Optimizing Apixaban Production: A Novel Synthetic Route for High-Purity Pharmaceutical Intermediates
Introduction to Advanced Apixaban Manufacturing
The global demand for direct oral anticoagulants continues to surge, placing immense pressure on supply chains to deliver high-purity active pharmaceutical ingredients (APIs) efficiently. Patent CN103694237A introduces a transformative approach to the synthesis of Apixaban, addressing critical bottlenecks in traditional manufacturing. This proprietary methodology shifts the paradigm from complex, low-yield pathways to a rational, step-economic design that prioritizes industrial feasibility. By leveraging a robust nitro-reduction strategy and optimized cyclization sequences, this technology offers a reliable pharmaceutical intermediate supplier solution that drastically simplifies the production landscape. The core innovation lies in the strategic construction of the piperidone scaffold prior to pyrazole ring formation, effectively bypassing the stability issues that plague earlier generations of synthetic routes.
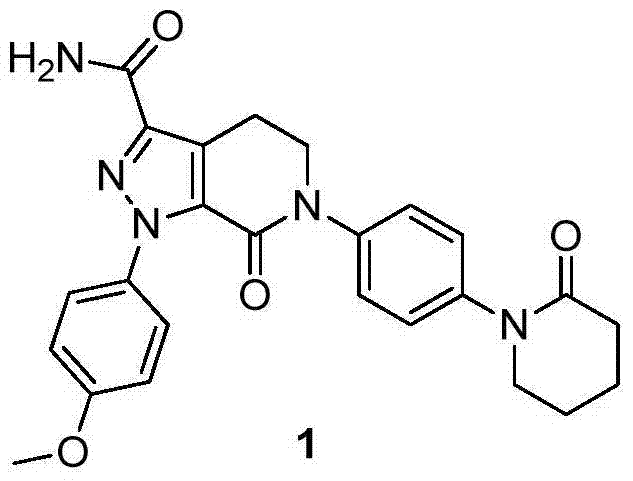
This technical breakthrough is not merely an academic exercise but a commercially viable pathway designed for commercial scale-up of complex pharmaceutical intermediates. The process begins with readily available starting materials, circumventing the reliance on scarce or prohibitively expensive halogenated aromatics. For procurement leaders, this translates to a more resilient supply chain less susceptible to raw material volatility. Furthermore, the enhanced selectivity of the reaction steps ensures that the final product meets stringent purity specifications with minimal downstream processing. As we delve deeper into the mechanistic advantages, it becomes clear that this route represents a significant leap forward in cost reduction in pharmaceutical manufacturing, offering a sustainable alternative for large-scale production facilities worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Apixaban has been hindered by inefficient route designs that compromise both yield and operational safety. Prominent prior art, such as the methodology described in patent WO03026652, relies heavily on p-iodoaniline derivatives as starting materials. This dependency introduces substantial cost burdens due to the high price of iodinated precursors compared to their chlorinated or nitrated counterparts. Moreover, the critical Ullmann coupling reaction required in these traditional routes demands harsh conditions, often involving elevated temperatures and stoichiometric amounts of copper catalysts. These conditions frequently lead to the formation of complex impurity profiles, necessitating rigorous and costly purification protocols that erode overall process efficiency. Additionally, the total yield of such legacy processes often hovers around single-digit percentages, rendering them economically unviable for competitive generic manufacturing.
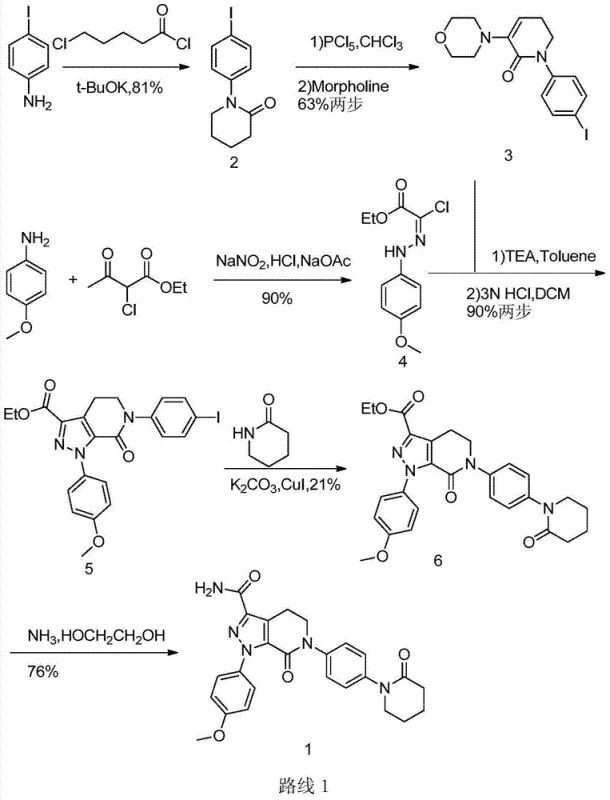
Another significant drawback in existing literature, exemplified by route CN101967145, involves the premature formation of sensitive enamine structures. These intermediates exhibit poor stability under acidic or strongly basic conditions, severely restricting the choice of reagents for subsequent transformations. The necessity to maintain neutral to weakly alkaline environments throughout multiple steps limits process flexibility and often results in incomplete reactions or side-product formation. Furthermore, the use of basic sulfides for nitro reduction in these older methods can be inconsistent, leading to variable quality and potential safety hazards due to the generation of toxic by-products. Collectively, these limitations create a fragile manufacturing process that struggles to meet the rigorous demands of modern high-purity pharmaceutical intermediate production.
