Advanced Acid-Catalyzed Esterification for Commercial Scale Flurbiprofen Derivatives
The pharmaceutical industry continuously seeks robust synthetic routes for non-steroidal anti-inflammatory drug (NSAID) derivatives that balance efficacy with manufacturing safety. Patent CN103664606A introduces a transformative preparation method for flurbiprofen ester compounds, addressing critical limitations in existing technologies. This innovation utilizes a direct acid-catalyzed esterification strategy, replacing hazardous halogenated intermediates with stable, commercially available alcohols or diesters. By operating within a temperature range of 20-150°C and employing common organic solvents, the process simplifies the reaction system while maintaining high conversion rates. The methodology eliminates the need for dangerous reagents such as acetyl bromide or acetyl chloride, which are traditionally required for activating the carboxylic acid group. This shift not only enhances operator safety but also streamlines the downstream purification workflow, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking sustainable production models.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of flurbiprofen esters, particularly flurbiprofen axetil, has relied on reactive halogenated precursors that pose significant logistical and safety challenges. Traditional patents describe methods utilizing 1-bromoethanol acetate or 1-chloroethanol acetate, substances that are volatile, unstable, and difficult to store or transport over long distances. The preparation of these precursors inherently involves toxic reagents like bromine or acetyl chloride, creating a high-risk environment for production personnel and requiring specialized corrosion-resistant equipment. Furthermore, these conventional routes often necessitate complex purification techniques such as molecular distillation to remove trace impurities, driving up capital expenditure and operational costs. The instability of the halogenated intermediates can lead to inconsistent batch quality and potential decomposition during storage, complicating supply chain management for global pharmaceutical manufacturers who require consistent raw material quality.
The Novel Approach
The disclosed invention circumvents these issues by employing a direct esterification pathway using flurbiprofen, an acid catalyst, and either a stable alcohol or a diester compound. This approach leverages the nucleophilic attack of the alcohol oxygen on the carbonyl carbon of the acid, facilitated by protonation from catalysts like p-toluenesulfonic acid. By avoiding the formation of acid chlorides or the use of alkyl halides, the reaction system remains chemically stable and predictable throughout the process. The use of common solvents such as acetonitrile or toluene allows for easy recovery and recycling, further enhancing the economic viability of the method. Additionally, the workup procedure involves standard liquid-liquid extraction and washing steps, which are easily scalable in large reactors without the need for exotic processing equipment. This fundamental shift in synthetic strategy transforms a hazardous, laboratory-scale curiosity into a robust, industrial-grade manufacturing process suitable for high-purity pharmaceutical intermediate production.
Mechanistic Insights into Acid-Catalyzed Esterification
The core of this synthesis lies in the efficient protonation of the carboxylic acid group of flurbiprofen by a Brønsted acid catalyst, which increases the electrophilicity of the carbonyl carbon. In the presence of an alcohol or diester, the nucleophilic oxygen attacks this activated carbonyl, forming a tetrahedral intermediate that subsequently eliminates water or acetic acid to yield the ester product. The reaction equilibrium is driven forward by the removal of byproducts or by using an excess of the alcohol reactant, ensuring high conversion rates even at moderate temperatures ranging from 20°C to 150°C. The choice of catalyst, such as benzenesulfonic acid or sulfuric acid, is critical for minimizing side reactions while maintaining a rapid reaction rate. This mechanistic simplicity ensures that the process is forgiving to minor variations in reaction conditions, a key attribute for maintaining consistency in commercial scale-up of complex pharmaceutical intermediates.
Impurity control is meticulously managed through a multi-stage workup protocol designed to remove unreacted starting materials and acidic residues. Following the reaction, the mixture is concentrated and dissolved in ethyl acetate, followed by washing with saturated sodium bicarbonate solution to neutralize any remaining acid catalyst and unreacted flurbiprofen. This step is crucial for preventing acid-catalyzed degradation of the product during storage. Subsequent washing with water ensures the removal of inorganic salts, while drying agents like anhydrous sodium sulfate eliminate trace moisture that could hydrolyze the ester bond. Final purification via silica gel column chromatography using ethyl acetate and alkane eluents removes any remaining organic impurities, resulting in a product with purity levels often exceeding 98%. This rigorous purification sequence guarantees that the final API intermediate meets the stringent quality standards required for injectable or oral formulations.
