Advanced Catalytic Synthesis of Eslicarbazepine Intermediates for Commercial Scale-up
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing chiral active pharmaceutical ingredients (APIs), and the preparation of Eslicarbazepine intermediates stands as a prime example of this technological evolution. Patent CN110790708A introduces a groundbreaking methodology for the preparation of key Eslicarbazepine intermediates, specifically utilizing an asymmetric catalytic hydrogenation strategy that fundamentally alters the economic and technical landscape of production. This innovation moves away from the traditional reliance on stoichiometric chiral resolving agents or less efficient transfer hydrogenation systems, instead employing molecular hydrogen gas activated by sophisticated chiral iridium complexes. By leveraging chiral tridentate ligands such as phosphine-nitrogen-nitrogen (PNN) or phosphine-nitrogen-sulfur (PNS) architectures, the process achieves exceptional stereocontrol directly from the prochiral ketone precursor. For R&D directors and process chemists, this patent represents a significant leap forward in atom economy and process simplicity, offering a robust alternative to legacy methods that have long plagued the supply chain with high material costs and complex purification burdens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Eslicarbazepine has been hindered by inherent inefficiencies in chiral separation and suboptimal hydrogenation techniques. Early methods, such as those described by Portela, relied heavily on the resolution of racemic Licarbazepine using chiral acids like tartaric anhydride derivatives.  This classical resolution approach creates a fundamental ceiling on yield, as it theoretically discards 50% of the valuable starting material—the unwanted enantiomer—which must either be wasted or subjected to energy-intensive racemization and recycling loops. Furthermore, alternative catalytic routes, such as the transfer hydrogenation methods disclosed by Novartis, utilize formic acid-triethylamine mixtures as hydrogen donors. While avoiding high-pressure hydrogen equipment, these transfer hydrogenation processes generate stoichiometric amounts of amine and carbon dioxide by-products, necessitating cumbersome column chromatography for purification. These legacy processes not only inflate the cost of goods sold (COGS) due to reagent waste but also introduce significant environmental hazards through the generation of chemical waste streams that require specialized treatment before disposal.
This classical resolution approach creates a fundamental ceiling on yield, as it theoretically discards 50% of the valuable starting material—the unwanted enantiomer—which must either be wasted or subjected to energy-intensive racemization and recycling loops. Furthermore, alternative catalytic routes, such as the transfer hydrogenation methods disclosed by Novartis, utilize formic acid-triethylamine mixtures as hydrogen donors. While avoiding high-pressure hydrogen equipment, these transfer hydrogenation processes generate stoichiometric amounts of amine and carbon dioxide by-products, necessitating cumbersome column chromatography for purification. These legacy processes not only inflate the cost of goods sold (COGS) due to reagent waste but also introduce significant environmental hazards through the generation of chemical waste streams that require specialized treatment before disposal.
The Novel Approach
In stark contrast to these legacy methodologies, the technology disclosed in CN110790708A utilizes direct asymmetric hydrogenation with molecular hydrogen, eliminating the formation of stoichiometric by-products entirely. 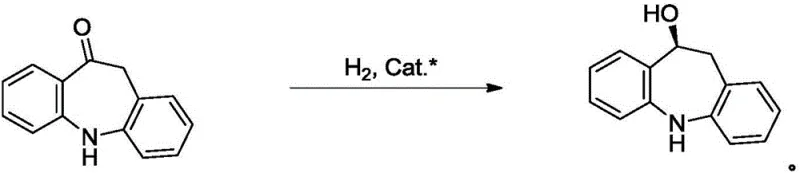 The core innovation lies in the use of specific chiral iridium catalysts bearing tridentate ligands that are uniquely tuned to recognize the subtle steric differences in the 5,11-dihydro-10H-dibenzo[b,f]azepine-10-one substrate. This direct hydrogenation pathway operates under relatively mild conditions, typically between 25°C and 50°C, and utilizes hydrogen pressures of 1.8 to 2.0 MPa, which are readily achievable in standard industrial hydrogenation reactors. By bypassing the need for chiral resolving agents and avoiding the by-product clutter of transfer hydrogenation, this novel approach streamlines the downstream processing workflow. The result is a cleaner reaction profile that facilitates simpler isolation procedures, such as concentration and crystallization, rather than resource-intensive chromatographic separations, thereby drastically reducing the operational complexity and solvent consumption associated with large-scale manufacturing.