The Novel Approach
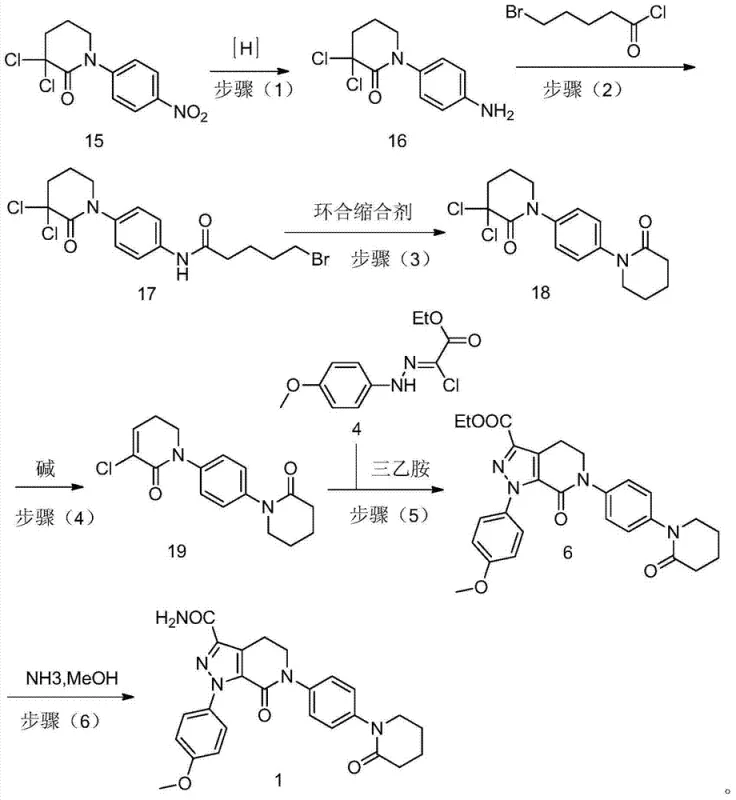
In stark contrast, the novel methodology outlined in CN103694237A re-engineers the synthetic sequence to maximize stability and yield. By initiating the synthesis with N-(4-nitrophenyl)-3,3-dichloro-2-oxopiperidine, the process establishes the core piperidone ring early, avoiding the instability associated with enamine intermediates in later stages. This strategic reordering allows for the use of robust reagents and standard operating conditions that are easily controlled in a multi-ton reactor environment. The elimination of expensive iodine-based reagents and the replacement of toxic triethyl orthoformate with safer alternatives significantly lowers the raw material cost basis. This approach not only enhances the economic viability of the project but also aligns with green chemistry principles by reducing the environmental footprint of the manufacturing process.
The superiority of this new route is further evidenced by its impressive total yield, which reaches approximately 37.65% based on p-nitroaniline, a substantial improvement over the 7.33% observed in older iodine-dependent routes. The reaction conditions are meticulously optimized to minimize by-product formation, facilitating easier isolation and purification of the target compounds. For supply chain managers, this reliability means shorter lead times and a consistent supply of critical intermediates. The process is inherently scalable, utilizing common solvents and reagents that are readily available in the global chemical market. This ensures that reducing lead time for high-purity pharmaceutical intermediates is not just a theoretical benefit but a tangible operational reality for manufacturers adopting this technology.
Mechanistic Insights into Nitro Reduction and Cyclization
The cornerstone of this innovative synthesis is the efficient reduction of the nitro group in Compound 15 to the corresponding aniline derivative, Compound 16. This transformation is achieved using mild reducing agents such as tin(II) chloride dihydrate (SnCl2·2H2O), sodium dithionite (Na2S2O4), or zinc powder. Unlike catalytic hydrogenation which may require specialized high-pressure equipment, these chemical reduction methods offer flexibility and can be performed under reflux conditions in common solvents like methanol or DMF. The choice of reducing agent allows for fine-tuning the reaction kinetics to ensure complete conversion while preventing over-reduction or degradation of the sensitive piperidone ring. This step is crucial as it generates the nucleophilic amine required for the subsequent acylation, setting the stage for the construction of the second ring system.
