Advanced Benzofuran Cyclization: Scaling High-Purity Intermediates for Global Pharma
Introduction to Novel Benzofuran Manufacturing Technologies
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly benzofuran and benzothiophene derivatives which serve as critical building blocks for numerous active pharmaceutical ingredients. Patent CN1382133A introduces a transformative methodology for preparing these compounds through the cyclization of specific aromatic precursors. Unlike traditional approaches that rely on harsh conditions or expensive reagents, this invention utilizes a robust system comprising a carbonate base and a carboxylic acid anhydride medium. This technical breakthrough addresses long-standing challenges in impurity control and process scalability, offering a streamlined route to high-value intermediates such as 2-n-butyl-5-nitrobenzofuran. By leveraging simple inorganic bases like potassium carbonate alongside ubiquitous reagents like acetic anhydride, the process significantly lowers the barrier to entry for high-purity production.
The core innovation lies in the ability to cyclize aromatic compounds bearing a side chain with at least two carbon atoms, where one carbon is linked via oxygen or sulfur and the other exists as a carboxylic acid, all while maintaining an ortho-formyl group. This specific structural arrangement allows for an intramolecular condensation that is both chemically selective and operationally simple. The patent data highlights that this method is not merely a laboratory curiosity but a viable industrial process capable of delivering excellent yields without the need for transition metal catalysts or exotic ligands. For R&D directors and process chemists, this represents a significant opportunity to optimize existing synthetic routes, reduce waste streams, and improve the overall economic profile of benzofuran-based drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted benzofurans, particularly those with nitro groups essential for further functionalization, has relied heavily on Wittig-type reactions or similar phosphorus-mediated couplings. As noted in the background art of the patent, specifically referencing European patent EP-A-0471609, the preparation of n-butyl-2-nitro-5-benzofuran traditionally involves the reaction of 2-hydroxy-5-nitro-benzyltriphenylphosphonium bromide with valeryl chloride in the presence of pyridine. This conventional pathway presents several distinct disadvantages for large-scale manufacturing. Firstly, the preparation of the phosphonium salt itself requires stoichiometric amounts of triphenylphosphine, a relatively expensive reagent that contributes significantly to the raw material cost. Secondly, the reaction generates triphenylphosphine oxide as a byproduct, which is notoriously difficult to remove from the final product mixture, often requiring extensive chromatography or recrystallization steps that lower overall throughput.
Furthermore, the use of pyridine as a solvent or base introduces toxicity concerns and complicates waste management due to its strong odor and environmental impact. The multi-step nature of generating the phosphonium salt prior to the coupling reaction also extends the production timeline, increasing the risk of yield loss at each stage. From a supply chain perspective, reliance on specialized phosphorus reagents can introduce volatility, as these materials are subject to different market dynamics compared to commodity chemicals. Consequently, manufacturers utilizing these legacy methods often face higher cost-of-goods-sold (COGS) and greater regulatory scrutiny regarding residual impurities, making the search for alternative cyclization strategies a priority for modern process development teams.
The Novel Approach
In stark contrast to the phosphorus-heavy legacy routes, the method disclosed in CN1382133A offers a direct and atom-economical cyclization strategy. The process centers on treating the aromatic precursor directly with a carboxylic acid anhydride in the presence of a mild carbonate base. This approach fundamentally simplifies the reaction manifold by eliminating the need for pre-activation of the side chain into a phosphonium salt. The use of carboxylic anhydrides, such as acetic anhydride, serves a dual purpose: it acts as a dehydrating agent to facilitate the ring closure and potentially activates the carboxylic acid moiety towards nucleophilic attack by the phenolic oxygen. The selection of carbonate bases, specifically alkali metal carbonates like sodium carbonate or potassium carbonate, provides a sufficiently basic environment to deprotonate the phenol without promoting unwanted side reactions that stronger bases might induce.
