Advanced Synthesis of Fluoranthene Derivatives for High-Efficiency Blue OLED Manufacturing
The landscape of organic light-emitting diode (OLED) technology is continuously evolving, driven by the demand for higher efficiency and longer lifespans in next-generation flat-panel displays. A pivotal advancement in this domain is documented in patent CN102924296B, which discloses a novel class of fluoranthene-containing organic compounds designed specifically to overcome the limitations of traditional blue emitters. Unlike conventional rigid structures that suffer from poor processability, these new derivatives incorporate specific substituent groups—ranging from alkyl chains to electron-withdrawing cyano groups—to engineer superior solubility and film-forming properties. This strategic molecular design not only facilitates easier purification and processing but also maintains the high hole mobility and thermal stability intrinsic to the fluoranthene core. For industry leaders seeking a reliable OLED material supplier, understanding the synthetic nuances of these compounds is essential for securing a stable supply chain of high-performance electronic chemicals.
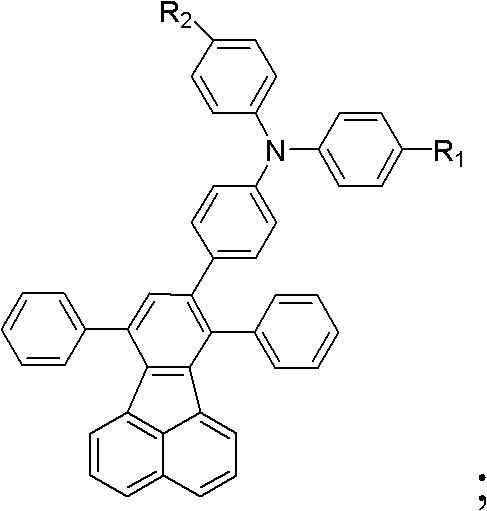
The development of efficient blue electroluminescent materials has long been a bottleneck in the commercialization of full-color OLED displays. Historically, materials like 7,8,10-triphenylfluoranthene (TPF) offered high fluorescence quantum efficiency but were plagued by synthetic complexity and low yields due to their rigid, planar structures which promote aggregation and crystallization. Subsequent iterations, such as diphenylamine-substituted derivatives, improved hole transport but often failed to address the fundamental issue of solubility, leading to difficulties in forming uniform thin films. The novel approach detailed in the patent data introduces a versatile modular synthesis where R1 and R2 groups can be independently tuned. By incorporating bulky alkyl or alkoxy chains, the intermolecular forces are effectively modulated, preventing unwanted crystallization during film deposition while enhancing the overall synthesis yield. This represents a significant leap forward in cost reduction in electronic chemical manufacturing, as higher yields and better solubility directly translate to reduced waste and lower production costs per kilogram of active material.
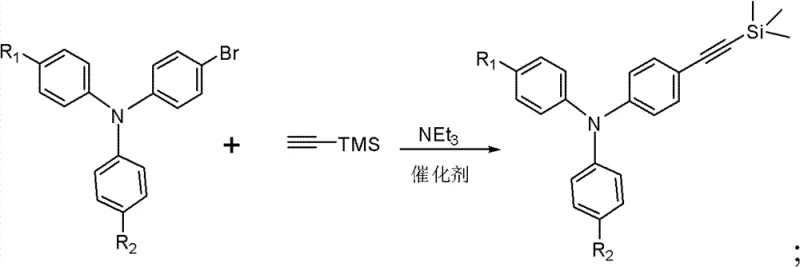
Mechanistically, the synthesis relies on a robust three-step sequence that balances reactivity with functional group tolerance. The initial step involves a palladium-catalyzed Sonogashira cross-coupling reaction, where a brominated triphenylamine precursor reacts with trimethylsilylacetylene. This reaction is typically conducted in triethylamine under an inert atmosphere, utilizing a catalyst system such as Pd(PPh3)2Cl2 or Pd(PPh3)4 combined with CuI. The use of the trimethylsilyl (TMS) protecting group is critical here, as it prevents polymerization of the acetylene moiety and allows for precise control over the subsequent cyclization step. Following the coupling, a mild deprotection step using NaOH in a THF/methanol mixture reveals the terminal alkyne. The final and most critical transformation is a thermal cyclization reaction performed at elevated temperatures (170°C) in xylenes. This step fuses the acetylene-linked triphenylamine with a 7,9-diphenyl-cyclopentene naphthalene-8-one ketone, constructing the extended conjugated fluoranthene system. The rigorous purification protocols described, including column chromatography and recrystallization, ensure that the final high-purity OLED material meets the stringent impurity specifications required for commercial device fabrication, minimizing trap states that could degrade device performance.
