Advanced Palladium-Catalyzed Reductive Cyclization for Scalable Indole Manufacturing
Advanced Palladium-Catalyzed Reductive Cyclization for Scalable Indole Manufacturing
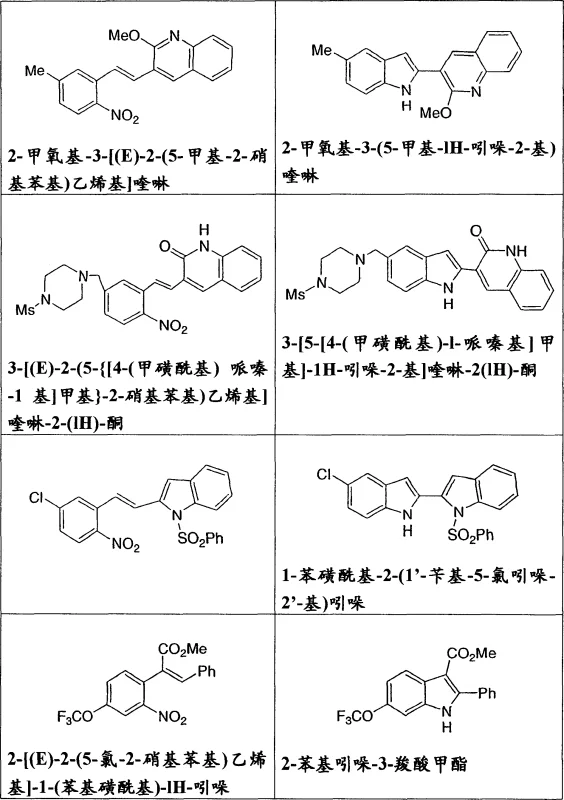
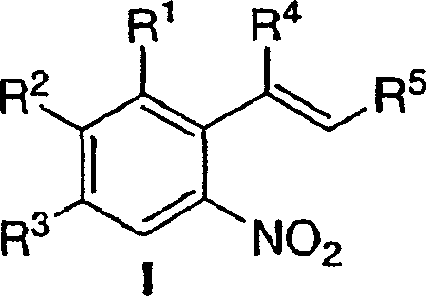
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic scaffolds, particularly substituted indoles which serve as critical pharmacophores in kinase inhibitors and hormonal regulators. Patent CN1798726A introduces a transformative methodology for the preparation of these valuable intermediates through a palladium-catalyzed reductive cyclization of ortho-nitrostyrenes. This technology represents a significant leap forward from historical precedents, offering a pathway that balances high chemical efficiency with operational simplicity. By leveraging in situ or preformed palladium catalysts in conjunction with aromatic diamine ligands, the process achieves the conversion of Formula I precursors into Formula II indole derivatives under remarkably mild conditions. For R&D directors and process chemists, this patent data provides a blueprint for accessing high-purity intermediates essential for KDR and GNRH inhibitor development, while simultaneously addressing the economic and environmental constraints faced by modern supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted indoles relied heavily on cross-coupling reactions or reductive cyclizations that imposed severe burdens on manufacturing scalability and environmental compliance. Early reports, such as those by Söderberg et al. in 1997, described palladium-catalyzed reductive cyclizations of 2-nitrostyrenes but necessitated the addition of stoichiometric amounts of tin(II) chloride (50 mol% SnCl2) to drive the reaction to completion. This reliance on tin reagents creates a massive downstream burden, requiring extensive workup procedures to remove toxic heavy metal residues from the final API intermediate. Furthermore, alternative high-pressure methods reported by Cennini et al. demanded extreme operating conditions, often exceeding 300 atmospheres of carbon monoxide pressure. Such harsh parameters not only escalate capital expenditure for specialized high-pressure equipment but also introduce significant safety risks and operational complexities that are untenable for large-scale commercial production.
The Novel Approach
The process disclosed in the present invention fundamentally disrupts these legacy limitations by enabling reductive cyclization without the need for stoichiometric tin additives or extreme pressures. By utilizing a optimized palladium catalyst system with loadings as low as 0.05 to 1.5 mol%, the reaction proceeds efficiently at moderate carbon monoxide pressures ranging from 5 to 90 psig, with specific embodiments demonstrating excellent results at merely 15 psig. This drastic reduction in pressure requirements allows the reaction to be performed in standard glass-lined or stainless steel reactors commonly available in multipurpose chemical plants, thereby removing a major barrier to entry for contract manufacturing organizations. Additionally, the substitution of phosphine ligands with aromatic diamines, such as 1,10-phenanthroline derivatives, enhances the stability of the catalytic species and simplifies the purification profile, leading to superior product quality and reduced processing time.
Mechanistic Insights into Palladium-Catalyzed Reductive Cyclization
The core of this technological advancement lies in the intricate interplay between the palladium center, the carbon monoxide reductant, and the nitro-aromatic substrate. The mechanism initiates with the coordination of the ortho-nitrostyrene to the active palladium(0) species, followed by the insertion of carbon monoxide which serves as the oxygen acceptor and reductant. Unlike traditional hydrogenation methods that might reduce other sensitive functionalities, this carbonylative pathway selectively targets the nitro group, reducing it through nitroso and hydroxylamine intermediates before triggering the intramolecular cyclization to form the indole ring. The use of aromatic diamine ligands plays a crucial role in stabilizing the palladium center against aggregation into inactive palladium black, ensuring a sustained catalytic turnover number throughout the reaction duration. This mechanistic efficiency allows for the tolerance of diverse functional groups, including alpha,beta-unsaturated ketones and aldehydes, which might otherwise be susceptible to over-reduction or polymerization under less controlled conditions.
