Advanced Catalytic Synthesis of Eslicarbazepine for Scalable API Production
Advanced Catalytic Synthesis of Eslicarbazepine for Scalable API Production
The pharmaceutical industry continuously seeks more efficient pathways for the production of high-value chiral intermediates, and the synthesis of Eslicarbazepine represents a critical challenge in the manufacture of next-generation antiepileptic medications. Patent CN102250005A introduces a groundbreaking preparation method that leverages advanced asymmetric catalytic hydrogenation to convert Oxcarbazepine directly into Eslicarbazepine with exceptional stereocontrol. This technology marks a significant departure from traditional resolution techniques, offering a route that maximizes atom economy while minimizing environmental impact through the use of molecular hydrogen as a clean reducing agent. By employing a sophisticated chiral catalyst system composed of a metal salt, a chiral diphosphine ligand, and a chiral nitrogen ligand, the process achieves high optical purity without the need for cumbersome separation steps. For R&D directors and process chemists, this patent provides a robust framework for developing a commercially viable synthesis that addresses both purity specifications and cost-efficiency targets inherent in modern API manufacturing.
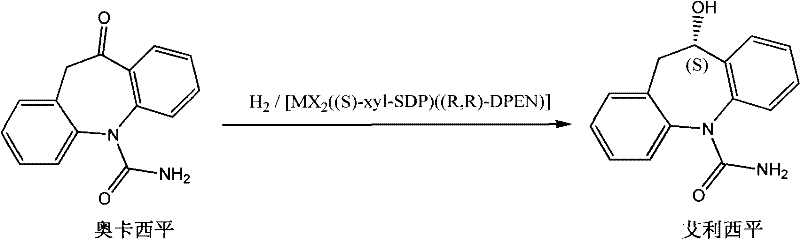
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Eslicarbazepine has been plagued by inefficiencies associated with classical resolution strategies and early transfer hydrogenation attempts. Traditional methods, such as those described in earlier patents, often involve the reduction of Oxcarbazepine to a racemic mixture followed by chiral resolution using expensive resolving agents like tartrate derivatives. This approach inherently suffers from a maximum theoretical yield of only 50%, as the undesired R-enantiomer is typically discarded or requires complex recycling loops to be useful. Furthermore, alternative methods utilizing transfer hydrogenation with formic acid and triethylamine mixtures introduce significant downstream processing burdens. These reactions generate stoichiometric amounts of amine salts as byproducts, necessitating rigorous purification protocols such as column chromatography to meet pharmaceutical grade standards. The reliance on mixed solvent systems in these older processes further complicates solvent recovery, increasing both the operational expenditure and the environmental footprint of the manufacturing process.
The Novel Approach
The methodology disclosed in CN102250005A fundamentally reengineers the synthesis by utilizing direct asymmetric hydrogenation, effectively bypassing the formation of racemic intermediates and the associated waste streams. By employing molecular hydrogen gas as the sole reducing agent, the reaction produces water as the only byproduct, eliminating the formation of difficult-to-remove amine salts entirely. This clean reaction profile allows for a simplified workup procedure where the product can be isolated through straightforward concentration and crystallization, removing the need for expensive and time-consuming chromatographic separations. Additionally, the process demonstrates remarkable flexibility regarding reaction conditions, operating effectively across a broad temperature range and utilizing common industrial solvents like ethanol or isopropanol. This shift not only enhances the overall yield by preserving the stereochemistry from the start but also aligns with green chemistry principles, making it an attractive option for sustainable pharmaceutical manufacturing.
Mechanistic Insights into Chiral Ruthenium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the precise design of the chiral catalyst system, specifically the complex denoted as [MX2((S)-xyl-SDP)((R,R)-DPEN)]. This catalyst operates through a synergistic interaction between the transition metal center, typically Ruthenium, and the chiral ligands that create a highly defined steric environment around the active site. The chiral diphosphine ligand, such as (S)-xyl-SDP, coordinates with the metal to establish the primary chiral pocket, while the chiral diamine ligand, (R,R)-DPEN, participates in the activation of the hydrogen molecule and the subsequent hydride transfer to the ketone substrate. This bifunctional catalytic mechanism ensures that the hydride delivery occurs exclusively from one face of the prochiral ketone group in Oxcarbazepine, resulting in the formation of the desired (S)-alcohol configuration with high fidelity. The structural integrity of these ligands is crucial, as subtle variations in the aryl or alkyl substituents can significantly influence the electronic properties and steric bulk, thereby tuning the enantioselectivity and reaction rate.