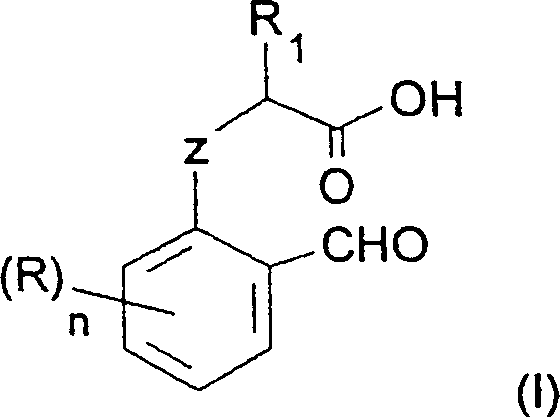
This novel methodology allows for the direct conversion of readily available starting materials into the target benzofuran core with high efficiency. The patent specifies that the reaction can be conducted in a variety of media, including neat anhydride or in the presence of organic solvents like dimethylformamide (DMF), which facilitates heat transfer and product recovery. The operational simplicity is further enhanced by the fact that the workup procedure involves standard techniques such as distillation to remove excess anhydride and solvent, followed by extraction and washing. This eliminates the complex purification burdens associated with phosphine oxide removal. By shifting to this carbonate-anhydride system, manufacturers can achieve a drastic reduction in process complexity, leading to a more robust and reliable supply of critical heterocyclic intermediates for the pharmaceutical sector.
Mechanistic Insights into Carbonate-Mediated Cyclization
The mechanistic pathway of this transformation is driven by the synergistic interaction between the carbonate base and the carboxylic acid anhydride. Initially, the carbonate base deprotonates the phenolic hydroxyl group of the starting aromatic compound, generating a phenoxide anion which is a potent nucleophile. Simultaneously, the carboxylic acid side chain interacts with the anhydride, likely forming a mixed anhydride intermediate that is more electrophilic than the free acid. This activation lowers the energy barrier for the subsequent intramolecular nucleophilic attack. The phenoxide oxygen attacks the activated carbonyl carbon of the side chain, initiating the ring closure that forms the five-membered furan ring fused to the benzene core. The presence of the ortho-formyl group is crucial, as it stabilizes the transition state and ensures the correct regiochemistry for the cyclization, preventing polymerization or intermolecular reactions.
For the specific synthesis of nitro-substituted derivatives, such as the key intermediate for 2-n-butyl-5-nitrobenzofuran, the process involves a preceding selective nitration step that demands precise control. The patent details a method where the O-alkylated aromatic precursor is subjected to nitration using a source of NO2+ in the presence of sulfuric acid. To ensure the nitro group is installed exclusively at the desired position (typically the 4-position relative to the oxygen linkage), the reaction is conducted at low temperatures, ranging from -5°C to 10°C. This thermal control is vital to suppress dinitration or oxidation of the sensitive aldehyde functionality. The use of a sulfuric-nitric acid mixture allows for the generation of the nitronium ion in a controlled manner, ensuring high regioselectivity.
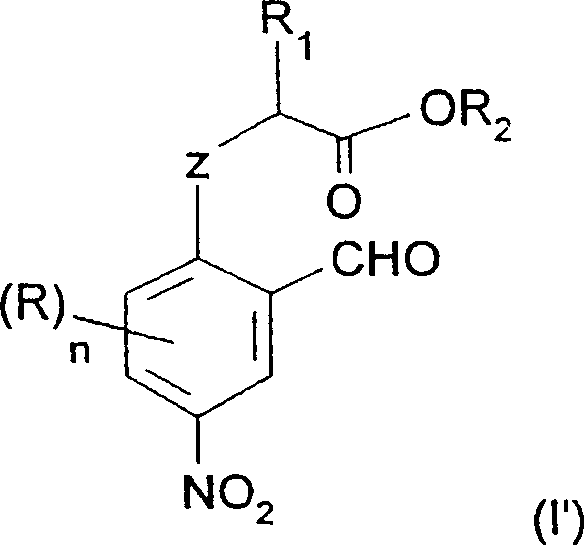
Following the nitration, the resulting nitro-aromatic acid is subjected to the cyclization conditions described above. The mechanism here remains consistent, but the electron-withdrawing nature of the nitro group enhances the acidity of the phenol, potentially facilitating the initial deprotonation step. However, the nitro group also deactivates the ring towards electrophilic attack, which is beneficial in preventing unwanted side reactions during the heating phase. The combination of selective low-temperature nitration followed by moderate-temperature cyclization (100°C to 140°C) creates a orthogonal set of conditions that maximize purity. This mechanistic understanding allows process chemists to fine-tune reaction parameters, such as base equivalents and solvent polarity, to minimize the formation of des-nitro impurities or ring-opened byproducts, ensuring the final API intermediate meets stringent quality specifications.
