Revolutionizing Flurbiprofen Manufacturing: A Deep Dive into Green Pd-Catalyzed Decarboxylative Coupling
Revolutionizing Flurbiprofen Manufacturing: A Deep Dive into Green Pd-Catalyzed Decarboxylative Coupling
In the competitive landscape of non-steroidal anti-inflammatory drug (NSAID) production, the efficiency and environmental footprint of synthetic routes are paramount. Patent CN101973869A introduces a transformative methodology for the synthesis of Flurbiprofen, a critical pharmaceutical intermediate and active pharmaceutical ingredient (API). This innovative approach leverages palladium-catalyzed decarboxylative coupling, a sophisticated organic transformation that streamlines the construction of the biphenyl backbone while simultaneously installing the requisite propionic acid side chain precursor. By integrating decarboxylation, methylation, and hydrolysis into a concise three-step sequence, this technology addresses the longstanding industry pain points of lengthy synthetic pathways and hazardous waste generation. For R&D directors and procurement strategists, understanding the mechanistic nuances and commercial implications of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity Flurbiprofen at optimized costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Flurbiprofen have historically been plagued by operational complexity and environmental inefficiencies. Conventional methods often involve multi-step sequences requiring harsh reaction conditions, expensive reagents, and the use of stoichiometric amounts of toxic heavy metals which necessitate rigorous and costly purification protocols to meet stringent residual metal specifications. Furthermore, older methodologies frequently suffer from low atom economy, generating substantial quantities of organic waste that complicate disposal and increase the overall carbon footprint of the manufacturing process. These factors collectively drive up the cost of goods sold (COGS) and introduce significant supply chain vulnerabilities, particularly when regulatory scrutiny on impurity profiles and environmental compliance intensifies. The reliance on fragile intermediates and sensitive reaction conditions in legacy processes also limits the ability to scale production rapidly to meet fluctuating market demands for this essential analgesic.
The Novel Approach
The methodology disclosed in CN101973869A represents a paradigm shift by utilizing a palladium-catalyzed decarboxylative coupling reaction as the cornerstone of the synthesis. This elegant strategy allows for the direct coupling of 4-halo-2-fluorobiphenyl with cyanoacetate derivatives, effectively merging bond formation and functional group installation in a single catalytic cycle. 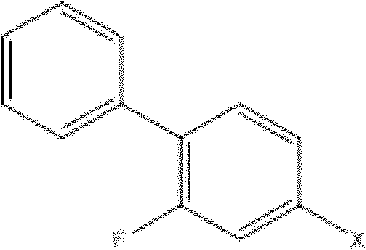
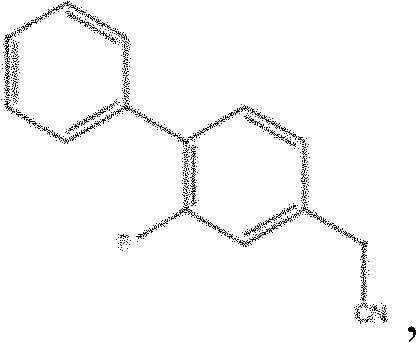 This innovation drastically simplifies the operational workflow, reducing the number of isolation steps and minimizing solvent consumption. The subsequent methylation and hydrolysis steps are performed under controlled conditions that maximize yield while maintaining a clean reaction profile. By replacing cumbersome traditional couplings with this modern catalytic approach, manufacturers can achieve a more robust and economically viable production process that aligns with the principles of green chemistry, ultimately offering a distinct competitive advantage in the global marketplace for high-purity Flurbiprofen.
This innovation drastically simplifies the operational workflow, reducing the number of isolation steps and minimizing solvent consumption. The subsequent methylation and hydrolysis steps are performed under controlled conditions that maximize yield while maintaining a clean reaction profile. By replacing cumbersome traditional couplings with this modern catalytic approach, manufacturers can achieve a more robust and economically viable production process that aligns with the principles of green chemistry, ultimately offering a distinct competitive advantage in the global marketplace for high-purity Flurbiprofen.
Mechanistic Insights into Pd-Catalyzed Decarboxylative Coupling
The core of this synthetic breakthrough lies in the intricate mechanism of the palladium-catalyzed decarboxylative coupling. The reaction initiates with the oxidative addition of the aryl halide, specifically 4-bromo-2-fluorobiphenyl or its chloro-analog, to the active palladium(0) species generated in situ from precursors such as diallylpalladium chloride. The choice of ligand is critical; bulky and electron-rich organophosphorus ligands, such as 9,9-dimethyl-4,5-bis(di-tert-butylphosphino)xanthene (Xantphos derivatives), facilitate the stabilization of the palladium center and promote the subsequent coordination of the cyanoacetate derivative.  Following coordination, the key decarboxylation event occurs, releasing carbon dioxide and generating a nucleophilic organopalladium species that undergoes reductive elimination to forge the new carbon-carbon bond. This mechanism not only drives the reaction forward thermodynamically through the entropy gain of CO2 release but also ensures high regioselectivity, minimizing the formation of homocoupling byproducts that often plague cross-coupling reactions.