Impurity control in this catalytic system is inherently managed by the high specificity of the hydrogenation reaction and the absence of side-reactive reagents. Unlike reduction methods using borohydrides which can lead to over-reduction or non-selective attack, the metal-ligand complex directs the reaction pathway strictly towards the carbonyl reduction. The patent data indicates that the catalyst loading can be optimized to extremely low levels relative to the substrate, which minimizes the risk of metal contamination in the final API. Furthermore, the use of single-component solvent systems facilitates efficient recycling, reducing the accumulation of trace impurities that often arise from solvent interactions in mixed systems. The robustness of the catalyst under hydrogen pressure ensures consistent performance over extended reaction times, allowing for complete conversion of the starting material and preventing the carryover of unreacted Oxcarbazepine, which is a critical quality attribute for downstream processing.
How to Synthesize Eslicarbazepine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating this laboratory-scale innovation into a reproducible manufacturing process. The procedure involves charging a pressure reactor with the Oxcarbazepine substrate and the pre-formed or in-situ generated chiral catalyst complex in a degassed alcoholic solvent. The system is then purged and pressurized with high-purity hydrogen, creating the necessary thermodynamic driving force for the reduction. Detailed standardized synthetic steps for this specific transformation are provided in the guide below.
- Charge the reactor with Oxcarbazepine substrate, the chiral catalyst complex [MX2((S)-xyl-SDP)((R,R)-DPEN)], and a degassed alcohol solvent under inert atmosphere.
- Replace the inert gas with high-purity hydrogen and pressurize the system to between 0.1 MPa and 16.9 MPa, then heat the mixture to a temperature range of 10°C to 100°C.
- Upon completion, concentrate the solvent, add water to induce crystallization, and filter the product to obtain high-purity Eslicarbazepine with excellent optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this asymmetric hydrogenation technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of stoichiometric resolving agents and the avoidance of complex salt byproducts drastically simplify the supply chain requirements, reducing the number of raw materials that need to be sourced and qualified. This simplification translates directly into reduced inventory complexity and lower working capital requirements for chemical procurement. Moreover, the ability to use molecular hydrogen, a commodity chemical, instead of specialized and costly reducing agents or resolving salts creates a more stable cost structure that is less susceptible to market volatility in niche fine chemical sectors.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic improvement in atom economy compared to resolution-based routes. By avoiding the inherent 50% yield loss associated with discarding the wrong enantiomer, the effective cost per kilogram of the active pharmaceutical ingredient is significantly lowered. Additionally, the removal of chromatographic purification steps reduces the consumption of silica gel and large volumes of elution solvents, which are major cost centers in API production. The simplified workup also shortens the batch cycle time, allowing for higher throughput in existing manufacturing facilities without the need for significant capital investment in new equipment.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial gases and common alcohol solvents enhances the resilience of the supply chain against disruptions. Unlike processes dependent on specialized chiral pool materials or custom-synthesized resolving agents which may have long lead times, the inputs for this hydrogenation process are commoditized and readily accessible from multiple global suppliers. This diversification of supply sources mitigates the risk of single-source dependency and ensures continuous production capability even during periods of market tightness. The robustness of the reaction conditions also implies a lower risk of batch failures due to sensitive reagent quality issues.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers a cleaner profile that simplifies waste management and compliance reporting. The absence of heavy metal waste streams associated with some alternative catalysts, combined with the lack of amine salt byproducts, reduces the burden on wastewater treatment facilities. The high efficiency of solvent recovery in single-solvent systems further minimizes volatile organic compound (VOC) emissions. These factors collectively support a more sustainable manufacturing operation, aligning with increasingly stringent global environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of this catalytic method over classical resolution?
A: Unlike classical resolution which theoretically limits yield to 50% due to the discard of the unwanted enantiomer, this asymmetric hydrogenation method utilizes the starting material efficiently to produce the desired S-enantiomer directly, significantly improving atom economy and reducing raw material costs.
Q: How does this process address purification challenges found in transfer hydrogenation?
A: Traditional transfer hydrogenation often relies on formic acid and amine mixtures which generate salt byproducts requiring complex separation like column chromatography. This method uses molecular hydrogen, producing no such salt byproducts, thereby simplifying the workup to a straightforward crystallization.
Q: Is the catalyst system suitable for large-scale industrial application?
A: Yes, the patent highlights the use of robust ligands and standard hydrogenation conditions compatible with industrial autoclaves. The ability to recover solvents and the high turnover potential of the catalyst system make it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eslicarbazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN102250005A for the production of high-value neurological therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our facility is equipped with state-of-the-art high-pressure hydrogenation reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of Eslicarbazepine meets the exacting standards required by global regulatory authorities.
We invite you to collaborate with us to leverage this efficient synthesis route for your supply chain needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this catalytic method can optimize your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable and cost-effective supply of this critical API intermediate.