How to Synthesize 2-n-butyl-5-nitrobenzofuran Efficiently
The synthesis of 2-n-butyl-5-nitrobenzofuran serves as a prime example of the utility of this patented technology, demonstrating how complex heterocycles can be assembled from simple commodity chemicals. The process begins with the preparation of the nitrated precursor, 2-(2-formyl-4-nitro-phenoxy)hexanoic acid, which is obtained through the selective nitration of the corresponding phenoxy-acetic acid derivative. Once this key intermediate is secured, the cyclization is performed by suspending the acid in a medium containing acetic anhydride and a catalytic amount of potassium carbonate. The mixture is then heated to reflux, typically around 130°C to 140°C, under an inert nitrogen atmosphere to prevent oxidation of the aldehyde or the newly formed furan ring. Detailed standardized synthesis steps for this transformation are provided in the guide below.
- Prepare the aromatic substrate containing a side chain with a carboxylic acid group and an ortho-formyl group, ensuring the presence of oxygen or sulfur linkage.
- Mix the substrate with a carboxylic acid anhydride (e.g., acetic anhydride) and an organic solvent like DMF in a reactor under inert atmosphere.
- Add a carbonate base such as potassium carbonate and heat the mixture to 100-140°C to effect cyclization, followed by extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1382133A offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the radical simplification of the raw material portfolio. By replacing expensive and specialized phosphonium salts with commodity chemicals like acetic anhydride and potassium carbonate, manufacturers can significantly reduce their exposure to price volatility in the fine chemical market. These inorganic bases and anhydrides are produced on a massive global scale, ensuring a stable and continuous supply even during periods of market disruption. This shift from specialty reagents to bulk commodities directly translates to a more predictable cost structure and improved margin protection for the final pharmaceutical intermediate.
Furthermore, the elimination of phosphorus-based waste streams simplifies environmental compliance and waste disposal logistics. Triphenylphosphine oxide is a solid waste that requires specific treatment protocols, whereas the byproducts of the carbonate-anhydride method are primarily carboxylic acids and salts that are easier to handle and treat in standard effluent plants. This reduction in hazardous waste volume not only lowers disposal costs but also aligns with increasingly strict green chemistry mandates imposed by regulatory bodies and corporate sustainability goals. The simplified workup procedure, which relies on distillation and liquid-liquid extraction rather than chromatography, also enhances the throughput capacity of existing manufacturing facilities, allowing for faster batch turnover and reduced lead times for customers.
- Cost Reduction in Manufacturing: The replacement of costly phosphorus reagents with inexpensive carbonate bases and acetic anhydride drives down the direct material cost per kilogram of product. Additionally, the simplified purification process reduces solvent consumption and energy usage associated with extensive recrystallization or chromatographic separation. This leaner manufacturing approach results in substantial cost savings that can be passed down the supply chain or retained as improved margin.
- Enhanced Supply Chain Reliability: Sourcing risks are minimized because the key reagents are widely available from multiple global suppliers, reducing dependency on single-source vendors for specialized catalysts. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent production schedules and reliable delivery performance for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-gram to kilogram scales in the patent examples without loss of efficiency. The use of standard unit operations like distillation and extraction facilitates easy technology transfer from pilot plant to commercial production. Moreover, the reduced generation of hazardous phosphorus waste simplifies regulatory reporting and environmental permitting, accelerating the timeline for commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzofuran synthesis technology. These answers are derived directly from the experimental data and claims within patent CN1382133A, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this process within their existing manufacturing frameworks and quality systems.
Q: What are the advantages of the carbonate-based cyclization method over traditional Wittig reactions?
A: The carbonate-based method eliminates the need for expensive phosphonium salts and avoids the generation of triphenylphosphine oxide waste, significantly simplifying purification and reducing raw material costs.
Q: How is regioselectivity controlled during the nitration of the benzofuran precursor?
A: Regioselectivity is achieved by conducting the nitration at low temperatures (-5°C to 10°C) using a sulfuric-nitric acid mixture, which directs the nitro group specifically to the 4-position of the phenoxy-acetic acid derivative.
Q: Can this process be scaled for commercial production of API intermediates?
A: Yes, the process utilizes common industrial reagents like acetic anhydride and potassium carbonate, and employs standard workup techniques like distillation and extraction, making it highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-n-butyl-5-nitrobenzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medicines. Our technical team has extensively analyzed the methodology presented in CN1382133A and possesses the expertise to implement this carbonate-mediated cyclization at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to commercial supply is seamless. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of benzofuran intermediate meets the exacting standards required by global pharmaceutical regulators.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized process can enhance your supply chain resilience and reduce overall manufacturing costs. Let us be your trusted partner in delivering high-quality heterocyclic intermediates for the global market.
