Revolutionizing Delta-Valerolactone Production: A Safe and Scalable Catalytic Route for Global Supply Chains
Revolutionizing Delta-Valerolactone Production: A Safe and Scalable Catalytic Route for Global Supply Chains
The global demand for high-performance cyclic esters like delta-valerolactone (DVL) is surging, driven by its critical applications in biodegradable polymers, pharmaceutical intermediates, and advanced coating formulations. However, traditional manufacturing methods have long been plagued by safety hazards, high raw material costs, and complex purification requirements. A groundbreaking technical advancement detailed in patent CN109651318B introduces a transformative one-step isomerization addition cyclization process that fundamentally reshapes the production landscape. This novel methodology utilizes readily available alkyl acetates and propylene oxide as feedstocks, catalyzed by a sophisticated lithium phosphate supported on LDHs-modified alumina. For R&D directors and procurement strategists seeking a reliable fine chemical intermediates supplier, this technology represents a paradigm shift towards greener, safer, and more economically viable manufacturing protocols that eliminate the reliance on dangerous peroxides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of delta-valerolactone has been dominated by two primary pathways, both of which suffer from severe intrinsic drawbacks that hinder scalable and cost-effective production. The first method involves the Baeyer-Villiger oxidation of cyclopentanone, a process that necessitates the use of expensive organic peracids such as peroxybenzoic acid or anhydrous peracetic acid. These oxidizing agents are not only costly but also introduce significant safety liabilities due to their thermal instability and potential for catastrophic runaway reactions. Furthermore, the reaction generates stoichiometric amounts of organic acid byproducts, requiring extensive neutralization with alkaline reagents like sodium carbonate, which drastically increases waste treatment costs and environmental burden. The second conventional route involves the dehydrogenation of 1,5-pentanediol, which produces n-valeric acid as a corrosive byproduct that damages reactor equipment and complicates product separation. These legacy technologies fail to meet the modern standards for cost reduction in fine chemical intermediates manufacturing due to their high operational expenditures and safety risks.
The Novel Approach
In stark contrast, the innovative process disclosed in the patent data leverages a direct reaction between alkyl acetates and propylene oxide, offering a streamlined and inherently safer alternative. This method operates under moderate conditions, typically between 150~350°C and pressures of 0~0.5MPa, utilizing a robust heterogeneous catalyst system. The core of this breakthrough lies in the use of a lithium phosphate active component supported on alumina that has been surface-modified with Layered Double Hydroxides (LDHs). This specific catalyst architecture enables a highly efficient isomerization and cyclization sequence in a single step. By employing catalytic distillation technology, the process achieves simultaneous reaction and separation, continuously removing the low-boiling alcohol byproduct to drive the equilibrium forward. This results in exceptional conversion rates reaching 99.9% and selectivity exceeding 95.0%, ensuring that the final product requires minimal downstream purification. 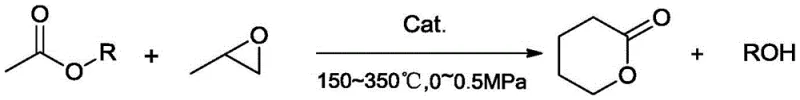
Mechanistic Insights into Li3PO4/LDHs-Al2O3 Catalyzed Cyclization
The exceptional performance of this synthesis route is rooted in the unique physicochemical properties of the LDHs-modified catalyst support. The layered double hydroxide structure imparts a highly ordered distribution of acid and base active sites on the alumina surface, which is critical for orchestrating the multi-step reaction mechanism. Initially, the basic sites on the catalyst facilitate the isomerization of propylene oxide, generating a reactive intermediate that is primed for nucleophilic attack. Subsequently, the acidic sites catalyze the transesterification and cyclization steps, effectively closing the ring to form the stable lactone structure. This synergistic cooperation between acid and base functionalities minimizes side reactions and polymerization, which are common pitfalls in lactone synthesis. The precise control over the active site distribution ensures that the reaction proceeds with high specificity, directly addressing the purity concerns of R&D Directors who require consistent quality for sensitive pharmaceutical applications.
Furthermore, the stability of the lithium phosphate active phase on the modified support ensures long catalyst life and resistance to leaching, which is essential for continuous industrial operations. The mechanism avoids the formation of corrosive carboxylic acids that plague the dehydrogenation of diols, thereby preserving the integrity of the reactor hardware. The detailed catalytic cycle illustrates how the substrate interacts with the surface species to lower the activation energy for ring closure while maintaining the structural integrity of the ester linkage. 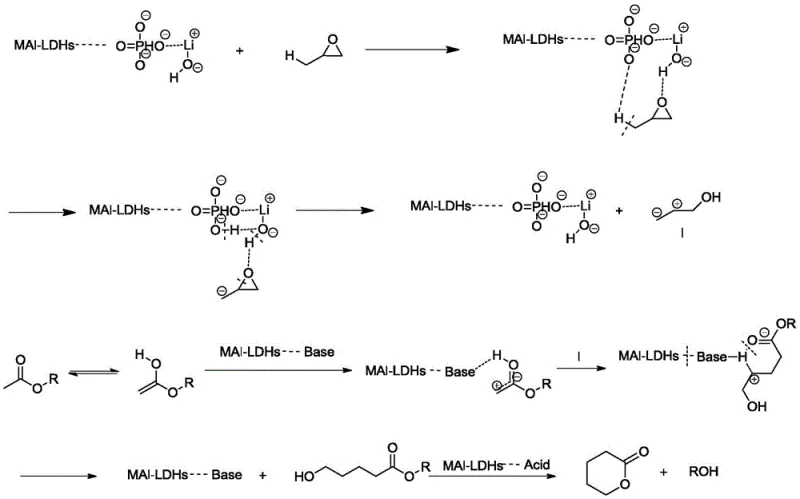
How to Synthesize Delta-Valerolactone Efficiently
Implementing this advanced synthesis route requires careful attention to catalyst preparation and reactor configuration to maximize yield and throughput. The process begins with the meticulous synthesis of the LDHs-modified alumina support, followed by the impregnation of the lithium phosphate active component via a controlled precipitation method. Once the catalyst is prepared and granulated, it is loaded into the reaction zone of a catalytic distillation column. The feedstock, consisting of alkyl acetate and propylene oxide in a molar ratio of approximately 1.01:1, is introduced continuously into the column. The system is operated at temperatures around 250°C, allowing the reaction to proceed while the volatile alcohol byproduct is distilled off from the top.