The core innovation lies in the use of specific chiral iridium catalysts bearing tridentate ligands that are uniquely tuned to recognize the subtle steric differences in the 5,11-dihydro-10H-dibenzo[b,f]azepine-10-one substrate. This direct hydrogenation pathway operates under relatively mild conditions, typically between 25°C and 50°C, and utilizes hydrogen pressures of 1.8 to 2.0 MPa, which are readily achievable in standard industrial hydrogenation reactors. By bypassing the need for chiral resolving agents and avoiding the by-product clutter of transfer hydrogenation, this novel approach streamlines the downstream processing workflow. The result is a cleaner reaction profile that facilitates simpler isolation procedures, such as concentration and crystallization, rather than resource-intensive chromatographic separations, thereby drastically reducing the operational complexity and solvent consumption associated with large-scale manufacturing.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The success of this synthetic route hinges on the precise architectural design of the chiral catalyst, which dictates the stereochemical outcome of the hydrogen addition. The patent highlights the efficacy of iridium complexes coordinated with chiral tridentate ligands, such as the PNN-type ligand found in catalyst C1. 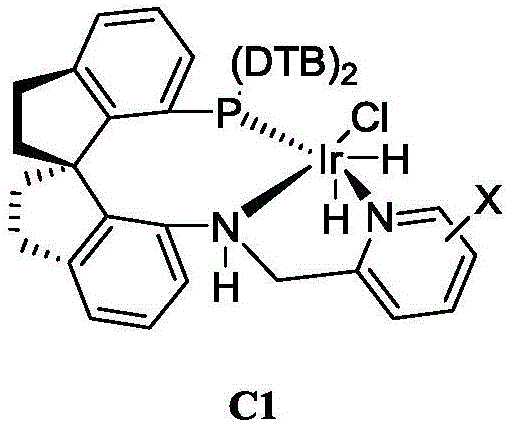 These ligands feature a rigid spiro-backbone that locks the coordination geometry around the metal center, creating a well-defined chiral pocket. When the prochiral ketone substrate coordinates to the iridium center, the bulky substituents on the ligand framework effectively block one face of the carbonyl group, forcing the hydride transfer to occur exclusively from the opposite face. This steric differentiation is particularly challenging for this specific substrate because the carbonyl group is flanked by two planar aromatic systems with minimal steric differentiation. However, the specific electronic and steric properties of the tridentate ligand system overcome this challenge, enabling high levels of enantioinduction. The mechanism likely proceeds through an outer-sphere or inner-sphere hydride transfer pathway where the amine proton on the ligand may participate in a bifunctional activation mode, stabilizing the transition state and enhancing both reaction rate and selectivity.
These ligands feature a rigid spiro-backbone that locks the coordination geometry around the metal center, creating a well-defined chiral pocket. When the prochiral ketone substrate coordinates to the iridium center, the bulky substituents on the ligand framework effectively block one face of the carbonyl group, forcing the hydride transfer to occur exclusively from the opposite face. This steric differentiation is particularly challenging for this specific substrate because the carbonyl group is flanked by two planar aromatic systems with minimal steric differentiation. However, the specific electronic and steric properties of the tridentate ligand system overcome this challenge, enabling high levels of enantioinduction. The mechanism likely proceeds through an outer-sphere or inner-sphere hydride transfer pathway where the amine proton on the ligand may participate in a bifunctional activation mode, stabilizing the transition state and enhancing both reaction rate and selectivity.
From an impurity control perspective, the mechanistic cleanliness of this catalytic cycle offers substantial advantages for pharmaceutical quality assurance. Unlike transfer hydrogenation which generates amine salts and requires extensive washing to remove trace organic bases, the direct hydrogenation method produces only the desired alcohol product and potentially trace amounts of the unreacted ketone. The high conversion rates reported in the patent examples, often exceeding 95% and reaching up to 99%, mean that the residual starting material is minimal. Furthermore, the high enantiomeric excess (ee) achieved directly from the reaction, ranging from 85% to over 99% depending on the specific ligand and solvent combination, reduces the burden on downstream purification. In cases where ultra-high optical purity is required, the process allows for a simple recrystallization step to upgrade the ee to >99.5%, ensuring that the final intermediate meets the rigorous specifications demanded by global regulatory agencies for chiral epilepsy medications without the need for chiral HPLC separation on a production scale.
How to Synthesize (S)-10,11-dihydro-5H-dibenzo[b,f]azepine-10-ol Efficiently
The practical implementation of this patented technology involves a straightforward yet highly controlled high-pressure hydrogenation procedure that is amenable to both laboratory optimization and industrial scale-up. The process begins with the careful preparation of the reaction vessel under an inert atmosphere to prevent catalyst deactivation by oxygen. Key parameters such as the choice of alcohol solvent (e.g., n-butanol, ethanol, or isopropanol) and the specific base (e.g., potassium tert-butoxide or sodium tert-butoxide) are critical for maximizing both conversion and enantioselectivity. The patent data suggests that n-butanol often provides superior results in terms of optical purity compared to shorter-chain alcohols. Once the reagents are charged, the system is pressurized with hydrogen and maintained at a constant pressure while stirring at ambient or slightly elevated temperatures. The reaction progress is monitored by hydrogen uptake, and upon completion, the workup involves simple aqueous extraction and solvent removal.