Furthermore, the impurity profile of the resulting indole products is significantly improved due to the absence of tin byproducts and the high selectivity of the catalyst system. In conventional tin-mediated processes, the formation of organotin waste complicates the isolation of the target molecule, often requiring chromatographic purification that is impractical on a metric ton scale. In contrast, the palladium-catalyzed method described here yields products that can often be isolated via simple crystallization or filtration, as evidenced by the high purity solids obtained in the experimental examples. This cleanliness of reaction is paramount for pharmaceutical applications where strict limits on genotoxic impurities and heavy metals must be adhered to, ensuring that the resulting intermediates meet the rigorous specifications required for subsequent coupling steps in API synthesis.
How to Synthesize Substituted Indoles Efficiently
Implementing this synthesis route requires careful attention to catalyst activation and gas-liquid mass transfer to maximize yield and minimize cycle time. The process begins with the preparation of the ortho-nitrostyrene precursor, which can be synthesized via condensation of nitro-aromatics with appropriate aldehydes or ketones. Once the substrate is prepared, it is charged into a pressure vessel along with the palladium source, such as palladium(II) trifluoroacetate or acetate, and the chosen diamine ligand. The choice of solvent is also critical, with polar aprotic solvents like DMF, DMAc, or NMP proving effective for solubilizing the reactants and stabilizing the catalytic cycle. Detailed standardized operating procedures for mixing, pressurization, and isolation are essential for reproducibility.
- Prepare the reaction mixture by combining the ortho-nitrostyrene precursor (Formula I) with a palladium source (0.05-1.5 mol%) and an aromatic diamine ligand in a suitable solvent like DMF or toluene.
- Pressurize the reactor with carbon monoxide (CO) to a mild pressure range of 5-90 psig, specifically optimizing around 15 psig for balanced kinetics and safety.
- Maintain the reaction temperature between 40°C and 150°C, typically around 70°C, to facilitate the reductive cyclization and isolate the high-purity indole product (Formula II).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of stoichiometric tin chloride removes a significant cost center associated with hazardous waste disposal and regulatory compliance, as the handling and treatment of organotin waste is both expensive and environmentally taxing. By shifting to a catalytic system that operates at near-atmospheric pressures, facilities can utilize existing infrastructure without the need for costly upgrades to high-pressure autoclaves, thereby accelerating the timeline from process development to commercial launch. This flexibility ensures a more resilient supply chain capable of adapting to fluctuating market demands without being bottlenecked by specialized equipment availability.
- Cost Reduction in Manufacturing: The transition from stoichiometric reductants to a low-loading catalytic system fundamentally alters the cost structure of indole production. By reducing the palladium loading to trace levels and eliminating the purchase and disposal of bulk tin salts, the direct material costs are substantially lowered. Furthermore, the simplified workup procedure reduces solvent consumption and labor hours associated with purification, leading to a leaner and more cost-effective manufacturing process that enhances overall margin potential for the final API.
- Enhanced Supply Chain Reliability: The robustness of this catalytic method against variations in substrate structure ensures a consistent supply of high-quality intermediates. Since the reaction conditions are mild and the catalyst system is stable, the risk of batch failures due to thermal runaways or pressure excursions is minimized. This reliability allows for tighter production scheduling and more accurate forecasting, which is critical for maintaining continuity in the supply of life-saving medications dependent on these kinase inhibitor scaffolds.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the linear scalability of this gas-liquid reaction facilitates a smooth transition from pilot plant to full commercial production. The reduced environmental footprint, characterized by lower energy consumption due to moderate temperatures and the absence of heavy metal waste, aligns perfectly with modern green chemistry initiatives. This compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile, a key factor for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed technology. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is vital for making informed decisions about process adoption and vendor selection.
Q: What are the primary advantages of this palladium-catalyzed method over traditional tin-mediated reductions?
A: The novel process eliminates the need for stoichiometric amounts of tin chloride (SnCl2), which was required in older methods to achieve suitable reaction rates. This removal drastically simplifies purification, reduces heavy metal waste disposal costs, and improves the environmental profile of the manufacturing process.
Q: What are the typical reaction conditions required for this reductive cyclization?
A: The process operates under significantly milder conditions compared to prior art, utilizing low catalyst loadings (0.05-1.5 mol% Pd) and moderate CO pressures (5-90 psig). Temperatures generally range from 40°C to 150°C, with 70°C being a preferred embodiment for optimal yield and selectivity.
Q: Can this methodology be applied to complex substrates containing sensitive functional groups?
A: Yes, the catalytic system demonstrates high functional group tolerance. It successfully processes substrates containing alpha,beta-unsaturated amides, ketones, aldehydes, and various heterocyclic substitutions like methoxy- and chloro-quinolines without compromising the integrity of the final indole structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Indoles Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless and secure. We are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of substituted indoles meets the exacting standards required for pharmaceutical grade intermediates. Our commitment to quality and consistency makes us an ideal partner for long-term supply agreements.
We invite you to engage with our technical procurement team to discuss how this advanced catalytic technology can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-value chemical solutions that drive your innovation forward.