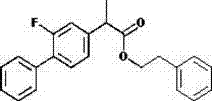
How to Synthesize Flurbiprofen Esters Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for producing various flurbiprofen ester derivatives with high reproducibility. The process begins with the precise mixing of flurbiprofen and an acid catalyst in a suitable organic solvent, followed by controlled heating and the gradual addition of the esterifying agent. This controlled addition prevents exothermic runaway and ensures uniform reaction progression. After the reaction reaches completion, indicated by TLC monitoring, the solvent is removed under reduced pressure, and the crude product is subjected to a systematic extraction and washing regime. The detailed standardized synthesis steps below outline the specific parameters for temperature, molar ratios, and purification techniques required to achieve optimal yields and purity.
- Mix flurbiprofen with an acid catalyst and organic solvent, then heat to 20-150°C before dropwise addition of esterifying agents.
- Concentrate the reaction mixture via rotary evaporation, extract with ethyl acetate and water, and separate the organic phase.
- Wash the organic phase with saturated sodium bicarbonate and water, dry, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere technical feasibility. The elimination of hazardous halogenated reagents drastically simplifies the sourcing of raw materials, as alcohols and diesters are commodity chemicals with stable global supply chains. This reduces the risk of supply disruptions caused by regulatory restrictions on toxic substances or the limited availability of specialized intermediates. Furthermore, the simplified operational requirements mean that production can be outsourced to a wider range of contract manufacturing organizations without needing specialized hazard containment facilities. The overall reduction in process complexity translates directly into lower manufacturing overheads and a more resilient supply network for critical pain management therapeutics.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous reagents like acetyl bromide with inexpensive, bulk-available alcohols significantly lowers the direct material costs associated with production. Additionally, the avoidance of complex purification steps such as molecular distillation reduces energy consumption and equipment depreciation costs. The ability to use standard glass-lined or stainless steel reactors without special corrosion protection further decreases capital investment requirements. These cumulative efficiencies result in a markedly lower cost of goods sold, allowing for more competitive pricing in the generic pharmaceutical market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: By relying on stable, non-volatile raw materials, the manufacturing process becomes far less susceptible to logistics delays and storage degradation issues. The extended shelf-life of the starting materials allows for larger batch planning and inventory buffering, ensuring continuous production even during periods of raw material scarcity. The robustness of the reaction conditions also minimizes the risk of batch failures due to minor environmental fluctuations, guaranteeing a steady flow of high-quality intermediates to downstream formulation partners. This reliability is paramount for maintaining the continuity of supply for essential analgesic medications in hospital and retail channels.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing unit operations that are standard in the fine chemical industry, facilitating a smooth transition from pilot plant to multi-ton commercial production. The reduction in toxic waste generation aligns with increasingly stringent environmental regulations, lowering the costs associated with waste disposal and environmental compliance auditing. The use of recyclable solvents and the generation of benign byproducts like acetic acid or water further enhance the sustainability profile of the manufacturing site. This eco-friendly approach not only mitigates regulatory risk but also appeals to corporate sustainability goals, strengthening the brand reputation of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this flurbiprofen ester synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios or sourcing strategies. The focus is on practical implementation details that impact cost, quality, and operational safety.
Q: What are the safety advantages of this new flurbiprofen ester synthesis method?
A: Unlike conventional methods requiring hazardous halogenated reagents like acetyl bromide or acetyl chloride, this process utilizes stable, non-toxic alcohols or diesters, significantly reducing operational risks and environmental pollution.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the method employs simple unit operations such as heating, distillation, and standard extraction, avoiding complex molecular distillation or sensitive conditions, making it highly suitable for mass production from 100 kgs to multi-ton scales.
Q: What purity levels can be achieved with this esterification technique?
A: Through optimized washing protocols using saturated sodium bicarbonate and final purification via silica gel column chromatography, the process consistently achieves high purity levels exceeding 98%, meeting stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flurbiprofen Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN103664606A are translated into reality with precision. We operate stringent purity specifications and maintain rigorous QC labs to verify that every batch of flurbiprofen ester intermediate meets the highest international standards. Our commitment to technical excellence ensures that our clients receive materials that are not only cost-effective but also fully compliant with regulatory requirements for API manufacturing.
We invite procurement leaders and R&D directors to collaborate with us to leverage these technological advancements for their product pipelines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency. Let us be your partner in delivering high-quality, safe, and affordable pharmaceutical intermediates to the patients who need them most.