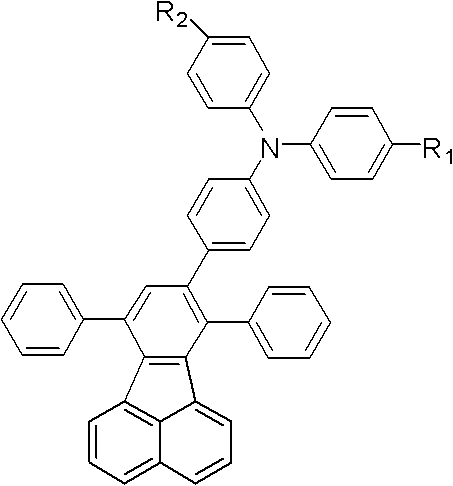
How to Synthesize Fluoranthene Derivatives Efficiently
The synthesis of these advanced fluoranthene derivatives requires precise control over reaction conditions to maximize yield and purity. The process begins with the preparation of the acetylene-functionalized triphenylamine intermediate, followed by deprotection and final cyclization. Each step demands careful monitoring of temperature, stoichiometry, and atmospheric conditions to prevent side reactions. The detailed standardized synthetic steps for producing these compounds, including specific molar ratios and workup procedures, are outlined in the guide below.
- Perform a Sonogashira coupling reaction between 4-bromo-4'-R1-4''-R2 triphenylamine and trimethylsilylacetylene using a Pd/Cu catalyst system in triethylamine.
- Deprotect the trimethylsilyl group by treating the intermediate with NaOH in a THF/Methanol mixture to generate the terminal ethynyl triphenylamine.
- Execute a thermal cyclization reaction between the ethynyl triphenylamine and 7,9-diphenyl-cyclopentene naphthalene-8-one in xylene at 170°C for 24 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible benefits beyond mere technical performance. The ability to tune solubility through simple alkyl or alkoxy substitutions means that the material can be processed using solution-based techniques or optimized for vacuum deposition with greater consistency. This flexibility reduces the risk of batch-to-batch variability, a common pain point in the sourcing of specialized organic semiconductors. Furthermore, the synthetic pathway utilizes readily available starting materials and standard catalytic systems, avoiding the need for exotic reagents that often create supply bottlenecks. This accessibility ensures a more resilient supply chain capable of meeting the fluctuating demands of the display industry.
- Cost Reduction in Manufacturing: The improved solubility of the final product significantly streamlines the purification process. Traditional rigid fluoranthene derivatives often require extensive and costly purification efforts to remove insoluble aggregates. By contrast, the substituted derivatives described in the patent can be purified more efficiently via standard column chromatography and recrystallization. This reduction in downstream processing time and solvent consumption leads to substantial cost savings. Additionally, the reported yields for the final cyclization step are consistently high, maximizing the output from expensive palladium catalysts and precious ketone precursors.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the use of diverse starting materials (R1/R2 groups) without altering the core reaction conditions. This means that if a specific alkyl bromide becomes scarce, the synthesis can be adapted to use an alternative chain length or an alkoxy variant without revalidating the entire process. Such flexibility is invaluable for maintaining commercial scale-up of complex polymer additives and small molecule emitters, ensuring that production schedules remain uninterrupted even when raw material markets fluctuate.
- Scalability and Environmental Compliance: The reaction conditions, particularly the use of triethylamine and xylenes, are well-established in industrial organic synthesis, facilitating straightforward scale-up from laboratory grams to metric ton quantities. The elimination of harsh acidic or basic conditions in the final cyclization step reduces the generation of hazardous waste streams. Moreover, the high thermal stability of the product ensures that the material can withstand the rigors of device fabrication without degradation, reducing the rate of device failure and the associated environmental cost of electronic waste.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these fluoranthene compounds. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on performance metrics and process capabilities.
Q: How does the substitution pattern affect the solubility of fluoranthene derivatives?
A: The introduction of alkyl chains (C1-C20) or alkoxy groups (C1-C12) at the R1 and R2 positions significantly disrupts molecular packing, thereby enhancing solubility in organic solvents compared to unsubstituted rigid fluoranthene cores.
Q: What is the thermal stability profile of these compounds for OLED applications?
A: The fluoranthene core provides inherent rigidity and high thermal stability, which is crucial for maintaining morphological stability in the emissive layer during device operation and vacuum deposition processes.
Q: Why is the Sonogashira coupling step critical in this synthesis?
A: This palladium-catalyzed cross-coupling efficiently links the electron-transporting triphenylamine unit with the acetylene linker, establishing the conjugated system necessary for effective hole transport and blue light emission.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoranthene Organic Compound Supplier
As the demand for high-efficiency blue OLED materials continues to surge, partnering with a manufacturer that possesses deep technical expertise is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch of fluoranthene derivative meets the exacting standards required for next-generation display technologies. We understand that the transition from lab-scale synthesis to industrial manufacturing involves complex challenges, and our team is equipped to navigate these hurdles with precision.
We invite global partners to collaborate with us to optimize their material sourcing strategies. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of reliability, quality, and innovation.
