Advanced Cyclization Technology for High-Purity Benzofuran Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance efficiency with regulatory compliance. Patent CN1200935C introduces a transformative method for preparing benzofuran and benzothiophene type compounds, which are critical scaffolds in modern drug discovery. This technology addresses the longstanding challenges associated with traditional cyclization methods by utilizing a carbonate base in a carboxylic acid anhydride medium. For R&D Directors and Procurement Managers, this represents a significant opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates. The process eliminates the need for costly phosphonium salts and harsh pyridine conditions, replacing them with more economical and environmentally benign reagents. By leveraging this intellectual property, manufacturers can achieve substantial cost reduction in fine chemical manufacturing while maintaining stringent quality standards required for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted benzofurans, such as 2-n-butyl-5-nitrobenzofuran, relied heavily on Wittig-type reactions or processes involving phosphonium salts as described in older literature like EP-A-0471609. These conventional methods typically require the preparation of 2-hydroxy-5-nitro-benzyltriphenylphosphonium bromide, which involves multiple steps and the use of triphenylphosphine. The subsequent reaction with valeryl chloride in the presence of pyridine generates significant amounts of triphenylphosphine oxide waste, complicating the purification process. Furthermore, the use of pyridine poses handling and environmental challenges due to its toxicity and odor. For supply chain heads, these factors translate into higher disposal costs and longer lead times for high-purity intermediates. The complexity of removing phosphorus-containing impurities often necessitates extensive chromatographic purification, which is not feasible for large-scale commercial production.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct cyclization strategy that bypasses the need for phosphonium intermediates entirely. The process involves cyclizing an aromatic compound bearing a specific side chain in the presence of a carbonate base, such as potassium carbonate or sodium carbonate, within a medium comprising a carboxylic acid anhydride. 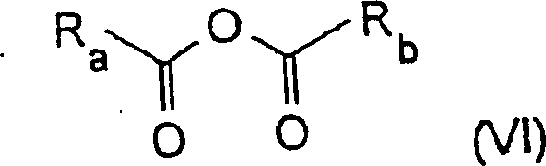 This shift in reagent strategy simplifies the reaction profile significantly. The use of anhydrides, such as acetic anhydride, serves a dual purpose as both a solvent component and a dehydrating agent that drives the cyclization equilibrium forward. This method allows for the use of common organic solvents like dimethylformamide (DMF), which facilitates product recovery and reduces the overall volume of anhydride required. For a reliable pharmaceutical intermediates supplier, this translates to a more streamlined operation with fewer unit operations, directly impacting the bottom line through reduced operational expenditure and improved throughput.
This shift in reagent strategy simplifies the reaction profile significantly. The use of anhydrides, such as acetic anhydride, serves a dual purpose as both a solvent component and a dehydrating agent that drives the cyclization equilibrium forward. This method allows for the use of common organic solvents like dimethylformamide (DMF), which facilitates product recovery and reduces the overall volume of anhydride required. For a reliable pharmaceutical intermediates supplier, this translates to a more streamlined operation with fewer unit operations, directly impacting the bottom line through reduced operational expenditure and improved throughput.
Mechanistic Insights into Carbonate-Promoted Cyclization
The core of this technological advancement lies in the mechanistic efficiency of the carbonate-promoted cyclization. The reaction targets aromatic compounds with a side chain containing at least two carbon atoms, where one carbon is linked to the benzene ring via an oxygen or sulfur atom, and the other exists in a carboxylic form. 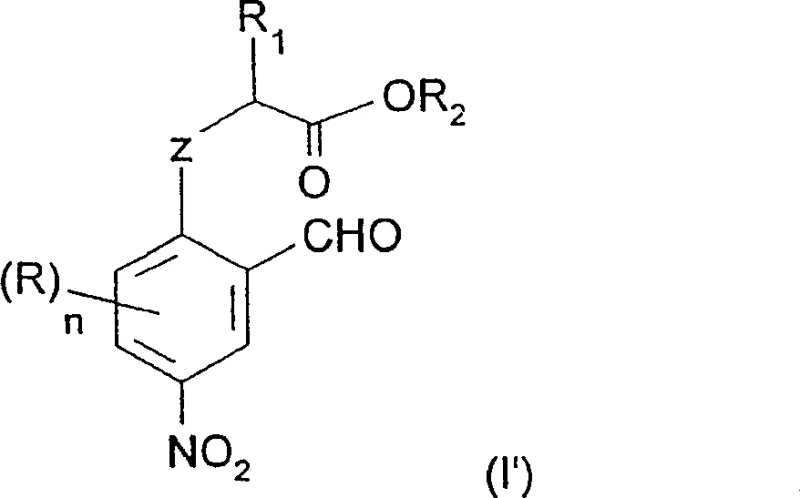 As shown in the structure above, the presence of a formyl radical in the ortho position relative to the side chain is crucial for the ring closure. The carbonate base acts as a mild yet effective catalyst that deprotonates the carboxylic acid moiety, generating a carboxylate anion. This anion then attacks the electrophilic center activated by the anhydride, facilitating the formation of the five-membered heterocyclic ring. Unlike strong bases that might degrade sensitive functional groups like the nitro group, carbonate bases offer a controlled basicity that preserves the integrity of the molecule. This selectivity is paramount for R&D teams focusing on impurity control, as it minimizes the formation of by-products that are difficult to separate.
