocarbamate ensures that the reaction proceeds smoothly to form 5-(2-mercapto-4-thiazolyl)-2-thiophenecarboxamide (Formula IV) with high specificity. This mechanism effectively suppresses the formation of tar-like impurities often seen in harsh bromination conditions, thereby simplifying the downstream purification process and boosting the overall yield of the key heterocyclic intermediate.
Impurity control is further enhanced by the mild reaction conditions employed throughout the synthesis. The use of catalytic copper bromide operates effectively at temperatures ranging from 70°C to 110°C, which is sufficient to drive the reaction kinetics without promoting thermal degradation of the sensitive thiophene and thiazole moieties. Moreover, the subsequent coupling of the thiol intermediate (Formula IV) with the chloro-amino alcohol side chain (Formula III) is conducted under controlled alkaline conditions using sodium hydroxide. This step is critical for preventing the hydrolysis of the amide group while ensuring the selective alkylation of the thiol sulfur. The careful modulation of pH and temperature during this nucleophilic substitution minimizes the formation of O-alkylated byproducts or bis-alkylated impurities. The final salt formation with hydrochloric acid is executed in methanol, a solvent choice that promotes the precipitation of the hydrochloride salt while keeping organic impurities in solution, thus acting as an effective purification step in itself.
How to Synthesize Arotinolol Hydrochloride Efficiently
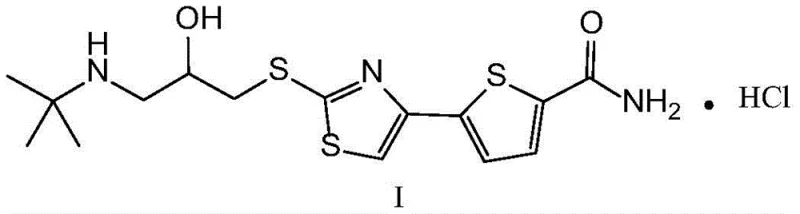
The synthesis of arotinolol hydrochloride via this patented route involves a logical sequence of four distinct chemical transformations that prioritize yield and purity. The process begins with the construction of the heterocyclic core, followed by the independent preparation of the amino-alcohol side chain, and concludes with their convergence and final salification. This modular approach allows for parallel processing of intermediates, which is highly advantageous for scaling up production timelines. The detailed standardized operating procedures, including specific molar ratios, solvent volumes, and temperature profiles optimized for maximum efficiency, are outlined in the comprehensive guide below for technical teams ready to implement this superior methodology.
- Prepare the key intermediate 5-(2-mercapto-4-thiazolyl)-2-thiophenecarboxamide (Formula IV) using 5-acetylthiophene-2-formamide and catalytic copper bromide in a one-pot reaction.
- Synthesize the side chain 1-chloro-3-[(2-methyl-2-propyl)amino]-2-propanol hydrochloride (Formula III) by reacting tert-butylamine with epichlorohydrin followed by acidification.
- Condense the thiazole intermediate (Formula IV) with the side chain (Formula III) under alkaline conditions to form free base arotinolol (Formula II).
- Convert the free base into the final hydrochloride salt (Formula I) using hydrochloric acid in methanol, followed by crystallization and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits regarding cost stability and operational reliability. The shift from stoichiometric hazardous reagents to catalytic systems fundamentally alters the cost structure of the manufacturing process. By eliminating the need for large quantities of liquid bromine or NBS, the process reduces the expenditure on expensive and regulated raw materials. Furthermore, the simplified workup procedures mean less time and resources are spent on waste treatment and solvent recovery, leading to substantial cost savings in the overall production budget. The robustness of the reaction conditions also implies a lower rate of batch failures, ensuring a more consistent supply of high-quality intermediates for downstream API synthesis.
- Cost Reduction in Manufacturing: The implementation of a catalytic bromination system drastically reduces the consumption of brominating agents, which are often subject to volatile market pricing and strict transportation regulations. By using copper bromide in catalytic amounts rather than stoichiometric quantities, the material cost per kilogram of the intermediate is significantly lowered. Additionally, the avoidance of noble metal catalysts in the side-chain synthesis removes a major cost driver found in alternative routes. The streamlined process also reduces energy consumption by shortening the reaction time and eliminating high-temperature distillation steps required for removing high-boiling solvents like DMSO, resulting in a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials such as 5-acetylthiophene-2-formamide and tert-butylamine ensures a resilient supply chain that is less susceptible to disruptions. Unlike processes that require specialized or custom-synthesized precursors, this method utilizes commodity chemicals that can be sourced from multiple global suppliers. The simplified purification steps, which avoid complex chromatographic separations or difficult crystallizations from high-boiling solvents, accelerate the production cycle time. This efficiency allows manufacturers to respond more quickly to market demand fluctuations, reducing lead times for high-purity pharmaceutical intermediates and ensuring continuous availability for client production schedules.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this green chemistry approach offers significant advantages for commercial scale-up. The reduction in hazardous waste generation, particularly the elimination of bromine-contaminated waste streams, simplifies compliance with increasingly stringent environmental protection laws. The use of common solvents like ethanol and methanol facilitates easier recycling and disposal compared to specialized polar aprotic solvents. This environmental friendliness not only lowers the cost of waste management but also future-proofs the manufacturing site against potential regulatory crackdowns on toxic emissions, making it a sustainable choice for long-term commercial production of complex heterocyclic compounds.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this route represents a superior choice for modern pharmaceutical manufacturing. Understanding these details is crucial for technical teams evaluating process transfer and for procurement specialists assessing the long-term viability of the supply source.
Q: What is the primary advantage of using catalytic copper bromide over liquid bromine in this synthesis?
A: The use of catalytic copper bromide significantly reduces the environmental hazard and toxicity associated with handling stoichiometric amounts of liquid bromine or N-bromosuccinimide (NBS). It simplifies the workup procedure, lowers raw material costs, and improves the overall atom economy of the bromination-cyclization step.
Q: How does this new method address solvent residue issues found in previous patents?
A: Unlike prior art methods that utilized high-boiling point solvents like DMSO in the final salting-out steps, this novel route employs lower boiling solvents such as methanol and ethanol for crystallization. This change drastically reduces the risk of excessive solvent residues in the final API, ensuring higher purity and easier compliance with ICH guidelines.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability. It features a shortened synthetic route, avoids expensive noble metal catalysts in the side-chain construction, and utilizes robust, one-pot reactions that minimize intermediate isolation steps, thereby enhancing throughput and operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arotinolol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the catalytic bromocyclization route described in CN115124522A and is fully prepared to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of arotinolol hydrochloride intermediate we produce adheres to the highest international quality standards.
We invite forward-thinking pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cost-effective and environmentally friendly technology. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will optimize your supply chain and enhance your product portfolio with high-quality cardiovascular intermediates.
