Advanced Synthesis of Fluoranthene Derivatives for High-Efficiency Blue OLED Manufacturing
The landscape of organic semiconductor materials is continuously evolving, driven by the relentless demand for higher efficiency and stability in display technologies. Patent CN102924296B introduces a significant advancement in this field by disclosing a novel class of fluoranthene-containing organic compounds tailored for organic electroluminescent applications. This intellectual property addresses critical limitations found in earlier generations of blue light-emitting materials, specifically focusing on molecular engineering to enhance solubility and thermal stability. By strategically incorporating electron-donating or electron-withdrawing substituent groups at specific positions on the triphenylamine moiety, the inventors have created a versatile platform for high-performance blue emitters. For procurement and R&D teams seeking a reliable OLED material supplier, understanding the structural nuances and synthetic accessibility of these compounds is paramount for securing a stable supply chain for next-generation flat-panel displays.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of blue light-emitting materials has been fraught with challenges regarding efficiency and operational stability compared to their green and red counterparts. Early benchmarks, such as the rigid fluoranthene derivative 7,8,10-triphenylfluoranthene (TPF), demonstrated high fluorescence quantum efficiency but suffered from significant processing drawbacks. The primary issue lay in the poor solubility of these unsubstituted aromatic systems, which severely hampered solution-processable fabrication techniques and often led to low synthesis yields. Furthermore, materials like 4-(7,10-diphenylfluoranthene)-N,N'-diphenylaniline (TPADPF), while improving hole transport through diphenylamine introduction, still exhibited tendencies towards easy crystallization. This crystallization results in poor film-forming properties, creating pinholes and defects in the emissive layer that drastically reduce device lifetime and luminous efficiency, making them less viable for commercial scale-up of complex electronic chemical manufacturing.
The Novel Approach
The methodology outlined in the patent data presents a robust solution by modifying the molecular architecture to include flexible alkyl chains, alkoxy groups, or specific functional groups like cyano or nitro moieties. This structural modification fundamentally alters the physical properties of the material without compromising the electronic characteristics of the fluoranthene core. The introduction of these substituents disrupts the strong pi-pi stacking interactions that typically lead to aggregation and crystallization, thereby significantly enhancing solubility in common organic solvents. Improved solubility directly translates to better processability during device fabrication, allowing for the formation of uniform, defect-free thin films. Additionally, the patent describes a streamlined synthetic route that leverages these substituents to facilitate purification, ultimately leading to higher overall yields and a more cost-effective production pathway for high-purity OLED material.
Mechanistic Insights into Pd-Catalyzed Coupling and Cyclization
The synthesis of these advanced fluoranthene derivatives relies on a sophisticated three-step sequence that ensures high regioselectivity and purity. The process initiates with a Sonogashira cross-coupling reaction, where a brominated triphenylamine precursor reacts with trimethylsilylacetylene. This transformation is catalyzed by a dual metal system typically comprising an organopalladium species, such as tetrakis(triphenylphosphine)palladium or bis(triphenylphosphine)palladium dichloride, alongside cuprous iodide as a co-catalyst. The reaction proceeds in triethylamine under an inert atmosphere, ensuring the sensitive acetylene functionality is preserved while efficiently forming the carbon-carbon bond. This step is critical as it installs the protected alkyne handle necessary for the subsequent ring-closing reaction, and the use of the trimethylsilyl protecting group prevents unwanted polymerization or side reactions during the coupling phase.
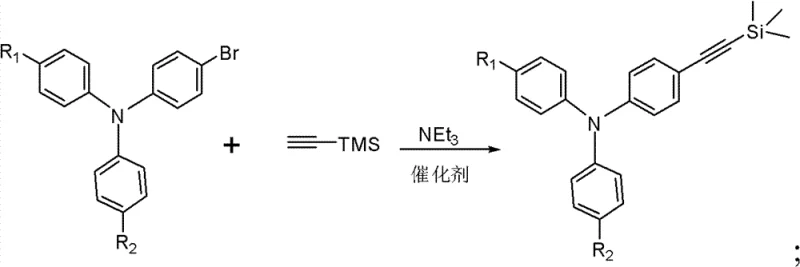
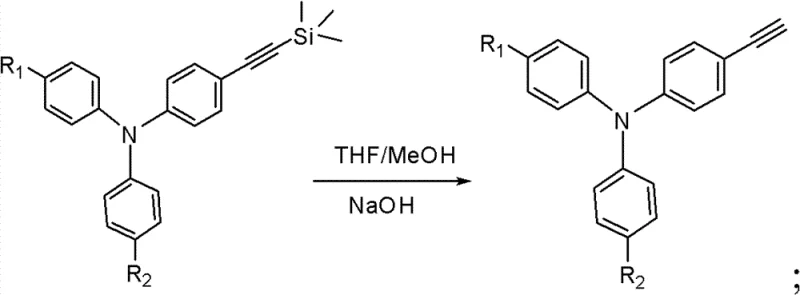
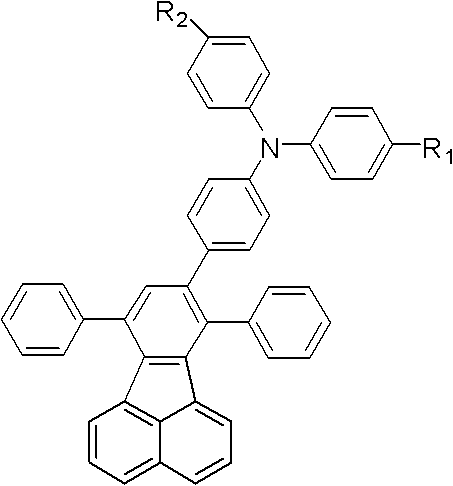
Following the initial coupling, the trimethylsilyl protecting group is removed via a mild deprotection strategy using sodium hydroxide in a mixed solvent system of tetrahydrofuran and methanol. This step reveals the terminal alkyne, which is highly reactive and ready for the final cyclization. The concluding step involves a thermal condensation reaction between the ethynyl-triphenylamine intermediate and 7,9-diphenyl-cyclopentenaphthalen-8-one. Conducted at elevated temperatures around 170°C in xylenes under inert gas, this reaction constructs the rigid fluoranthene core through an intramolecular cyclization mechanism. The high thermal stability of the resulting fluoranthene group, combined with the hole-transporting capability of the triphenylamine unit, creates a balanced charge transport material ideal for blue electroluminescence.