- Charge a high-pressure reactor with the ketone substrate, chiral iridium catalyst (e.g., C1 or C2), and a base such as potassium tert-butoxide under nitrogen protection.
- Add the appropriate alcohol solvent (e.g., n-butanol or ethanol) and pressurize the system with hydrogen gas to maintain a pressure between 1.8 and 2.0 MPa.
- Stir the reaction mixture at 25-50°C until hydrogen uptake ceases, then concentrate the solution and purify the crude product via extraction and recrystallization to achieve high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this asymmetric hydrogenation technology translates into tangible improvements in cost structure and supply reliability. The most significant economic driver is the elimination of the 50% material loss inherent in classical resolution processes. By converting the prochiral ketone directly into the desired (S)-enantiomer with high selectivity, the theoretical yield is doubled compared to resolution methods, effectively halving the raw material cost per kilogram of active intermediate. Additionally, the switch from expensive chiral resolving agents like acetyl tartaric anhydride to catalytic amounts of ligand significantly reduces the bill of materials. Although the chiral ligands themselves are sophisticated molecules, their usage at low catalytic loadings (often less than 1 mol%) ensures that their cost contribution is marginal compared to the savings gained from improved yield and reduced waste disposal costs.
- Cost Reduction in Manufacturing: The transition to direct asymmetric hydrogenation eliminates the need for stoichiometric chiral auxiliaries and the associated recycling loops for the unwanted enantiomer, leading to substantial cost savings in raw material procurement. Furthermore, the avoidance of column chromatography, which is required in transfer hydrogenation routes to remove amine by-products, drastically reduces solvent consumption and labor costs associated with purification. The simplified workup procedure, involving primarily extraction and crystallization, lowers the operational expenditure (OPEX) related to utility consumption and waste treatment, making the overall manufacturing process significantly more lean and cost-effective for high-volume production.
- Enhanced Supply Chain Reliability: Relying on molecular hydrogen as the reductant offers a distinct supply chain advantage over methods requiring specialized hydrogen donors like formic acid-amine azeotropes. Hydrogen is a commodity chemical available globally with a stable supply infrastructure, reducing the risk of raw material shortages that can disrupt production schedules. Moreover, the robustness of the catalyst system, which tolerates a range of alcohol solvents and bases, provides flexibility in sourcing. This flexibility ensures that manufacturing sites can adapt to local solvent availability without compromising reaction performance, thereby enhancing the resilience of the supply chain against regional logistical disruptions or price volatility in specific solvent markets.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard high-pressure hydrogenation equipment common in fine chemical manufacturing facilities, which facilitates a smooth transition from pilot plant to commercial tonnage production. From an environmental standpoint, the atom economy of using H2 gas is superior, as the only by-product is essentially none, contrasting sharply with the salt waste generated by resolution or transfer hydrogenation. This green chemistry profile aligns with increasingly stringent environmental regulations and corporate sustainability goals, reducing the carbon footprint of the manufacturing process and minimizing the liability associated with hazardous waste disposal, which is a critical factor for long-term operational licensing and community relations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology for Eslicarbazepine intermediate production. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this synthetic route.
Q: What represents the primary advantage of this iridium-catalyzed route over traditional resolution methods?
A: Unlike traditional resolution methods which inherently discard 50% of the material as the unwanted enantiomer, this asymmetric hydrogenation route theoretically allows for 100% atom economy by directly generating the desired (S)-enantiomer from the prochiral ketone, significantly reducing raw material costs.
Q: How does the catalyst loading impact the commercial viability of this process?
A: The patent demonstrates effective catalysis with low catalyst loading (e.g., 0.05 mol% in scaled examples), which is critical for commercial viability as it minimizes the cost contribution of the expensive chiral ligand and simplifies the removal of residual metal from the final pharmaceutical intermediate.
Q: Can the optical purity be further enhanced post-reaction?
A: Yes, while the reaction itself achieves high enantiomeric excess (up to 99.2% ee in optimized examples), the patent explicitly describes a recrystallization step using ethyl acetate and n-hexane that can upgrade the optical purity to >99.5% ee, ensuring compliance with stringent regulatory specifications for chiral drugs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eslicarbazepine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced catalytic technologies to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN110790708A can be successfully translated into robust manufacturing operations. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch for identity, potency, and chiral purity. Our facility is equipped with state-of-the-art high-pressure hydrogenation reactors and dedicated suites for handling sensitive catalytic reactions, allowing us to offer this specific Eslicarbazepine intermediate with the highest standards of quality and consistency required by top-tier drug manufacturers.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages of switching to this catalytic method compared to your current sourcing strategy. We encourage you to contact us to obtain specific COA data from our recent pilot batches and to request comprehensive route feasibility assessments tailored to your volume requirements. Let us collaborate to secure a sustainable and cost-efficient supply of this critical chiral building block for your antiepileptic drug formulations.