As shown in the structure above, the presence of a formyl radical in the ortho position relative to the side chain is crucial for the ring closure. The carbonate base acts as a mild yet effective catalyst that deprotonates the carboxylic acid moiety, generating a carboxylate anion. This anion then attacks the electrophilic center activated by the anhydride, facilitating the formation of the five-membered heterocyclic ring. Unlike strong bases that might degrade sensitive functional groups like the nitro group, carbonate bases offer a controlled basicity that preserves the integrity of the molecule. This selectivity is paramount for R&D teams focusing on impurity control, as it minimizes the formation of by-products that are difficult to separate.
Furthermore, the reaction conditions are optimized to ensure high conversion rates without compromising safety. The cyclization is typically conducted at temperatures ranging from 100°C to 140°C, which is accessible using standard heating equipment in most chemical plants. The use of an inert atmosphere, such as nitrogen or argon, prevents oxidative degradation of the reactants. The mechanism also allows for flexibility in the choice of the anhydride, with acetic anhydride being the preferred embodiment due to its availability and reactivity profile. This mechanistic robustness ensures that the process can be transferred from the laboratory to the pilot plant with minimal re-optimization. For technical teams, understanding this mechanism provides confidence in the scalability of the route, ensuring that the commercial scale-up of complex heterocyclic compounds can be achieved with consistent quality.
How to Synthesize 2-n-butyl-5-nitrobenzofuran Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and addition order of reagents to maximize yield and purity. The process begins with the preparation of the nitro-aromatic precursor, which can be obtained through selective nitration of the corresponding O-alkylated compound. Once the substrate is ready, it is introduced into the reactor containing the anhydride and solvent mixture. The detailed standardized synthesis steps see the guide below, which outlines the precise temperatures and mixing rates required for optimal performance. This section is designed to assist process engineers in setting up the reaction parameters correctly. Following the reaction, the workup involves simple extraction and washing steps, avoiding the need for complex distillation columns or crystallization trains that are often required for phosphorus-containing by-products. This simplicity is a key driver for operational efficiency.
- Prepare the reaction medium by mixing the aromatic substrate with a carboxylic anhydride and an organic solvent such as DMF.
- Add a carbonate base, preferably potassium carbonate, to the mixture under an inert nitrogen atmosphere.
- Heat the reaction mixture to reflux temperatures between 100°C and 140°C for several hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this cyclization technology offers profound benefits for procurement and supply chain management. The elimination of expensive phosphonium salts and the reduction in solvent usage directly contribute to cost reduction in manufacturing. By simplifying the purification process, manufacturers can reduce the time required for quality control testing and release, thereby enhancing supply chain reliability. The use of common reagents like potassium carbonate and acetic anhydride ensures that raw material sourcing is stable and not subject to the volatility associated with specialized reagents. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The primary economic advantage stems from the replacement of high-cost reagents with commodity chemicals. Traditional methods rely on triphenylphosphine and acyl chlorides, which are significantly more expensive than carbonates and anhydrides. Additionally, the waste stream is much cleaner, reducing the costs associated with hazardous waste disposal and treatment. The simplified workup procedure means less labor and energy are consumed during the isolation of the final product. These factors combine to lower the overall cost of goods sold, allowing for more competitive pricing in the market without sacrificing margin. This economic efficiency is vital for sustaining long-term partnerships in the competitive fine chemical sector.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available raw materials. Potassium carbonate and acetic anhydride are produced in large volumes globally, reducing the risk of supply disruptions that can occur with niche reagents. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain. For supply chain heads, this reliability translates into predictable lead times and the ability to plan inventory more effectively. Reducing lead time for high-purity intermediates becomes achievable when the production process is not bottlenecked by complex purification steps or scarce reagents.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations and mild reaction conditions. The absence of toxic pyridine and phosphorus waste simplifies environmental compliance, making it easier to obtain necessary permits for production expansion. The reduced environmental footprint aligns with the increasing demand for green chemistry solutions from end-users. Scalability is further supported by the ability to use continuous flow chemistry or large batch reactors without significant modification. This adaptability ensures that production capacity can be ramped up quickly to meet surges in demand, providing a strategic advantage in a dynamic market environment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific technical advantages and operational parameters detailed in the patent documentation. Understanding these aspects helps stakeholders make informed decisions about integrating this route into their manufacturing portfolios. The answers reflect the practical implications of the chemical data provided, focusing on yield, purity, and operational feasibility. This transparency is essential for building trust between suppliers and their technical partners.
Q: What are the advantages of using carbonate bases over phosphonium salts in benzofuran synthesis?
A: Using carbonate bases eliminates the need for expensive phosphonium reagents and pyridine, simplifying the workup process and reducing hazardous waste generation significantly.
Q: Can this cyclization method be scaled for commercial production?
A: Yes, the process operates at atmospheric pressure with standard heating and uses common solvents, making it highly suitable for commercial scale-up of complex heterocyclic compounds.
Q: What purity levels can be achieved with this novel cyclization route?
A: The method yields products with high NMR purity, often exceeding 95%, without requiring complex chromatographic purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-n-butyl-5-nitrobenzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the production of high-value pharmaceutical intermediates. Our technical team has extensively evaluated the cyclization method described in CN1200935C and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-n-butyl-5-nitrobenzofuran meets the highest industry standards. We are committed to delivering consistent quality that supports your drug development and commercialization goals.
We invite you to collaborate with us to optimize your supply chain for benzofuran derivatives. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the tangible benefits of this technology. Contact us today to discuss how we can support your project with reliable, cost-effective, and high-quality chemical solutions. Let us help you accelerate your timeline to market with our proven manufacturing expertise.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