Following coordination, the key decarboxylation event occurs, releasing carbon dioxide and generating a nucleophilic organopalladium species that undergoes reductive elimination to forge the new carbon-carbon bond. This mechanism not only drives the reaction forward thermodynamically through the entropy gain of CO2 release but also ensures high regioselectivity, minimizing the formation of homocoupling byproducts that often plague cross-coupling reactions.
Impurity control is inherently built into this catalytic system through the precise modulation of reaction parameters. The patent specifies a temperature range of 120°C to 160°C and a reaction time of 16 to 24 hours, conditions that are optimized to ensure complete conversion of the starting material while preventing the degradation of the sensitive nitrile intermediate. The use of specific solvents like mesitylene or dimethylformamide further enhances the solubility of reactants and stabilizes the transition states involved in the catalytic cycle. By avoiding the use of excess strong bases or aggressive nucleophiles in the initial coupling step, the process mitigates the risk of hydrolyzing the nitrile group prematurely or causing dehalogenation of the fluorobiphenyl core. This meticulous control over the reaction environment results in a crude product with a superior impurity profile, significantly reducing the burden on downstream purification units and ensuring that the final API meets the rigorous quality standards required for pharmaceutical applications.
How to Synthesize Flurbiprofen Efficiently
The practical implementation of this synthesis involves a sequential protocol designed for maximum efficiency and reproducibility. The process begins with the preparation of the nitrile intermediate via the aforementioned decarboxylative coupling, followed by a selective methylation step if a simple cyanoacetate is used, or direct hydrolysis if a methyl-substituted cyanopropionate is employed. The final step involves the hydrolysis of the nitrile group to the corresponding carboxylic acid under either acidic or basic conditions, followed by acidification to precipitate the pure Flurbiprofen. This modular approach allows for flexibility in raw material sourcing and process optimization based on specific plant capabilities. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-yielding pathway.
- Perform Pd-catalyzed decarboxylative coupling of 4-halo-2-fluorobiphenyl with cyanoacetate derivatives using organophosphorus ligands.
- Execute methylation of the resulting nitrile intermediate using methyl halides and strong base hydrides.
- Conduct final hydrolysis under acidic or basic conditions to yield Flurbiprofen, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers profound strategic benefits that extend beyond mere technical feasibility. The simplification of the manufacturing process directly translates into substantial cost savings by reducing the consumption of raw materials, solvents, and energy. The elimination of complex purification steps associated with traditional heavy metal catalysts lowers the operational expenditure related to waste treatment and quality control testing. Furthermore, the use of widely available and inexpensive starting materials, such as 4-halo-2-fluorobiphenyl and cyanoacetates, insulates the supply chain from the volatility of exotic reagent markets, ensuring a stable and continuous flow of production inputs. This resilience is crucial for maintaining uninterrupted supply to downstream API manufacturers and finished dosage form producers.
- Cost Reduction in Manufacturing: The streamlined three-step process significantly lowers the overall production cost by minimizing unit operations and maximizing atom economy. The decarboxylative coupling eliminates the need for pre-functionalized organometallic reagents, which are often expensive and unstable, replacing them with robust carboxylic acid derivatives. Additionally, the generation of only inorganic salts and carbon dioxide as byproducts simplifies effluent treatment, leading to drastic reductions in environmental compliance costs. This economic efficiency allows for more competitive pricing strategies in the global NSAID market without compromising on quality margins.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals and robust catalytic systems, this method reduces the risk of supply disruptions caused by the scarcity of specialized reagents. The tolerance of the reaction to various halogen substituents (chlorine or bromine) provides procurement teams with the flexibility to source the most cost-effective and available aryl halide at any given time. This adaptability ensures that production schedules can be maintained even when specific raw material markets experience temporary shortages, thereby enhancing the overall reliability of the supply chain for high-purity Flurbiprofen.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard reactor configurations and moderate pressure conditions. The green chemistry attributes, specifically the avoidance of toxic heavy metal waste and the production of benign byproducts, facilitate easier regulatory approval and permit acquisition in jurisdictions with strict environmental laws. This compliance readiness accelerates the time-to-market for new production lines and safeguards the manufacturer against future regulatory tightening, ensuring long-term operational sustainability and commercial viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing portfolios or sourcing strategies.
Q: What are the key advantages of the decarboxylative coupling method for Flurbiprofen?
A: This method significantly shortens the synthetic route to three steps, eliminates the need for toxic heavy metal removal processes associated with traditional catalysts, and generates only inorganic salts and CO2 as byproducts, aligning with green chemistry principles.
Q: How does this process impact impurity profiles compared to conventional routes?
A: The use of specific organophosphorus ligands and controlled reaction temperatures (120°C-160°C) minimizes side reactions, resulting in a cleaner crude product that requires less intensive purification, thereby enhancing overall yield and purity specifications.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes readily available raw materials like 4-halo-2-fluorobiphenyl and common solvents, and the reaction conditions are robust enough for scale-up from kilogram to multi-ton annual production without compromising safety or efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flurbiprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing processes in the modern pharmaceutical industry. Our team of expert chemists has extensively analyzed the potential of the Pd-catalyzed decarboxylative coupling route described in CN101973869A and is fully prepared to implement this technology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Flurbiprofen we deliver adheres to the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage these advanced synthetic capabilities for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization as your trusted partner in fine chemical manufacturing.
