Optimizing Apixaban Intermediate Production for Commercial Scale-Up and Cost Efficiency
The pharmaceutical industry continuously seeks robust synthetic routes for critical anticoagulant medications, and patent CN103626759A presents a significant advancement in the preparation of a key Apixaban intermediate. This specific intermediate, chemically defined as 1-(4-methoxyphenyl)-7-carbonyl-6-[4-(2-carbonylpiperidinyl)phenyl]-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylate, serves as a pivotal building block in the manufacturing of Factor Xa inhibitors. The disclosed methodology addresses long-standing challenges in yield optimization and process simplicity by shifting from traditional ethyl acetate solvent systems to a dichloromethane-based protocol. By meticulously adjusting reactant ratios and reaction conditions, this innovation achieves a substantial increase in product yield while maintaining exceptional purity levels suitable for stringent regulatory requirements. For R&D directors and procurement specialists, understanding this technological shift is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality. The strategic implementation of this synthesis route not only enhances production efficiency but also mitigates supply chain risks associated with low-yielding processes.
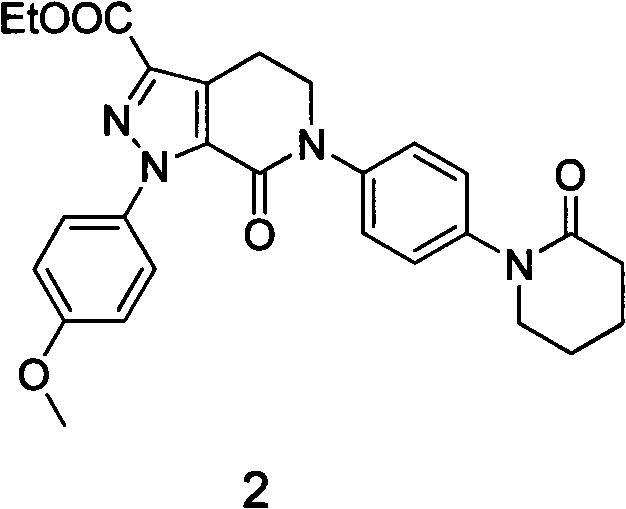
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this vital anticoagulant precursor relied heavily on ethyl acetate as the primary reaction solvent, a method documented in earlier patents such as WO03049681 and US2003181466. These conventional protocols typically employed a stoichiometric 1:1 molar ratio of reactants, which often resulted in incomplete conversion and necessitated extensive purification steps like column chromatography. The reliance on column chromatography is particularly detrimental in a commercial setting due to high operational costs, significant solvent consumption, and difficulties in scaling up for industrial production. Furthermore, the reported yields under these traditional conditions hovered around 67%, indicating a substantial loss of valuable raw materials and increased waste generation. The lack of reported purity data in older literature further complicates the assessment of material suitability for downstream API synthesis, creating uncertainty for supply chain heads who require guaranteed specifications. These inefficiencies collectively contribute to higher manufacturing costs and longer lead times, posing a barrier to cost reduction in API manufacturing.
The Novel Approach
In contrast, the novel approach detailed in the patent data introduces a transformative solvent system utilizing dichloromethane, which fundamentally alters the reaction kinetics and solubility profiles of the intermediates. By increasing the molar ratio of the hydrazine derivative to the enaminone substrate to a range of 1:1.1 to 1:3, the process drives the equilibrium towards product formation, effectively minimizing unreacted starting materials. This adjustment, combined with the use of an organic base such as triethylamine under reflux conditions, facilitates a smoother cyclization process that is less prone to side reactions. The subsequent acid treatment step is optimized to occur at controlled low temperatures, ensuring the stability of the sensitive pyrazolo-pyridine core while promoting the removal of protecting groups or byproducts. Crucially, this method replaces the cumbersome column chromatography with a straightforward recrystallization step using common solvents like ethanol, drastically simplifying the isolation procedure. This streamlined workflow not only boosts the yield to approximately 80% but also ensures a purity profile exceeding 99.5%, making it ideal for high-purity pharmaceutical intermediate demands.