- Prepare the heterogeneous catalyst by loading lithium phosphate onto an LDHs-modified alumina support via precipitation and calcination.
- Load the catalyst into the reaction section of a catalytic distillation column equipped with rectification and stripping sections.
- Feed alkyl acetate and propylene oxide continuously into the column at 150-350°C, separating the low-boiling alcohol byproduct overhead while collecting delta-valerolactone from the bottom.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel manufacturing protocol offers substantial strategic benefits that extend far beyond simple yield improvements. The primary advantage lies in the drastic simplification of the raw material supply chain. By replacing scarce and expensive cyclopentanone or 1,5-pentanediol with commodity chemicals like ethyl acetate and propylene oxide, manufacturers can achieve significant cost reduction in manufacturing without compromising on product quality. These feedstocks are produced on a massive global scale, ensuring a stable and resilient supply line that is less susceptible to market volatility. Additionally, the elimination of hazardous peroxides removes the need for specialized storage facilities and rigorous safety protocols associated with explosive materials, thereby lowering insurance premiums and operational overheads.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the utilization of low-cost, bulk commodity feedstocks. Unlike the Baeyer-Villiger route which consumes expensive oxidizing agents and generates large volumes of salt waste requiring disposal, this catalytic method produces a clean alcohol byproduct that can often be recycled or sold as a co-product. The high selectivity of the catalyst means that raw material utilization is maximized, reducing the cost per kilogram of the final active ingredient. Furthermore, the integration of reaction and separation in a catalytic distillation column reduces capital expenditure on equipment and lowers energy consumption compared to batch processes with separate purification units.
- Enhanced Supply Chain Reliability: Sourcing raw materials for chemical production is often a bottleneck, but this method relies on alkyl acetates and propylene oxide, which are ubiquitous in the petrochemical industry. This abundance ensures that production schedules are not disrupted by raw material shortages, a common issue with niche precursors like cyclopentanone. The robustness of the heterogeneous catalyst also contributes to supply continuity, as it allows for long campaign runs without frequent shutdowns for catalyst replacement. This reliability is crucial for maintaining just-in-time delivery commitments to downstream customers in the pharmaceutical and polymer sectors.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is exceptionally clean. It avoids the generation of corrosive acidic waste streams and eliminates the risk of peroxide explosions, making it easier to obtain regulatory approvals for plant expansion. The continuous nature of the catalytic distillation process facilitates seamless scale-up from pilot plants to commercial-scale production facilities ranging from 100 kgs to 100 MT annual capacity. The reduced environmental footprint aligns with the increasing global pressure for sustainable chemical manufacturing, enhancing the brand value of companies adopting this green technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this delta-valerolactone synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities.
Q: Why is the new alkyl acetate route superior to the traditional Baeyer-Villiger oxidation?
A: The traditional Baeyer-Villiger method relies on expensive and hazardous organic peroxides, posing significant safety risks and generating acidic waste. The new route uses cheap, stable alkyl acetates and propylene oxide, eliminating peroxide hazards and simplifying waste treatment.
Q: How does the LDHs-modified catalyst improve selectivity?
A: The Layered Double Hydroxides (LDHs) modification creates a regular distribution of acid-base active sites on the alumina surface. This specific arrangement facilitates the isomerization and cyclization steps simultaneously, achieving selectivity over 95% and minimizing byproduct formation.
Q: What are the separation advantages of using catalytic distillation?
A: Catalytic distillation combines reaction and separation in one unit. It continuously removes the low-boiling alcohol byproduct (ROH) from the top, shifting the equilibrium towards the product and allowing for the continuous collection of high-purity delta-valerolactone from the bottom without complex downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Delta-Valerolactone Supplier
At NINGBO INNO PHARMCHEM, we understand that the successful commercialization of advanced chemical intermediates requires more than just a laboratory recipe; it demands engineering excellence and rigorous quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is smooth and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of catalytic distillation processes, and our stringent purity specifications guarantee that every batch of delta-valerolactone meets the exacting standards required for pharmaceutical and electronic applications. With our rigorous QC labs, we ensure that impurity profiles are tightly controlled, delivering a product that supports your downstream synthesis without failure.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this new route can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this superior delta-valerolactone into your product portfolio.