How to Synthesize Fluoranthene Organic Compound Efficiently
Executing this synthesis requires strict adherence to inert atmosphere techniques and precise temperature control to maximize yield and minimize impurities. The process is designed to be scalable, moving from gram-scale laboratory optimization to potential multi-kilogram production. The initial coupling reaction typically requires reflux conditions for extended periods, often up to 48 hours, to ensure complete conversion of the brominated starting material. Subsequent purification steps involve standard silica gel column chromatography, which is facilitated by the enhanced solubility of the intermediates. The final cyclization step is the most energy-intensive, requiring heating at 170°C for 24 hours, but it delivers the final product with excellent structural integrity. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Perform Sonogashira coupling of 4-bromo-triphenylamine derivatives with trimethylsilylacetylene using Pd/Cu catalysts in triethylamine.
- Execute deprotection of the trimethylsilyl group using NaOH in a THF/Methanol mixture to yield the terminal alkyne intermediate.
- Conduct thermal cyclization with 7,9-diphenyl-cyclopentenaphthalen-8-one in xylene at 170°C under inert atmosphere to form the final fluoranthene core.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the transition to this new class of fluoranthene derivatives offers tangible strategic benefits beyond mere performance metrics. The enhanced solubility of these materials simplifies the downstream processing requirements, reducing the need for aggressive solvents or high-energy dispersion techniques during device manufacturing. This ease of processing translates directly into cost reduction in electronic chemical manufacturing by lowering energy consumption and minimizing waste generation associated with failed coating runs. Furthermore, the synthetic route utilizes readily available starting materials and established catalytic systems, mitigating the risk of raw material shortages that often plague specialty chemical supply chains. The robustness of the three-step synthesis ensures a consistent supply of high-quality material, essential for maintaining production schedules in the fast-paced display industry.
- Cost Reduction in Manufacturing: The improved solubility and film-forming properties of the substituted fluoranthene compounds eliminate the need for complex formulation adjustments or expensive additives often required for poorly soluble emitters. By streamlining the purification process through better solubility profiles, manufacturers can achieve substantial cost savings in solvent usage and chromatography media. Additionally, the higher synthesis yields reported in the patent examples mean less raw material is wasted per kilogram of final product, optimizing the overall cost of goods sold without compromising on the stringent purity specifications required for electronic applications.
- Enhanced Supply Chain Reliability: The synthetic pathway relies on commodity chemicals such as triethylamine, xylene, and standard palladium catalysts, which are widely available from multiple global suppliers. This diversification of the supply base reduces dependency on single-source vendors for exotic precursors, thereby enhancing supply chain resilience. The ability to synthesize various derivatives by simply changing the R1 and R2 substituents allows for flexible production planning, enabling manufacturers to quickly adapt to specific customer requirements for different emission wavelengths or charge transport properties without retooling the entire production line.
- Scalability and Environmental Compliance: The reaction conditions, while requiring elevated temperatures for the final step, do not necessitate extreme pressures or hazardous reagents that would complicate regulatory compliance. The use of standard organic solvents allows for established recovery and recycling protocols, aligning with modern environmental sustainability goals. The scalability of the Sonogashira coupling and thermal cyclization steps has been demonstrated in similar industrial contexts, suggesting a smooth path from pilot plant to commercial scale-up of complex polymer additives and small molecule OLED materials, ensuring long-term availability for large-area display manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and application scope of the fluoranthene derivatives. Understanding these details is crucial for technical teams evaluating the feasibility of integrating these materials into existing device architectures.
Q: Why are alkyl or alkoxy substituents introduced into the fluoranthene structure?
A: Introducing substituents like alkyl or alkoxy groups significantly improves the solubility of the material, which enhances synthesis yield and film-forming properties, addressing the crystallization issues of unsubstituted analogs.
Q: What represents the key catalytic step in this synthesis route?
A: The initial Sonogashira coupling reaction utilizing a palladium and copper catalyst system is critical for installing the acetylene functionality required for the subsequent ring-closing cyclization.
Q: What is the primary application of these fluoranthene derivatives?
A: These compounds are designed as emitting layer materials for organic electroluminescent devices, specifically targeting high-efficiency and stable blue light emission for next-generation displays.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoranthene Organic Compound Supplier
As the demand for high-performance blue OLED materials continues to surge, partnering with an experienced CDMO is essential for navigating the complexities of organic semiconductor synthesis. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis critical for preventing device degradation. We understand that the reliability of your display panels depends on the quality of every molecule, and our commitment to excellence guarantees that our fluoranthene derivatives meet the highest industry standards for efficiency and stability.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthetic routes can reduce your overall material costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Let us collaborate to bring the next generation of efficient and stable blue electroluminescent devices to market faster and more economically.