Mechanistic Insights into Dichloromethane-Mediated Cyclization
The core of this synthetic breakthrough lies in the nuanced interaction between the enaminone substrate and the hydrazine derivative within the dichloromethane medium. The reaction initiates with a nucleophilic attack by the hydrazine nitrogen on the electrophilic carbon of the enaminone, facilitated by the polar aprotic nature of dichloromethane which stabilizes the transition state. Unlike ethyl acetate, dichloromethane provides a superior solvation environment for the organic base, enhancing its ability to deprotonate intermediates and drive the cyclization forward. The formation of the pyrazole ring is a critical step that determines the structural integrity of the final molecule, and the optimized conditions ensure that this ring closure occurs with high regioselectivity. The presence of the organic base also helps in scavenging the hydrochloric acid byproduct generated during the initial condensation, preventing acid-catalyzed degradation of the sensitive intermediates. This mechanistic clarity allows process chemists to fine-tune reaction parameters with confidence, knowing exactly how each variable influences the outcome.
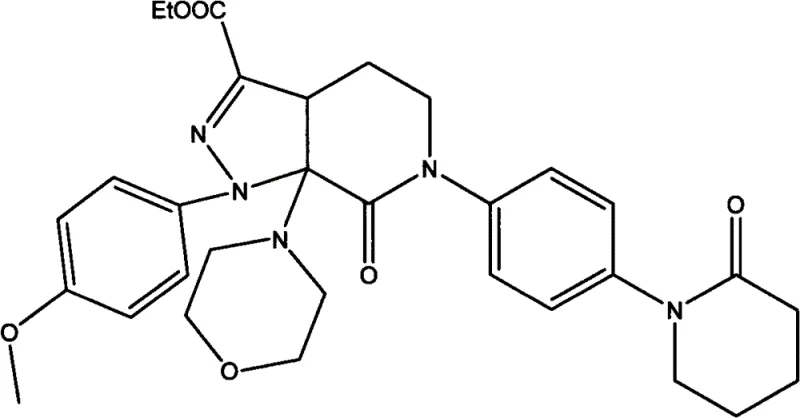
Following the initial cyclization, the reaction mixture undergoes an acid-mediated transformation that is equally critical for achieving the final product structure. The addition of inorganic acid, preferably hydrochloric acid, at temperatures between -5°C and 5°C triggers the hydrolysis and rearrangement necessary to form the stable carbonyl functionality. This low-temperature control is essential to prevent over-acidification which could lead to ring opening or decomposition of the heterocyclic system. The patent suggests that the intermediate formed after the base-catalyzed step possesses a specific configuration that is susceptible to acid-catalyzed aromatization or tautomerization. By carefully managing the acid concentration and addition rate, manufacturers can ensure that the impurity profile remains minimal, avoiding the formation of difficult-to-remove isomers. This level of control over the reaction mechanism translates directly into a more robust and reproducible manufacturing process, reducing batch-to-batch variability. For quality assurance teams, this mechanistic understanding provides the rationale behind the stringent purity specifications achievable with this route.
How to Synthesize Apixaban Intermediate Efficiently
Implementing this optimized synthesis route requires strict adherence to the specified reaction parameters to maximize yield and purity outcomes. The process begins with the precise weighing and dissolution of Compound 3 and Compound 4 in dichloromethane, ensuring that the volume-to-mass ratio aligns with the recommended 10 to 30 ml/g range. The addition of the organic base must be controlled to maintain the desired pH environment throughout the reflux period, which typically lasts between 9 to 12 hours depending on the scale. Once the initial cyclization is complete, the reaction mixture is cooled, and the inorganic acid is introduced slowly to manage the exotherm and maintain the critical temperature window. Detailed standardized synthesis steps see the guide below for exact operational procedures.