Following the reduction, the process employs a tandem acylation-cyclization sequence to form the second piperidone ring. Compound 16 reacts with 5-bromopentanoyl chloride in the presence of a base and a catalyst like DMAP to form the amide intermediate Compound 17. Subsequent treatment with a strong base, such as lithium tert-butoxide or potassium hydroxide, induces an intramolecular nucleophilic substitution, closing the ring to yield Compound 18. This cyclization is highly regioselective and proceeds with high efficiency, minimizing the formation of oligomeric by-products. The final elimination step, converting Compound 18 to the enamine Compound 19 using lithium carbonate at 105°C-110°C, is particularly noteworthy for its mildness compared to traditional dehydrohalogenation methods. This careful control of reaction conditions preserves the integrity of the molecule, ensuring that the final coupling with Compound 4 proceeds smoothly to form the pyrazolo-pyridine core with exceptional purity.
How to Synthesize Apixaban Efficiently
The synthesis of Apixaban via this patented route involves a logical six-step sequence that transforms simple starting materials into the complex final API. The process begins with the preparation of the key nitro-piperidone intermediate, followed by reduction, acylation, and cyclization to build the molecular scaffold. The final stages involve the construction of the pyrazole ring and subsequent ammonolysis to install the primary amide functionality. Each step has been optimized for maximum yield and minimal waste generation, making it an ideal candidate for technology transfer. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are provided in the guide below to assist R&D teams in replicating this high-efficiency process.
- Reduction of N-(4-nitrophenyl)-3,3-dichloro-2-oxopiperidine (Compound 15) using SnCl2 or Zn powder to obtain the amine intermediate (Compound 16).
- Acylation of Compound 16 with 5-bromopentanoyl chloride followed by base-mediated cyclization to form the piperidone ring system (Compound 18).
- Elimination reaction using Li2CO3 to generate the enamine precursor (Compound 19), followed by condensation with Compound 4 and final ammonolysis to yield Apixaban.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain strategists, the adoption of this novel synthetic route offers compelling economic and operational benefits. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for exotic iodinated aromatics and replacing them with commodity chemicals like p-nitroaniline and 5-bromopentanoyl chloride, the cost of goods sold (COGS) is significantly reduced. This shift not only lowers the direct material costs but also mitigates the risk of supply disruptions associated with niche reagents. Furthermore, the avoidance of toxic and irritating reagents such as triethyl orthoformate reduces the regulatory burden and safety costs associated with handling hazardous materials, contributing to a safer and more compliant manufacturing environment.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis directly translates to lower operational expenditures. By achieving a total yield that is multiples higher than legacy routes, the amount of starting material required per kilogram of final product is drastically decreased. This efficiency gain reduces solvent consumption, energy usage for heating and cooling, and waste disposal costs. Additionally, the simplified purification protocols mean less time and resources are spent on chromatography or recrystallization, allowing for faster batch turnover and higher throughput in existing production facilities without the need for major capital investment.
- Enhanced Supply Chain Reliability: Dependence on a single source for critical intermediates is a major vulnerability in the pharmaceutical supply chain. This route utilizes widely available building blocks that can be sourced from multiple suppliers globally, enhancing supply security. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed batches or out-of-specification results. This reliability is crucial for maintaining continuous production schedules and meeting the Just-In-Time delivery expectations of downstream API manufacturers and finished dosage form producers.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental profile of a synthesis route becomes a key differentiator. This method generates fewer hazardous by-products and avoids the use of heavy metal catalysts often required for cross-coupling reactions. The reduced waste load simplifies effluent treatment and lowers the environmental compliance costs. Moreover, the process is designed with scalability in mind, utilizing unit operations that are standard in the fine chemical industry. This ensures that the transition from pilot scale to multi-ton commercial production is seamless, enabling rapid response to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Apixaban synthesis technology. These insights are derived directly from the patent data and practical experience in scaling similar chemical processes. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provide clarity on reaction specifics, safety considerations, and the strategic advantages of this approach over conventional methods.
Q: How does this new route improve upon traditional Apixaban synthesis methods?
A: Unlike prior art routes that rely on expensive iodobenzene derivatives or harsh Ullmann coupling conditions, this patented method utilizes cost-effective p-nitroaniline derivatives. It avoids sensitive enamine structures early in the synthesis, resulting in fewer by-products, easier purification, and a significantly improved total yield suitable for industrial scale-up.
Q: What are the critical reaction conditions for the key cyclization steps?
A: The process employs mild to moderate conditions. For instance, the elimination step to form Compound 19 utilizes Li2CO3 at 105°C-110°C, while the final pyrazole ring closure occurs in toluene with triethylamine at 95°C-110°C. These conditions minimize thermal degradation and ensure high purity of the final API intermediate.
Q: Is this synthesis route compliant with green chemistry principles?
A: Yes, the route eliminates the need for toxic and irritating reagents such as triethyl orthoformate found in other methods. By reducing the number of purification steps and avoiding heavy metal catalysts often associated with cross-coupling reactions, the process generates less hazardous waste and aligns better with modern environmental compliance standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apixaban Supplier
The technological advancements detailed in Patent CN103694237A represent a significant opportunity for optimizing the production of anticoagulant therapies. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these laboratory innovations into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this route are fully realized in practice. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Apixaban intermediate meets the highest international standards, providing our partners with unwavering confidence in product quality.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance your supply chain efficiency and profitability in the competitive anticoagulant market.