- React Compound 3 and Compound 4 in dichloromethane under reflux with an organic base such as triethylamine for 9 to 12 hours.
- Treat the reaction mixture with inorganic acid, preferably hydrochloric acid, at low temperatures between -5°C and 5°C.
- Isolate the crude product via extraction and purify through recrystallization using ethanol or acetonitrile to obtain high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound benefits for procurement managers and supply chain leaders focused on efficiency and cost containment. The elimination of column chromatography in favor of recrystallization represents a major reduction in processing time and solvent usage, directly impacting the cost of goods sold. Higher yields mean that less raw material is required to produce the same amount of finished intermediate, effectively lowering the unit cost without compromising on quality standards. This efficiency gain is particularly valuable in the volatile market of pharmaceutical raw materials, where margin pressures are constant. Furthermore, the use of readily available solvents like dichloromethane and ethanol simplifies logistics and reduces dependency on specialized reagents that might face supply disruptions. These factors combine to create a more resilient supply chain capable of meeting tight production schedules.
- Cost Reduction in Manufacturing: The shift to a higher-yielding process inherently reduces the cost per kilogram of the produced intermediate by maximizing raw material utilization. Eliminating the need for silica gel chromatography removes a significant expense category related to consumables and waste disposal, leading to substantial cost savings. The simplified workup procedure also reduces labor hours and energy consumption associated with solvent evaporation and fraction collection. Additionally, the ability to recycle mother liquors from the recrystallization step further enhances the economic viability of the process. These cumulative efficiencies allow for a more competitive pricing structure while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Utilizing common, commercially available reagents and solvents minimizes the risk of supply bottlenecks that can delay production timelines. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even with different batches of starting materials. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who depend on just-in-time delivery models. Moreover, the scalability of the recrystallization purification method ensures that production can be ramped up quickly to meet surges in demand without requiring new equipment investments. Such flexibility strengthens the partnership between the intermediate supplier and the pharmaceutical client.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, avoiding unit operations that are difficult to translate from the lab to the plant floor. Reduced solvent consumption and the absence of solid waste from chromatography columns contribute to a lower environmental footprint, aligning with modern green chemistry principles. This compliance with environmental standards reduces the regulatory burden and potential fines associated with hazardous waste management. The high purity achieved through recrystallization also minimizes the need for reprocessing, further conserving resources and energy. Overall, this approach supports sustainable manufacturing practices that are increasingly demanded by global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and sourcing of this specific anticoagulant intermediate. These answers are derived directly from the patented technology and practical manufacturing experience to provide clarity on process capabilities. Understanding these details helps stakeholders make informed decisions about integrating this material into their supply networks. The focus is on verifying the feasibility, quality, and logistical advantages of the described synthesis method.
Q: What is the primary advantage of using dichloromethane over ethyl acetate in this synthesis?
A: Using dichloromethane as the solvent significantly improves the reaction yield from approximately 67% to around 80% while simplifying the post-reaction workup process.
Q: How does the molar ratio of reactants affect the purity of the Apixaban intermediate?
A: Optimizing the molar ratio of Compound 3 to Compound 4 between 1:1.1 and 1:3 ensures complete conversion of the limiting reagent, minimizing impurities and enhancing overall product purity up to 99.5%.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method avoids complex column chromatography by utilizing recrystallization for purification, making it highly scalable and cost-effective for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apixaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced synthetic methodologies like the one described in CN103626759A to deliver superior pharmaceutical intermediates to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Apixaban intermediate meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex chemical transformations efficiently, providing you with a secure source of critical materials. By choosing us, you gain access to a partner dedicated to optimizing your supply chain through innovation and reliability.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can support your drug development goals. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to our optimized supply model. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to handle your project volumes. Let us collaborate to ensure the uninterrupted success of your anticoagulant medication production.
